Mount Sinai Heart
Mount Sinai Heart is celebrated internationally as a leader in all facets of cardiology care, cardiac surgery, and advanced research.
Mount Sinai Heart
Our team of award-winning physicians has invigorated the science of cardiovascular medicine, pioneering treatments for arrhythmias, coronary artery disease, heart failure, valvular disease, and vascular disease.
This specialty report highlights:
- FREEDOM study confirms therapy for patients with diabetes and multivessel disease
- Mount Sinai Heart earns Joint Commission certification in Comprehensive Cardiac Care
- FAMILIA trial demonstrates child health intervention in underserved communities
- Multicenter APOLLO trial evaluates options for patients with severe mitral regurgitation
- NIH hybrid trial compares two minimally invasive treatments for multivessel disease
- Developing a machine learning model to predict response to high-dose statins
- Cardiac Catheterization Laboratory demonstrates complex coronary cases online
- Successful human trial treats atrial fibrillation with pulsed field ablation
- AVIATOR 2 study finds discrepancy between perceived and empiric risks after PCI
- Review finds two PCSK9 inhibitors safe and effective for high-risk patients
- Significant gains in a next generation of LVAD
- Three-dimensional heart organoids signal a step forward in drug testing
- Aerosol-delivered gene therapy shows promise in treating pulmonary hypertension
- Message from the Director: Valentin Fuster, MD, PhD
Cardiac Care: FREEDOM Study Confirms Therapy for Patients With Diabetes and Multivessel Disease
A long-term follow-up to the landmark FREEDOM trial is helping to establish an optimal standard of care for patients with diabetes and multivessel coronary artery disease. The FREEDOM Follow-On Study, led by researchers at Mount Sinai Heart, found that patients in this high-risk population who are treated with coronary artery bypass grafting (CABG) survived significantly longer than those treated with percutaneous coronary intervention (PCI) with drug-eluting stents.
Over a follow-up period of 7.5 years, all-cause mortality was 18.7% for the CABG group and 23.7% for the PCI cohort. Moreover, the study found that younger patients fared the best, with the difference most significant for those under age 63. “These data support current recommendations that CABG be considered the preferred strategy for patients with diabetes and multivessel disease,” says the study’s principal investigator, Valentin Fuster, MD, PhD, Director of Mount Sinai Heart and Physician-in-Chief of The Mount Sinai Hospital. The FREEDOM Follow-On Study, published in the Journal of the American College of Cardiology in November 2018, tracked 943 patients. It is the successor report to the FREEDOM (Future Revascularization Evaluation in Patients with Diabetes Mellitus: Optimal Management of Multivessel Disease) trial, which randomized 1,900 patients with diabetes.
That original study, which concluded in 2012, showed that revascularization through CABG for patients with diabetes and multivessel coronary artery disease resulted in a significantly reduced risk of all-cause mortality, myocardial infarction (MI), and stroke compared to treatment with PCI. The median follow-up time of the trial, 3.8 years, was considered relatively short because of the prolonged nature of atherosclerotic cardiovascular disease. Consequently, Mount Sinai developed a longer-term, follow-on study, and 25 of the original international centers agreed to participate.
Adults with diabetes are two to four times more likely to die from heart disease than those without it, according to the American Heart Association. The FREEDOM Follow-On Study is the first to demonstrate the long-term mortality benefit of CABG compared to PCI in patients with diabetes and to show that the greatest benefits accrue to patients under 65. It is expected to add to the body of evidence that CABG plus guideline-directed medical therapy is the optimal treatment pathway for this vulnerable population.
An editorial that accompanied publication of the study suggested that a newer generation of ultra-thin-strut drug-eluting stents—which have shown reductions in death and MI through decreases in stent thrombosis and restenosis—might narrow the mortality gap between CABG and PCI. In addition, newer classes of type 2 diabetes drugs that not only impact blood glucose levels but can markedly reduce cardiovascular deaths and events are starting to reconfigure the medical landscape. These therapies include sodium glucose co-transporter 2 (SGLT2) inhibitors and GLP-1 receptor agonists. While acknowledging how much therapies have evolved since FREEDOM, Dr. Fuster says those changes do not appear to be sufficient to bridge the mortality gap between PCI and CABG.
The results also underscore the importance of aggressive medical treatment and prevention to keep patients from reaching advanced stages of diabetes, hypertension, and hypercholesterolemia—forestalling the need for either CABG or PCI, he says. “We have to pay attention to how we can prevent this late stage of disease by taking care of obesity,” Dr. Fuster says. “About 70% of these patients were obese, and we can do a lot to prevent this.”
Cardiac Care: Mount Sinai Heart Earns Joint Commission Certification in Comprehensive Cardiac Care

Mount Sinai Heart has earned The Joint Commission’s Comprehensive Cardiac Center (CCC) certification, becoming one of only four cardiac organizations in the United States—and the only one on the East Coast—to hold the honor. The certification indicates consistent patient care and comprehensive excellence, and recognizes compliance with The Joint Commission’s strict national standards.
“Mount Sinai Heart is honored to receive this award from The Joint Commission that recognizes our outstanding center and quality of care,” says Valentin Fuster, MD, PhD, Director of Mount Sinai Heart and Physician-in-Chief of The Mount Sinai Hospital. “We are proud of our exceptional cardiology and cardiovascular services and will continue to offer superior treatment to patients across the world.”
