Division of Endocrinology, Diabetes and Bone Disease
We provide the highest quality care to patients and are at the forefront of research on endocrine diseases and diabetes.
Endocrinology, Diabetes and Bone Disease
The Hilda and J. Lester Gabrilove Division of Endocrinology, Diabetes and Bone Disease provides the highest quality care to patients and is at the forefront of research on endocrine diseases and diabetes.
This specialty report highlights:
- A data-driven and personalized approach to managing weight and metabolism
- A large-scale study that suggests shared genetic architecture for PCOS diagnosis criteria
- Helping a patient pursue a passion for auto racing
- A combination of two drug classes induces robust replication in human beta cells
- Prolactin levels do not rise among transgender women treated with estrogen
- A new treatment for endometriosis stems from a Mount Sinai discovery
- Message from the Chief: Andrea Dunaif, MD
Data-Driven and Personalized Approach to Managing Weight and Metabolism
A 40-year-old patient was seeking help, with some urgency. Her type 2 diabetes was poorly controlled, and her A1C was 12.6. She was losing her vision and sensation in her feet, and her physician said she might soon be a candidate for a liver transplant. A year later, after following a personalized nutrition and exercise program recommended by Mount Sinai diabetes and obesity experts, she lost 35 pounds, her vision and sensation were significantly better, liver function was normal, and her A1C was 6.8.
This outcome—exemplary but achievable—was produced by two complementary programs, the Weight and Metabolism Management Program at The Mount Sinai Hospital and the Mount Sinai PhysioLab at Mount Sinai St. Luke’s. The programs together offer an evidence-based, personalized approach that helps identify and address internal and external factors that may be contributing to patients’ weight gain.
“As an endocrinologist, I’m attuned to making sure there are no other, secondary reasons for the weight gain—whether it is due to medications a patient is taking or hormonal issues such as thyroid dysfunction, excess cortisol production, or menopause,” says Reshmi Srinath, MD, Medical Director of the Weight and Metabolism Management Program, and Assistant Professor of Medicine (Endocrinology, Diabetes and Bone Disease), Icahn School of Medicine at Mount Sinai.
The program’s team consists of endocrinologists, nurse practitioners, nutritionists, and exercise physiologists, in collaboration with bariatric surgeons. A patient starts with a comprehensive evaluation by an endocrinologist to assess patterns of weight gain, risk factors and complications, and optimization of chronic medications to avoid those that cause weight gain. A nutritionist reviews the patient’s eating habits and develops a personalized meal plan. Patients may choose to undergo a quick assessment of their resting metabolic rate.
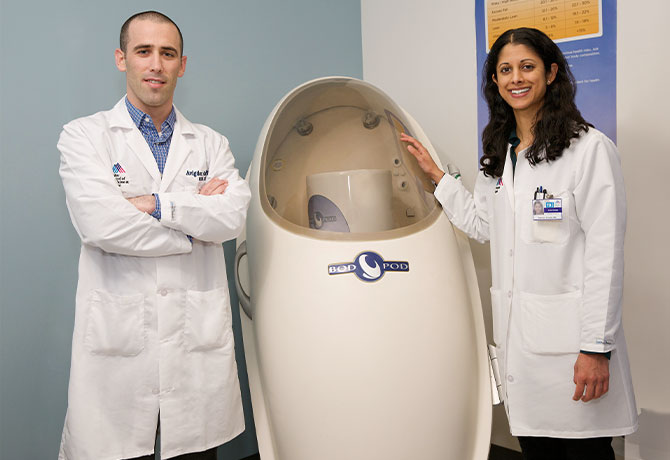
At the Mount Sinai PhysioLab, the patient is offered a uniquely comprehensive range of additional testing options, including a 24-hour metabolic chamber, one of only a half dozen in the world. The chamber was originally available only to researchers, but it is now available for clinical management and commercial use. Users range from elite athletes who want to improve their performance to patients who need to lose weight because of serious comorbidities but feel “lost,” says Avigdor Arad, PhD, RDN, CDE, Director of the Mount Sinai PhysioLab.
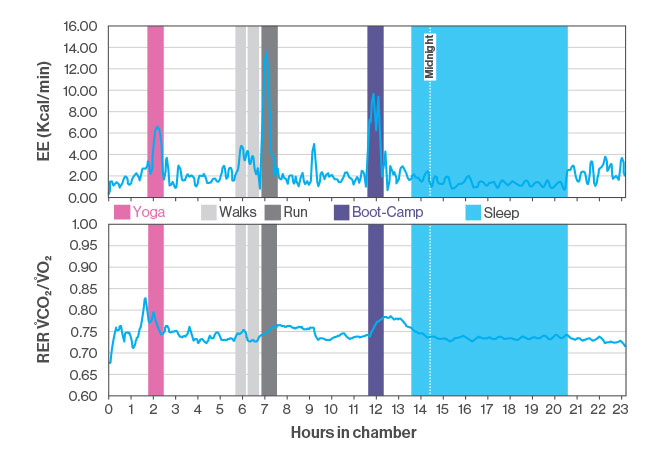
Both programs use an indirect calorimetry technique, measuring oxygen consumption and carbon dioxide production to assess precisely the number of calories the patient burns at rest and throughout the day, and therefore the exact number of calories they need to consume for weight loss, as well as how the patient uses fuel. “For example, at rest, in the fasted state, and during light-moderate exercise, stored fat should be the predominant fuel for energy production. Individuals with impaired ability to rely on fat in these circumstances might be at risk for developing obesity, diabetes, heart disease, and other metabolic disorders,” Dr. Arad says.
A comprehensive assessment at the Mount Sinai PhysioLab may include:
- Overnight Chamber, which provides what Dr. Arad calls a “gold standard” measure of total energy expenditure and oxidation profile. In a test that typically runs for 24 hours, the patient performs daily activities in a chamber that resembles a small dormitory room with a bed, phone, window, toilet, sink, bicycle, and computer. Meals and snacks are provided through a vacuum-sealed window.
- Resting Metabolic Rate Chamber, in which the patient lies down in a small room for an hour, and the number of calories burned at rest and in a fasting state is assessed.
- Exercise Testing Chamber, in which the patient runs on a treadmill or pedals a stationary bicycle, determining the number of calories burned to support the exercise bout and the fuel types the patient is using to produce energy.
- Bod Pod, in which an air-displacement plethysmograph measures the percentage of body fat and lean body mass.
If diet and lifestyle changes are not effective, physicians at the Weight and Metabolism Management Program can guide patients through medication options, Dr. Srinath says. They include liraglutide, which works on a gut hormone called GLP-1 to suppress appetite; bupropion/naltrexone, an appetite suppressant; lorcaserin, which raises serotonin levels; or a regimen of Qsymia, which has phentermine to speed the metabolism, and topiramate, a drug for migraine that suppresses appetite. Patients with a BMI > 40 also can be referred to the bariatric surgery team or to a gastroenterologist who specializes in noninvasive endoscopic procedures.
The 40-year-old patient underwent a comprehensive assessment with Dr. Arad. “Her body was unable to use fat very readily and efficiently for energy production, which might explain the weight gain and poor blood-sugar control,” he says. In the past, the patient had tried a high-fat diet paired with low-intensity exercise. Dr. Arad’s team helped her design a plan that better suited her physiology, medical condition, and preferences. She switched to high-intensity exercise—mainly cycling—and to a diet that was low fat and high in protein and fiber, with a moderate amount of low-glycemic carbohydrates. Soon after she started following the program, she began to lose weight, particularly around the belly, and her blood sugar, cholesterol, and blood pressure improved. “A year later, she is a different person,” Dr. Arad says, “much healthier physically and metabolically.”
Large-Scale Study Suggests Shared Genetic Architecture for PCOS Diagnosis Criteria
In the largest genetic analysis of polycystic ovary syndrome (PCOS) performed to date, an international consortium, including researchers from the Icahn School of Medicine at Mount Sinai, has performed a meta-analysis of genome-wide association studies (GWAS) that shows shared genetic architecture and biology for different—and competing—diagnostic criteria used to define the syndrome.
PCOS is one of the most common endocrine disorders in reproductive-age women, and it is a leading cause of infertility and type 2 diabetes. The etiology of PCOS is unknown. Accordingly, the diagnostic criteria are based on expert opinion and have been controversial. The National Institutes of Health (NIH) criteria require the presence of hyperandrogenism and ovulatory dysfunction. The Rotterdam criteria added polycystic ovarian morphology as a criterion and required two of the three criteria for the diagnosis of PCOS. This change added two additional groups—hyperandrogenism and polycystic ovarian morphology, and ovulatory dysfunction and polycystic ovarian morphology—to the diagnosis of PCOS.