The advanced CCC certification has been offered since 2017 by The Joint Commission, exclusively to hospitals with a comprehensive service line in cardiology and cardiovascular services. To earn the two-year certification, Mount Sinai Heart underwent a rigorous two-day on-site survey at The Mount Sinai Hospital in November 2018, in which two reviewers from The Joint Commission evaluated performance in key areas, including: management of heart disease, acute myocardial infarction, percutaneous coronary interventions, coronary artery bypass graft surgery, cardiac valve disease, dysrhythmias, and heart failure; transition of care from the Emergency Department to diagnosis, treatment, and outpatient follow-up; collaboration among disciplines; cardiac rehabilitation of patients; training of staff; identification of risk factors and prevention of cardiac disease; and the use of data collection to monitor and measure outcomes.
Preparing for the visit involved a vast collaborative effort. “We met every week for 10 months, making sure all of our policies and procedures are up to date,” says Beth Oliver, DNP, RN, Senior Vice President of Cardiac Services, Mount Sinai Health System. “And we had tremendous support from physicians and their teams.” They included Cardiovascular Surgery, led by David H. Adams, MD, Cardiac Surgeon-in-Chief of the Mount Sinai Health System; the Emergency Department; the Quality Assurance Department; the Cardiac Catheterization Laboratory, and many others throughout the hospital. “This accomplishment emphasizes and validates our commitment to providing exceptional care to our patients,” says David L. Reich, MD, President and Chief Operating Officer of The Mount Sinai Hospital.
CCC certification is a voluntary program for hospitals with extensive cardiac care services seeking to evaluate and advance patient care across an array of cardiac domains, according to The Joint Commission. “Developed in partnership with a technical advisory panel of clinicians with specific expertise in the field, and with input from the American Heart Association, the program is designed to recognize hospitals demonstrating consistent, comprehensive excellence,” the Commission says.
The Joint Commission certification follows another honor for Mount Sinai Heart, which each year cares for 65,000 patients. The Cardiac Intensive Care Unit at The Mount Sinai Hospital has received the gold-level Beacon Award for Excellence from the American Association of Critical-Care Nurses. This award honors units that excel in every facet of patient care. Mount Sinai’s is the only cardiac intensive care unit in New York State to have this recognition.
Global Health: FAMILIA Trial Demonstrates Child Health Intervention in Underserved Communities

FAMILIA, a cluster-randomized trial developed by Valentin Fuster, MD, PhD, Director of Mount Sinai Heart and Physician-in-Chief of The Mount Sinai Hospital, has demonstrated that a school-based education intervention is an effective strategy for instilling healthy behaviors among preschoolers, according to an abstract presented in November 2018 at the American Heart Association Scientific Sessions.
The “Family-Based Approach in a Minority Community Integrating Systems-Biology for Promotion of Health” (FAMILIA) trial enrolled 600 families in Harlem, including 562 children ages three to five, over the last four years. It was intended to promote cardiovascular health among children while reducing their chances of developing risk factors for heart disease.

“There is good data showing that part of our behavior as adults develops between ages three and five,” says Dr. Fuster, a pioneer in the study of atherosclerotic disease. “If this age is so important, why wouldn’t we use this window of opportunity to teach children to make health a priority for the rest of their lives?”
Funded by a $3.8 million grant from the American Heart Association, FAMILIA is based on health interventions that Dr. Fuster developed in Bogota, Colombia, and Spain. Like those programs, FAMILIA is exploring how a child’s behavior, environment, and genetics may lead to heart disease, with the goal of reducing the risk of obesity, heart attack, stroke, and type 2 diabetes by creating a family-based “culture of health.”
The specific objective of the FAMILIA trial was to “assess the impact of a preschool-based health promotion education intervention in an underserved community.” It enrolled children in 15 Head Start preschools, forming a cohort that was 51% female, 54% Hispanic/Latino, and 37% African American. Children were randomized 3:2 to receive either their school’s normal curriculum (control) or to receive 50 hours of heart-health educational intervention over four months. At the start, each child was interviewed by a team member with experience in early childhood education, using tools that were pictorial and structured like an interactive game.
Based on the results, children were given a KAH (knowledge, attitudes, and habits) score. The children who received intervention learned lessons including how the heart works; how to select healthy foods; how to regulate their emotions; and how to stay physically active and encourage their families to be active, too.
After four months, researchers interviewed the preschoolers again. The primary outcome was the change from baseline in the KAH score of the child. As secondary outcomes, researchers evaluated the changes in KAH subcomponents. Linear mixed-effects models were used to test for intervention effects. Researchers found that the overall KAH score rose 11.8% from baseline in the intervention group, compared with 5.5% in the control group. The average difference in the change of KAH between the intervention and control groups was 2.9 points. Changes in physical activity and understanding of the human body and heart were the main drivers of this effect, researchers said. As part of a long-term vision, the team plans to conduct follow-ups of the children after five and ten years to assess the sustainability of the intervention effects.
FAMILIA also includes a parallel program for the parents and caregivers. Some adults meet in small groups to help each other get healthier through peer support, while others receive individualized lifestyle counseling and a personal-activity monitoring device. Results from that program are expected in late 2019. “What we are finding is a significant benefit in all respects,” Dr. Fuster says of both adults and children in FAMILIA. “Their knowledge, their attitude, and their habits are quite positive, and this is very exciting.”
Cardiovascular Surgery: Multicenter APOLLO Trial Evaluates Options for Patients With Severe Mitral Regurgitation
Participation in a new U.S. Food and Drug Administration pivotal clinical trial is underway at The Mount Sinai Hospital, which could result in an alternative to open-heart mitral valve replacement surgery. The APOLLO trial, which began in October 2017, is a multicenter, global, prospective, randomized trial that is evaluating the safety and efficacy of the Medtronic Intrepid™ Transcatheter Mitral Valve Replacement (TMVR) system in patients with severe mitral regurgitation who are candidates for conventional mitral valve replacement surgery.