The researchers explored the genetic basis of PCOS by conducting a meta-analysis of GWAS involving more than 10,000 women of European ancestry with PCOS and 100,000 controls from seven studies. These studies included 2,540 NIH-criteria cases, 2,669 Rotterdam-criteria cases, and 5,184 self-reported cases from the personal genetics company 23andMe. With the benefit of this sample size, researchers were able to identify three new susceptibility loci near the genes PLGRKT, ZBTB16, and MAPRE1 that reached genome-wide significant associations with PCOS. Further, the study replicated 11 previously reported loci. All the new loci contain genes plausibly linked to both metabolic and reproductive features of PCOS.
The study was the first to compare the genetics of PCOS identified by the NIH criteria compared to the Rotterdam diagnostic criteria. There were no differences genetically in the disease defined by these criteria or by self-report, except for one locus that was more strongly associated with the NIH criteria.

Additional analyses provided further insight into the biology of PCOS. There was evidence that the same genes contribute to PCOS, obesity, fasting insulin levels, type 2 diabetes, lipid levels, and coronary artery disease. In addition, genetic variants associated with menopause timing, depression, and premature male-pattern balding play a causal role in PCOS. The association between PCOS and male-pattern balding is particularly intriguing, since it is the first genetic evidence for shared disease biology in men. The genetic link with depression supports epidemiologic studies that have found an increased risk for depression in women with PCOS.
“One of our most important findings is simply that all the fighting that goes on in the field over which diagnostic criteria to use is unnecessary,” says Andrea Dunaif, MD, Chief of the Hilda and J. Lester Gabrilove Division of Endocrinology, Diabetes and Bone Disease at the Icahn School of Medicine at Mount Sinai, and a senior author of the study, published in PLOS Genetics in December 2018. “We can be pretty broad in our diagnosis with the criteria we have and be assured we are going to find the same genetic forms of PCOS.”
“This study also indicates the enormous power of GWAS to provide novel insights into disease biology. For the first time, we’re making real progress in understanding the causal pathways leading to PCOS and the diseases that are genetically related to it,” notes Dr. Dunaif, an international authority on the disease. Ultimately, she hopes that these biologic insights will enable the development of novel therapies for PCOS.
Helping a Patient Pursue a Passion for Auto Racing

Any patient with type 1 diabetes faces daily challenges, but Lance Bergstein has an added degree of difficulty: he is a serious amateur race car driver, and a sudden drop in blood sugar could be potentially lethal during a race. To keep him on track, a personalized approach was devised by his physician, Carol J. Levy, MD, Director of the Mount Sinai Diabetes Center, a leader in diabetes treatment and technology research.
Until recently, Mr. Bergstein followed a strict routine to maintain his blood sugar at safe levels, pricking his finger 30 minutes before each race to get a reading and then eating a snack to raise his glucose level just enough to stay within the normal range of 120 mg/dL to 150 mg/dL for the duration of the competition. If his blood sugar dropped too low, it could affect his brain function and impair his judgment, but high blood sugar levels could lead to other problems, including blurry vision and a greater risk of long-term complications, such as blindness, kidney damage, and stroke. Working closely together, Dr. Levy and Mr. Bergstein decided that diabetes-based technology, including the Dexcom G6 continuous glucose monitor (CGM), is the best method to monitor his blood glucose level as he competes in races that last as long as 24 hours.
Mount Sinai was part of a priority-access program for patients to use the G6 before it became more widely available; the device was approved by the U.S. Food and Drug Administration in March 2018. Dr. Levy, who herself has had type 1 diabetes since childhood, was one of the earlier clinicians to wear one of the new monitors. “Any person who has type 1 diabetes is basically continually walking on a tightrope, trying to maintain their blood sugar in a normal range,” says Dr. Levy, Associate Professor of Medicine (Endocrinology, Diabetes and Bone Disease), Icahn School of Medicine at Mount Sinai.
The G6 continuous glucose monitor, she says, represents a significant advance over previous iterations, because it does not require finger sticks for calibration, and it lets patients track blood sugar levels to determine whether the level is stable, rising, or dropping in real time. The results can be transmitted wirelessly every few minutes to a smartphone. Alarms are set to warn patients if their levels are too high or too low. The tiny sensor filament is inserted into the abdomen with an applicator and is worn for 10 days. The monitor also allows for the data to be shared with up to five other people.
In the case of Mr. Bergstein, who races Porsches for teams in New York and in Florida, the phone with the readout is mounted on the steering wheel, and if he needs to make an adjustment, he sips orange juice or water from straws threaded through his helmet. “Being able to keep my blood sugar in range with more ease and precision has completely changed my life,” he says.
Dr. Levy points out that not every patient who comes to the Diabetes Center wants to use every new piece of technology that comes along. “We have to find the place that we can meet the patient from a safety perspective at their level of comfort,” she says. Still, she adds, diabetes treatment has come a long way over the last decade, and patients and health care professionals are enthusiastic about the newest advances. “It’s exciting to be part of this era of care for patients with diabetes,” she says, “and to continue to work with patients both in clinical care and in moving forward the development of devices for disease management, such as glucose sensors and other diabetes-management technology.”
Combination of Two Drug Classes Induces Robust Replication in Human Beta Cells

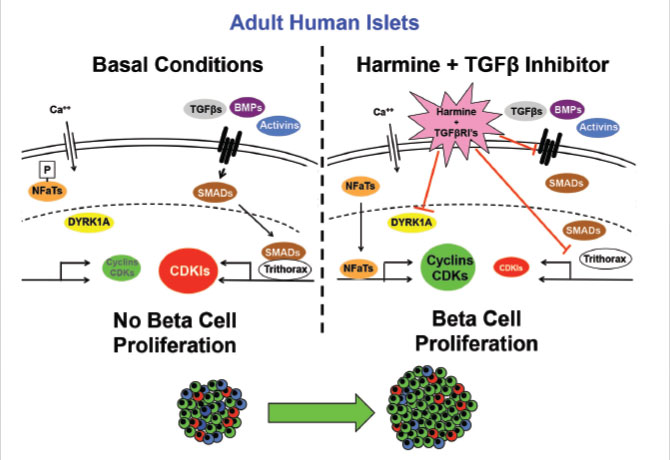
Researchers at the Icahn School of Medicine at Mount Sinai have discovered a novel combination of two classes of drugs that induces the highest rate of proliferation ever observed in adult human beta cells—the cells in the pancreas that produce insulin. The finding involved one drug that inhibits the enzyme dual specificity tyrosine-regulated kinase 1A (DYRK1A) and another that inhibits transforming growth factor beta superfamily (TGFβSF) receptors. Together, they caused the cells to proliferate robustly, generating a labeling index of 5 to 8 percent.
“For the first time, we are able to see rates of replication that are sufficient to replenish beta cell mass in human beings,” says Andrew F. Stewart, MD, Director of the Diabetes, Obesity and Metabolism Institute and the Irene and the Dr. Arthur M. Fishberg Professor of Medicine (Endocrinology, Diabetes and Bone Disease) at the Icahn School of Medicine. Dr. Stewart was lead author of the study, which was published in December 2018 in the journal Cell Metabolism.
The paper builds upon two earlier studies by Dr. Stewart and his team. One of them, published in Nature Medicine in 2015, showed that the drug harmine drove sustained division and multiplication of adult human beta cells in culture. The harmine treatment led to normal control of blood sugar in mice whose beta cells had been replaced with human beta cells. However, the proliferation rate was 1.5 to 3 percent, lower than needed to rapidly expand beta cells in people with diabetes. The second study, published in 2017 in Nature Communications, conducted the largest genomic study of insulinomas—benign pancreatic tumors that secrete insulin—and uncovered multiple pathways to human beta cell proliferation.
In the 2018 study, Dr. Stewart and his team explored a suggestion in the “insulinoma genomic recipe” that a combination of two classes of drugs—a DYRK1A inhibitor such as harmine with a TGFβSF inhibitor drug—would be able to synergistically increase beta cell regeneration. This proved to be true. The study also found that this was a class effect, achieved by many different DYRK1A inhibitors and many different TGFβ superfamily inhibitors.
“Beta cell regeneration is a ‘holy grail’ for the treatment of diabetes,” says Peng Wang, PhD, Associate Professor of Medicine (Endocrinology, Diabetes and Bone Disease), Icahn School of Medicine at Mount Sinai, and first author on the study. “The drug combination that we have identified can induce beta cell proliferation at rates that are likely to be effective in people with type 1 and type 2 diabetes.” The National Institutes of Health and the JDRF Beta Cell Regeneration and Survival Program supported the study.