David H. Adams, MD, the Marie-Josee and Henry R. Kravis Professor and Chair of Cardiovascular Surgery at the Icahn School of Medicine at Mount Sinai, and the Cardiac Surgeon-in-Chief of the Mount Sinai Health System, is the national co-investigator for the study, along with Martin Leon, MD, of New York-Presbyterian/Columbia University Irving Medical Center.
“This system is unique in that it is a self-expanding stent technology that requires no sutures, tethers, or anchors to attach and conform to a patient’s native mitral annulus,” Dr. Adams says. “It enables a rapid catheter-based deployment of the prosthesis through a cardiac puncture, which we believe will benefit patients because it eliminates the invasiveness of a sternotomy or the use of a heart-lung machine.”
Up to 1,380 participants will be enrolled in the APOLLO study, with patients randomized on a 1:1 basis to undergo TMVR implantation or standard-of-care mitral valve surgery. Patients are eligible if they are 18 years old or older and have moderate to severe symptomatic regurgitation, and a CT scan confirms they are suitable candidates for bioprosthetic mitral valve replacement. Exclusion factors include predicted risk of mortality due to comorbidities, previous mitral valve surgery, and a mitral anatomy that precludes management of the subvalvular apparatus or full chordal sparing. There will also be a single-arm cohort for patients who are ineligible for conventional mitral valve surgery.
“TMVR is a rapidly growing field with a range of emerging devices and approaches,” says Ahmed El-Eshmawi, MD, Assistant Professor of Cardiovascular Surgery at the Icahn School of Medicine. “This trial will enable us to compare TMVR versus standard surgery. It also enables us to study the high-risk patient population in the single arm, and we expect it will result in invaluable information regarding the periprocedural risks and clinical outcomes of TMVR, which hopefully will one day mirror the outstanding results now achieved with transcatheter aortic valve replacement.”
Dr. El-Eshmawi says the primary endpoint for the APOLLO study is the composite of all-cause mortality, stroke, reoperation, and cardiovascular hospitalization at one year. Patients will be monitored after the procedure, starting with a CT scan after 30 days to ensure the valve is well-seated on the annulus. “Because TMVR is truly minimally invasive, we expect there should be a more rapid recovery, with decreased morbidity for patients who receive it,” he says. “It may also facilitate valve treatment in high-risk patients in the future.”
The trial is continuing to enroll patients and has a completion date of 2021, and Dr. El-Eshmawi is optimistic about the potential of TMVR: “Being a leading Mitral Valve Repair Reference Center, Mount Sinai will continue to pursue therapeutic advances that offer our patients the safest and most effective options for treatment.”
For more information on enrollment, call: 212-659-6820
The Icahn School of Medicine at Mount Sinai receives royalty payments from Edwards Lifesciences and Medtronic related to Dr. Adams’s intellectual property.
Coronary Revascularization: NIH Hybrid Trial Compares Two Minimally Invasive Treatments for Multivessel Disease
An international clinical trial, led by a team at Mount Sinai, could answer longstanding questions in treatment of cardiovascular disease by comparing the safety and effectiveness of two well-established treatments for the 15% of heart patients who have a specific pattern of multivessel coronary artery disease (CAD).

The Hybrid trial, launched in October 2017, is a multicenter, prospective, randomized trial that is comparing hybrid coronary revascularization (HCR)—a combination of minimally invasive coronary bypass surgery and percutaneous coronary intervention (PCI)—versus PCI with drug-eluting stents alone. HCR and PCI are alternatives to open-heart bypass surgery, both providing a shorter recovery time and avoiding sternotomy. The Hybrid trial, funded by the National Institutes of Health (NIH), is using rigorous scientific methods to evaluate the outcomes for the two procedures over five years of planned follow-up.
“This might be a new paradigm in the treatment of coronary disease,” says the principal investigator of the trial, John D. Puskas, MD, Chair of Cardiovascular Surgery at Mount Sinai St. Luke’s, and Director of Surgical Coronary Revascularization for the Mount Sinai Health System.
“Before HCR was introduced, when a patient with multivessel coronary artery disease needed an intervention, the options were either PCI with stents or open surgery. HCR is in between, a combination of the less invasive stenting with the more durable internal mammary artery bypass.” The trial’s co-principal investigator is Gregg Stone, MD, of the Columbia University Vagelos College of Physicians and Surgeons. The Data Coordinating Center for the trial is being led by Emilia Bagiella, PhD, and Alan J. Moskowitz, MD, of Mount Sinai’s International Center for Health Outcomes and Innovation Research (inCHOIR).
The current study was preceded by the NIH-sponsored Hybrid observational trial, an 18-month non-randomized trial also led by Dr. Puskas, which followed the outcomes of 300 patients who received HCR or PCI at 11 sites in the United States and established the feasibility of a prospective trial. Its results were published in July 2016 in the Journal of the American College of Cardiology.
The multicenter, randomized Hybrid trial—a broader, longer-term study—aims to enroll 2,354 patients aged 18 and older who have coronary disease involving the left anterior descending (LAD) artery and/or left main coronary artery, with at least one other coronary artery requiring treatment. Subjects will be randomized 1:1, with one group receiving PCI with drug-eluting stents. The other patients will undergo HCR, which involves grafting the left internal mammary artery (LIMA) to the LAD in one stage, and in a second stage performing PCI with drug-eluting stents for the remaining blocked non-LAD vessels.