The drug combination is not without its hurdles. The drugs have effects on other organs in the body, and at the moment there is no molecule that is able to deliver a drug specifically to the beta cell. The study envisioned possible solutions, including transport molecules such as beta cell specific GLP1 analogues or monoclonal antibodies. “We have the packages to deliver,” Dr. Stewart says, “but now we need a courier system to deliver them to the exact beta cell address.”
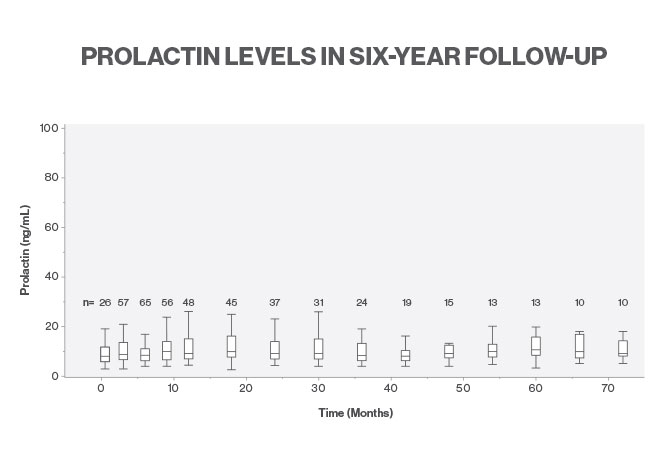
Prolactin Levels Do Not Rise Among Transgender Women Treated With Estrogen
Levels of prolactin, the hormone associated with milk production, did not rise significantly in transgender patients undergoing the feminizing hormone therapy treatment of estradioladministered with the antiandrogen spironolactone, according to a novel study led by a physician-scientist at the Icahn School of Medicine at Mount Sinai. The findings indicate that gender-affirming hormone treatment for transgender women is significantly safer than was previously thought.

The Standards of Care for transgender women, issued by the World Professional Association for Transgender Health, currently suggests that endocrinologists closely monitor prolactin levels in patients receiving hormone therapy. “Slight elevations in prolactin can be a sign of more concerning disease processes involving the brain or nearby structures,” says Joshua D. Safer, MD, Executive Director of the Mount Sinai Center for Transgender Medicine and Surgery. “So, when we do find elevated prolactin levels, we’re obligated to look for more serious problems.”
The study was the first to follow prolactin levels over time in transgender patients receiving the hormone regimen that is standard in the United States. In the retrospective study, which was published in July 2018 in the journal Endocrine Practice, researchers reviewed the medical records of 98 patients at Boston Medical Center who were receiving trans feminine hormone therapy. In some cases, as many as six years of data were available to Dr. Safer and his co-authors, Kelly J. Chan and Jason R. Bisson. In the cohort studied, 79 percent of patients were on oral estradiol, 19 percent were on conjugated equine estrogens, and 2.5 percent received transdermal estradiol.
Despite the extended exposure to high doses of estrogens and testosterone-lowering medication, the study found no statistically significant relationship between estradiol and prolactin levels among the patients. In addition, none of the patients were diagnosed with prolactinoma, benign but hormone-producing tumors on the pituitary gland.
The study adds to the evidence-based literature in transgender medicine and suggests that prolactin could be removed from the list of hormone levels to be monitored in the Standards of Care, which are to be revised in 2019. “Our study included about 100 women and looked at their data points over about six years,” Dr. Safer says. “These are real data, as opposed to a bunch of experts in a room saying, ‘Estrogen is known to stimulate prolactin so maybe we should check it, just to be safe.’”
The National Institutes of Health made a program announcement in January 2018 declaring the study of transgender medicine to be a priority. “Things are moving forward as a whole, just very slowly,” Dr. Safer says.
New Treatment for Endometriosis Stems From a Mount Sinai Discovery
A drug that recently received approval from the U.S. Food and Drug Administration (FDA) for the treatment of pain associated with the gynecological disorder endometriosis had its genesis two decades ago in the laboratory of Stuart C. Sealfon, MD, at the Icahn School of Medicine at Mount Sinai.