The primary endpoint is the occurrence of major adverse coronary and cerebrovascular events (MACCE)—including death, myocardial infarction, stroke, or revascularization. The secondary objectives are to determine the impact of HCR compared to PCI on health status and quality of life. Follow-up after the procedure will be conducted at 30 days, six months, and at six-month intervals over five years. Data will primarily be collected via phone by the trial’s Data Coordinating Center and will primarily focus on patient-reported MACCE and quality of life.
Enrollment will end in March 2020, and the estimated completion date of the study is March 2024. As of January 2019, approximately 200 patients were enrolled at 35 sites in the United States. The NIH and Hybrid trial leadership are recruiting an additional 50 clinical sites in other nations, including Germany, England, Israel, and Poland.
“Patients generally prefer a less invasive procedure to a more invasive procedure. Of course, they also want a durable procedure,” says Dr. Puskas, a leader in coronary artery revascularization. “If hybrid therapy—this middle-of-the-road approach—will give patients the long-term benefit of the bypass graft without the large incision, then maybe this is something that a significant portion of patients will choose to have in the future.”
For more information on enrollment, go to: hybridtrial.org
Coronary Revascularization: Developing a Machine Learning Model to Predict Response to High-Dose Statins
A machine learning model has been developed to predict which individuals will undergo changes in plaque morphology—specifically a thickening of the fibrous cap—that are associated with improved cholesterol transport following high-intensity statin therapy. The research is part of the ongoing YELLOW studies of high-dose statins, led by Annapoorna S. Kini, MD, Director of the Cardiac Catheterization Laboratory and the Zena and Michael A. Wiener Professor of Medicine at the Icahn School of Medicine at Mount Sinai.
Since 2013, Dr. Kini has been conducting YELLOW (Reduction in Yellow Plaque by Aggressive Lipid-Lowering Therapy) studies regarding the use of high-dose statins (rosuvastatin). Her team has found that some patients treated with intensive statin therapy exhibited significant changes in plaque morphology, a finding that they say identifies a potential independent link between fibrous cap thickening (FCT) and improved cholesterol efflux capacity.
In the substudy, Dr. Kini says, “we created a model to understand these people whose fibrous cap thickened,” using transcriptomic data from their clinical trial of rosuvastatin to better understand the mechanisms underlying the extensive data demonstrating the beneficial effects of statins.
FCT is typically measured with optical coherence tomography (OCT), which requires coronary artery catheterization. The machine learning substudy could lead to a less invasive tool that will help determine personalized dosages for patients, says Dr. Kini, who presented the study at the American College of Cardiology conference in March 2018. Physicians often encounter patients who take high-dose statins and suffer adverse clinical events, despite improvements in LDL cholesterol levels, she says. “These patients complain, ‘Doctor, I’m doing everything right. I’m eating right. I’m doing exercise. I’m taking medication, but still I develop a blockage,’” Dr. Kini says. In search of an explanation, she and her team looked for biomarkers, comparing the gene expression of those who responded well to high-dose statins with the gene expression of those who did not.
FCT was measured using OCT in 69 patients at baseline and after 8 to 12 weeks of 40 mg rosuvastatin therapy. Researchers used two machine learning models—binomial elastic net regression and K-top-scoring-pairs (KTSP)—to analyze gene expression data using FCT response as the dependent variable. Both the elastic net and KTSP models were able to predict FCT responders with high accuracy. The elastic net model, using 73 genes, had an accuracy of 92.8%, sensitivity of 94.1%, and specificity of 91.4%. The KTSP model, using 18 genes, had an accuracy of 95.7%, sensitivity of 94.3%, and specificity of 97.1%.
“We could do all this in just a blood test,” Dr. Kini says of a longer-term goal. “That way we could personalize the medicine to determine who will respond to statins, who will not, and what other medicine will work on them. Is there any other medicine that will work? That is the research we are doing now.”
Coronary Revascularization: Demonstrating Complex Coronary Cases Online
Complex Coronary Cases, a live webcast from The Mount Sinai Hospital’s Cardiac Catheterization Laboratory, is in its tenth year of demonstrating therapeutic options for structural heart disease. The monthly series, which has been viewed more than 650,000 times in 138 countries, was founded in 2009 by Samin K. Sharma, MD, Director of Clinical and Interventional Cardiology at The Mount Sinai Hospital, and the Anandi Lal Sharma Professor of Medicine in Cardiology at the Icahn School of Medicine at Mount Sinai; and Annapoorna S. Kini, MD, Director of the Cardiac Catheterization Laboratory, and the Zena and Michael A. Wiener Professor of Medicine at the Icahn School of Medicine.

In real time, viewers can participate in an online discussion with a moderator as they are guided through a complex procedure by Dr. Kini and Dr. Sharma, leaders in percutaneous coronary intervention (PCI). The New York State Department of Health in 2018 awarded the highest “two star” safety rating to Dr. Sharma, Dr. Kini, and the Cardiac Catheterization Laboratory for PCI, marking the 20th consecutive year that the laboratory or its physicians have been recognized for safety rates significantly exceeding the statewide average. In the 115th program, in January 2019, Dr. Sharma and Dr. Kini treated an 86-year-old woman with chronic stable angina and severe calcific bifurcation disease. The patient successfully underwent revascularization of the complex calcific left main bifurcation using rotational atherectomy with intravascular ultrasound guidance.
“The purpose of the live webcasts is to educate interventional cardiologists, of all levels of expertise, to confidently treat structural heart disease,” Dr. Sharma says. “It is a multidisciplinary forum to discuss all available medical, surgical, and interventional treatment paradigms.”
To view webcasts, go to: ccclivecases.org/new/live-webcast
Cardiac Electrophysiology: Successful Human Trial Treats Atrial Fibrillation With Pulsed Field Ablation
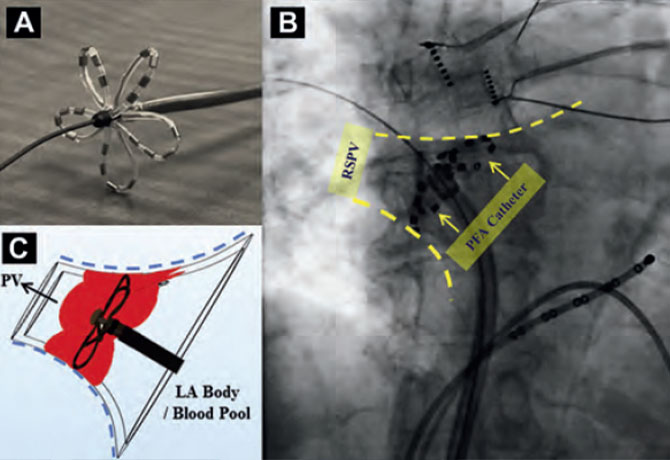
For the first time, treatment of atrial fibrillation with pulsed electrical fields was successfully applied in a human trial to isolate pulmonary veins (PV) during either endocardial catheter ablation or epicardial surgical ablation. The clinical trial was headed by Vivek Y. Reddy, MD, Director of Cardiac Arrhythmia Services, and the Leona M. and Harry B. Helmsley Charitable Trust Professor of Medicine in Cardiac Electrophysiology at the Icahn School of Medicine at Mount Sinai.

The pulsed field ablation (PFA) method avoids risk to nontarget tissue and eliminates the risk of coagulative necrosis while simultaneously offering rapid ablation with minimal complications, Dr. Reddy reported in the August 2018 Journal of the American College of Cardiology (JACC). The data were also presented at the Late Breaking Clinical Trial Sessions of the Heart Rhythm Society’s 39th Annual Scientific Sessions in May 2018. “Pulsed field ablation was acutely safe and feasible using both percutaneously delivered endocardial and surgically placed epicardial catheters,” says Dr. Reddy, lead author of the JACC article. “Our data could help usher in a new era of tissue-specific, ultra-rapid ablation of atrial fibrillation.”
Researchers conducted the trial at clinical centers in Prague, Czech Republic, and Bordeaux, France, with 22 patients (12 male and 10 female), using both of the delivery systems made by Iowa Approach Inc. One system used an epicardial linear catheter for PV, plus left atrium (LA) encircling, to generate a posterior box lesion during concomitant cardiac surgery. The other utilized an endocardial over-the-wire catheter for percutaneous transeptal PV isolation. Endocardial LA-PV voltage maps were created before and after encirclement in all cases, using either the Boston Scientific Rhythmia™ or Biosense Webster CartoR electroanatomical mapping systems.
The 22 patients were ablated under general anesthesia: seven epicardial patients during valve or coronary artery bypass grafting surgery, and 15 endocardial patients, each of whom was experiencing paroxysmal atrial fibrillation. Box lesions were successful in six of the seven epicardial patients. The only failure was related to equipment malfunction. The catheter PV isolation approach was successful in all 15 patients. There were no complications, and total PFA energy delivery time was as short as 60 seconds per patient.
Ablation is the most commonly used method for the catheter treatment of cardiac arrhythmias. Traditional thermal energy sources ablate all tissue types indiscriminately and depend on conductive heating or cooling. In contrast, PFA does not employ thermal energy, but instead ablates through the creation of microscopic pores in cell membranes, a process known as electroporation. This technology is not new. However, instead of delivering a single very high voltage pulse, from 900 to 2,500 V, this method is unique in delivering multiple, ultra-rapid millisecond pulses, as in the treatment of cancer patients.
PFA is highly tissue-specific, making it ideal for ablation of myocardial tissue, which has a very low threshold for ablation. And because of its speed, with energy delivered in a few seconds, it can induce necrosis in targeted, specific tissues and leave unharmed collateral structures such as the esophagus, arteries, and nerves. Additional studies are certainly warranted, as Dr. Reddy and his team see significant promise in this ultra-rapid technique for ablation of atrial fibrillation.
The clinical trials were supported by the manufacturer of the ablation catheters, Iowa Approach Inc. Dr. Reddy has served as a consultant for Biosense Webster, Boston Scientific, and Iowa Approach, and he has stock options in Iowa Approach.
Clinical Data Management: AVIATOR 2 Study Finds Discrepancy Between Perceived and Empiric Risks After PCI
A study of patients with atrial fibrillation (AF) undergoing percutaneous coronary intervention (PCI) suggested that physician perspectives on ischemic and bleeding risk related to antithrombotic therapies (ATT) are poorly aligned with empiric risk estimates. This misalignment results in the possible overtreatment of low-risk patients with these therapies, and the undertreatment of high-risk patients.