The drug, Orilissa™, approved by the FDA in July 2018, is the first orally active gonadotropin-releasing hormone (GnRH) antagonist. It specifically helps to ease the moderate to severe pain that accompanies endometriosis as tissue that forms in the lining of the uterus also grows outside the uterus. In the disorder, which affects roughly one in ten women of reproductive age, the excess tissue growth is often accompanied by pain during menstruation, intercourse, or urination.
“Orilissa is a drug that resulted from the basic research we conducted at Mount Sinai, and it will help millions of women,” says Dr. Sealfon, the Sarah B. and Seth M. Glickenhaus Professor and Chair Emeritus of the Department of Neurology. “At Mount Sinai, we discovered how to clone the drug target that was needed to develop this new drug.”
More than two decades ago, Dr. Sealfon led the Mount Sinai team that cloned the gonadotropin-releasing hormone receptor (GnRHR) and genetically engineered host cells that express GnRHR. GnRH, which is secreted by the hypothalamus, plays a key role in controlling reproduction and acts via its receptor, GnRHR. In 1992, Dr. Sealfon authored a paper describing the cloning procedure of the receptor in the journal Molecular Endocrinology, and the next year, his paper on the receptor’s primary structure was published in Molecular and Cellular Endocrinology. This research provided a better understanding of the interplay of hypothalamic, pituitary, and gonadal hormones, which underlie pharmacotherapy and the reproductive system. Two U.S. patents, in 1998 and 1999, assigned these inventions to Mount Sinai.
Orilissa—also known by its generic name, elagolix—is an oral pill taken daily in doses of either 150 mg or 400 mg. It enables women to dial down the reproductive system, suppressing the luteinizing hormone and the follicle-stimulating hormone, which leads to decreased blood concentrations of estradiol and progesterone. This reduces the growth of excess tissue, or lesions, that form on the ovaries, fallopian tubes, or areas near the uterus, including the bowel and bladder, that characterize endometriosis and cause pain.
The 20 years it took for elagolix to move from Dr. Sealfon’s laboratory to the marketplace demonstrates the length of time it can take for basic scientific discoveries to bear fruit. The drug was released by AbbVie, a global pharmaceutical company, in cooperation with Neurocrine Biosciences, Inc. In two phase III clinical trials, Orilissa has also been shown to be helpful in the treatment of uterine fibroids, which causes bleeding or pain in millions of women, and for which there are currently only limited nonsurgical treatment options.
Message from the Chief: Andrea Dunaif, MD
I’m exceptionally proud that 2018 has been another exciting year for research and clinical care in the Division of Endocrinology, Diabetes and Bone Disease at the Icahn School of Medicine at Mount Sinai. Andrew F. Stewart, MD, and his team have continued their innovative studies of human beta cell regeneration. Most recently, they have reported that a novel combination of two classes of drugs induces the highest rate of proliferation ever observed in adult human beta cells.

I have been one of the lead investigators in an international consortium that has conducted the largest genetic analysis of polycystic ovary syndrome (PCOS). This research has provided important new insights into the underlying biology of PCOS and shown that there are no genetic differences between PCOS diagnosed according to the National Institutes of Health criteria compared to cases diagnosed according to the Rotterdam criteria. Joshua D. Safer, MD, and colleagues reported that prolactin levels did not increase in transgender patients undergoing feminizing hormone therapy, adding to the evidence base in transgender medicine. An important treatment for endometriosis was approved by the U.S. Food and Drug Administration, based on foundational research by Stuart C. Sealfon, MD.
In the clinical arena, the Weight and Metabolism Management Program at The Mount Sinai Hospital and the Mount Sinai PhysioLab at Mount Sinai St. Luke’s helped patients meet their health goals with unique protocols informed by precise metabolic monitoring. Carol J. Levy, MD—with a combination of advanced devices and personalized care—helped a patient with type 1 diabetes pursue his dream of racing high-performance cars. And The Mount Sinai Hospital was again nationally ranked by U.S. News & World Report in diabetes and endocrinology. Building on the momentum generated by the past year’s accomplishments, we look forward to a new year of innovation and discovery.
Andrea Dunaif, MD, Chief of the Hilda and J. Lester Gabrilove Division of Endocrinology, Diabetes and Bone Disease, and the Lillian and Henry M. Stratton Professor of Molecular Medicine, Icahn School of Medicine at Mount Sinai