AVIATOR 2 (Antithrombotic Strategy Variability In Atrial Fibrillation and Obstructive Coronary Disease Revascularized with PCI) also found that physicians’ priorities when prescribing ATT regimens were not well aligned with those of their patients. When asked, “Which two factors were most important in making your medication selection,” 93.8% of doctors cited safety, and 89.9% cited efficacy. When patients were asked what they were most concerned about when undergoing PCI, 63.4% cited heart attack, and 50.6% cited stroke. This indicates that the priority for the physician is the procedure itself, while their patients are more concerned with what happens afterward.
“I believe that a patient-centered approach to clinical research is of paramount importance,” says the AVIATOR 2 principal investigator, Roxana Mehran, MD, Professor of Medicine (Cardiology), and Population Health Science and Policy, Icahn School of Medicine at Mount Sinai. “We can develop many strong therapeutic regimens, but if we do not take our patients’ needs and opinions into account, we are not going to make much headway in terms of results.”
AVIATOR 2 enrolled 515 patients with nonvalvular AF undergoing PCI at 11 sites in the United States and Europe. ATT regimens were chosen at the discretion of the patients’ physicians, who recorded their perspectives on patient bleeding and ischemic risk using AVIATOR, a novel smartphone survey developed at the Icahn School of Medicine. Patients also used the app to provide feedback on their own health concerns. The resulting data were compared against precalculated risk scores, including CHA₂DS₂-VASc, which estimates the risk of stroke based on age, sex, and history of congestive heart failure, hypertension, stroke, vascular disease, and diabetes; and HAS-BLED, which estimates the risk of major bleeding based on factors including hypertension, renal disease, and prior major bleeding.
Presented in April 2018 at the Society for Cardiovascular Angiography and Interventions (SCAI) Scientific Sessions, the 30-day results of AVIATOR 2 found that compared with the empiric CHA₂DS₂-VASc score, the physician risk assessment was underestimated in 26.9% of patients and overestimated in 22.2% of patients. The results further revealed that physician risk assessment for stent thrombosis risk was overestimated in 71.5% of patients and underestimated in 4.6% of patients.
“This is the first time we have collected subjective physician risk assessments, and it was quite striking to see the significant discrepancy when compared with the empiric real-time risk assessments,” says Dr. Mehran, Director of Interventional Cardiovascular Research and Clinical Trials at the Zena and Michael A. Wiener Cardiovascular Institute at the Icahn School of Medicine. Dr. Mehran is planning a third AVIATOR study with a more robust smartphone app and a much larger patient population. “AVIATOR 2 is a relatively small study, but it speaks volumes to the potential of understanding physician and patient perspectives in comparison with empiric data in the effort to improve patient outcomes,” she says.
Clinical Data Management: Review Finds Two PCSK9 Inhibitors Safe and Effective for High-Risk Patients
Two human monoclonal antibodies that target the PCSK9 (proprotein convertase subtilisin/kexin type 9) enzyme significantly reduce low-density lipoprotein levels (LDLs) and the risk of atherosclerotic cardiovascular disease (ASCVD) events among high-risk patient groups, according to a state-of-the-art review conducted by researchers at Mount Sinai Heart and published in July 2018 in the Journal of the American College of Cardiology.

“We know from extensive genetic research conducted at Mount Sinai that individuals with low levels of PCSK9 have low concentrations of LDL particles in their blood and thus a reduced risk for an ASCVD,” says co-author Robert Rosenson, MD, Professor of Medicine (Cardiology) at the Icahn School of Medicine at Mount Sinai. “Clinical trials confirm that these PCSK9 inhibitors are highly effective, safe drugs for lowering LDL cholesterol and ASCVD events among patients with stable cardiovascular disease and acute coronary syndrome. The benefit of treatment is especially high in patients with multivessel coronary artery disease, peripheral arterial disease, and chronic kidney disease.”
The review analyzed the biology of PCSK9 and distinguished the human monoclonal antibody approach from antisense or small interfering ribonucleic acid (siRNA) oligonucleotides, which achieve a lower level of reduction in LDL levels. “Our goal in conducting this review was to provide a treatment algorithm for the use of PCSK9 in high-risk patients,” Dr. Rosenson says.
The review found that clinical trials of the PCSK9 inhibitors alirocumab and evolocumab resulted in a 55% to 72% reduction in LDL cholesterol (LDL-C) levels among patients with LDL cholesterol levels ≥70 mg/dL on maximally tolerated oral therapies such as statins and ezetimibe. In one clinical trial, FOURIER (Further Cardiovascular Outcomes Research With PCSK9 Inhibition in Subjects With Elevated Risk), patients who received evolocumab had a 9.8% composite endpoint of cardiovascular death, myocardial infarction (MI), stroke, hospitalization for angina, or revascularization versus 11.3% of the placebo group after an average two years of follow-up. In the ODYSSEY OUTCOMES trial (Evaluation of Cardiovascular Outcomes After an Acute Coronary Syndrome During Treatment With Alirocumab), 9.5% of patients receiving alirocumab experienced a primary endpoint (cardiovascular death, MI, ischemic stroke, or hospitalization for angina) versus 11.1% of the placebo group.
One high-risk subgroup that are candidates for PCSK9 inhibitor therapy are patients with statin-associated muscle symptoms. “Patients who had a prior myocardial infarction and either stop statin treatment or are placed on a lower dose to prevent muscle symptoms are at a very high risk for a recurring event,” Dr. Rosenson says. “Because these inhibitors do not interact with or enter any other tissue in the body, they have the ultimate safety profile, enabling us to achieve a significant reduction in patient LDL-C levels without any muscle-related adverse event.”
The review also challenged conventional wisdom on LDL cholesterol targets. Although an LDL-C target of < 70 mg/dL is widely accepted and reaffirmed by data, Dr. Rosenson says the consensus was that anything lower than 30 to 40 mg/dL might affect the ability of organs such as the brain, testes, or ovaries to synthesize cholesterol. “We found that there is no harm in reducing LDL cholesterol to levels < 25 mg/dL, because the lower the cholesterol rate, the lower the ASCVD event rate,” he says. “This therapy is so safe, so potent, and so effective that it supports the concept that there may be no lower limit for LDL-C in high-risk patients.”
Heart Failure: Significant Gains in a Next Generation of LVAD

Mount Sinai Heart is an early adopter of a next-generation left ventricular assist device (LVAD), HeartMate 3™, which was approved as a bridge to transplant in August 2017 and as destination therapy in October 2018 by the U.S. Food and Drug Administration.
“Patients with end-stage heart failure who receive LVAD support live longer and enjoy an improved quality of life, but devices have been fraught with complications,” says Noah Moss, MD, Assistant Professor of Medicine (Cardiology), Icahn School of Medicine at Mount Sinai, and Medical Director of Mechanical Circulatory Support, The Mount Sinai Hospital. “The HeartMate 3 has nearly eliminated one of the main complications of LVAD support, pump thrombosis, and has reduced the incidence of the worst complication, stroke.” Dr. Moss says Mount Sinai is “very comfortable” with the device, implanting it in 32 patients in 2018.
HeartMate 3, made by Abbott, is powered by a pump consisting of a single rotating disk that is fully magnetically levitated, which reduces trauma to the blood while providing appropriate flow. It is surgically attached to the left ventricle and ascending aorta, pumping blood from a failing heart that is unable to effectively eject it on its own. A driveline connects the pump to a controller outside the patient’s body, about the size of a large cellphone. Mount Sinai Heart participated in MOMENTUM 3, a multicenter, randomized trial of HeartMate 3. Early two-year outcomes found that for patients with advanced heart failure, the device was superior to a mechanical-bearing axial-flow pump. The study found that the rate of suspected pump thrombosis was 15.7% in the axial-pump group versus 1.1% in the HeartMate 3 group; and the stroke rate was 19.2% in the axial-pump group versus 10.1%, the lowest ever for a continuous-flow LVAD.
Sean P. Pinney, MD, Professor of Medicine (Cardiology), Icahn School of Medicine, and Director of Heart Failure and Transplantation, Mount Sinai Health System, says, “It is clear from this recent study and past ones that patients on LVAD support are not only alive but living a meaningful life, which makes the advent of LVADs so important as both a bridge to transplant and as destination therapy for patients with end-stage heart failure.”
Translational Research: Three-Dimensional Heart Organoids Signal a Step Forward in Drug Testing

Three-dimensional heart organoids, which function like a single ventricular chamber of a miniature human heart, have been developed by a leader in heart mechanics and tissue engineering, Kevin D. Costa, PhD, Associate Professor of Medicine (Cardiology), and Director of Cardiovascular Cell and Tissue Engineering at the Icahn School of Medicine at Mount Sinai. These beating “mini-hearts”—engineered from stem cells and suspended in a jar of fluid—could help change the future of drug discovery and development, Dr. Costa says.
“We are hoping that by having ready access to human mini-hearts for testing, if a drug is going to fail, we will find out early in the drug development process. And if a promising drug isn’t quite perfect, you could reformulate it or change the dosage, which you don’t typically get a chance to do once patients are involved,” says Dr. Costa, who is Chief Scientific Officer and scientific co-founder of Novoheart, a Vancouver-based, publicly traded company that produces the organoids.
Dr. Costa developed the first rat heart organoid chamber a decade ago while at Columbia University. He moved to Mount Sinai, he says, in part because of its excellent human stem cell program and the outstanding resources and opportunities for collaboration in the Cardiovascular Research Center (CVRC), led by Roger Hajjar, MD, the Arthur and Janet Ross Professor of Medicine (Cardiology). Novoheart’s CEO and co-founder, Ronald Li, MD, an expert in cardiac stem cells and electrophysiology, was also a CVRC faculty member at the time, leading to a “perfect” mix of their expertise to create a human mini-heart, Dr. Costa says.
The strategy is to combine bioengineering and stem cell technologies to grow miniature human hearts using cardiac cells derived from human pluripotent stem cells. After these pluripotent stem cells are differentiated into human heart muscle cells, they are combined with a collagen-based material in a liquid suspension, with fibroblast cells added for support. The mixture is poured into a mold that determines the size and shape of the organoid and then firmed in a specially designed bioreactor. The cells adhere to the collagen matrix and make tissue that mimics the heart’s structure, which can then be tested within the bioreactor to evaluate responses to drug treatments or other interventions, or to study various cardiac diseases.

The U.S. Food and Drug Administration is re-evaluating the way it screens new drugs for cardiovascular side effects and may incorporate organoids like Novoheart’s as part of their future regulatory process. Mice or other animals are normally used to test the safety of compounds before they are tried out in humans. But while mice are genetically close to humans, their hearts beat faster, have a different vascular anatomy, and are too small to sustain arrhythmias that a human heart could sustain. As a result, tests in mice are poorly predictive of the heart’s response to drugs in humans.
To put its cardiac assays to the test, Novoheart partnered with Pfizer in a blinded study of 17 drug compounds. Without the investigators’ knowing the identity of the drugs that were being tested, the bioengineered heart tissues showed promising results, predicting cardiac effects correctly in 15 of the samples, with one false positive and one false negative. Novoheart is also collaborating with Pfizer’s rare diseases research unit to develop an organoid model for Friedreich’s ataxia. Replicating the arrhythmias and other complications of the disease in the organoid could lead to novel therapies.
Why nudging the cells into a three-dimensional heart-shaped organoid leads them to function more like an actual heart is still unknown. “We are very much interested in teasing it out,” Dr. Costa says. Other challenges remain, including creating more sophisticated heart models and finding a way to use the mini-hearts to screen large numbers of compounds efficiently.
Kevin D. Costa, PhD, holds equity in Novoheart Holdings.
Translational Research: Aerosol-Delivered Gene Therapy Shows Promise in Treating Pulmonary Hypertension
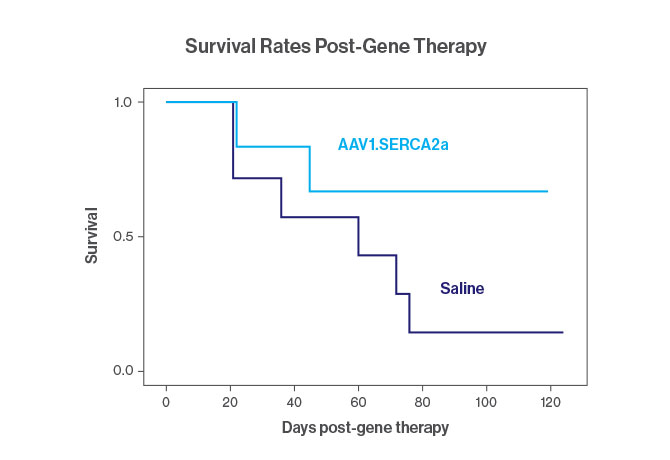
Researchers from Mount Sinai Heart have made advances in developing a novel aerosol-delivered gene therapy for pulmonary hypertension (PH) in a large animal model. The therapy was developed by the laboratory of Roger Hajjar, MD, the Arthur and Janet Ross Professor of Medicine (Cardiology), and Director of the Cardiovascular Research Center at the Icahn School of Medicine at Mount Sinai. It combines an aerosolized form of the SERCA2a (sarcoplasmic reticulum Ca2+-ATPase 2a) gene with a viral vector—an engineered adeno-associated virus (AAV)—to deliver the gene to the narrowed blood vessels in the lungs.

“We’ve proven in past studies that animals had lower pulmonary vascular resistance after treatment with SERCA2a gene therapy, and are now excited about the prospect of bringing this novel approach to humans in clinical trials to treat a very serious disease,” says Dr. Hajjar, an international leader in gene therapy. Past research relied on a sprayer device inserted directly into the trachea via the endotracheal tube to deliver the therapeutic gene. However, as Dr. Hajjar points out, this methodology is not well suited for use in severely ill patients with PH, who would benefit from a minimally invasive delivery system. To that end, he developed a nebulization delivery system for his latest study in animal models. In the study, which was published in August 2018 in the journal Pulmonary Circulation, nebulization delivery of the SERCA2a gene therapy was examined in Yucatan miniature swine. The animals underwent pulmonary vein banding to establish PH, and two months after surgery, 13 animals were randomized to receive either gene therapy or saline, using nebulization delivery. To demonstrate that nebulization was at least comparable to the intratracheal aerosol method, researchers also delivered blue dye via nebulization or the intratracheal sprayer. Nebulization delivery resulted in a more homogeneous distribution of the dye compared to the intratracheal sprayer delivery method, which concentrated the dye in lung central segments.
“We found that SERCA2a delivery through nebulization is safe and results in an even distribution of vectors in both the left and right lung lobes,” says Dr. Hajjar, senior author of the study. “Just as important, we learned there was no loss of efficacy in treatment by using a less invasive means of vector delivery.” The study supported previous findings that airway delivery prevented PH progression in rodents and swine models by increasing the expression of the SERCA2a protein. It improved the animals’ heart and lung function, including pulmonary pressure and vascular resistance, while limiting pulmonary blood vessel remodeling. The latter is a pernicious hallmark of PH, characterized by a proliferation of smooth muscle cells and a thickening and narrowing of lung blood vessels, which can lead to right ventricle failure and premature death.
According to Dr. Hajjar, clinical trials using AAV vector delivery of SERCA2a to treat pulmonary hypertension could begin at Mount Sinai over the next 12 months. This could pave the way for a commercially available product within several years, filling a void for patients with this rare but rapidly progressing disease.
Message from the Director: Valentin Fuster, MD, PhD
A landmark trial confirming the standard of care for patients with diabetes and multivessel coronary artery disease; a randomized study that is instilling heart-healthy behaviors among underserved Harlem preschoolers and their families; and a new certification from The Joint Commission as a Comprehensive Cardiac Center—these are some of the latest achievements of Mount Sinai Heart. This 2019 issue of the Heart Specialty Report also features an aerosol-delivered gene therapy for pulmonary hypertension; an innovative method for ablation of atrial fibrillation; and novel “mini-heart” organoids that show promise in drug discovery and development.

Through research, education, and clinical care, Mount Sinai Heart is improving people’s lives in New York and around the world. Mount Sinai is leading a global clinical trial evaluating the safety of a transcatheter mitral valve replacement system for patients with severe mitral regurgitation. We also are leading an international trial that is comparing two minimally invasive treatments for patients with multivessel coronary artery disease. Interventionalists at The Mount Sinai Hospital’s Cardiac Catheterization Laboratory are sharing their expertise by developing a machine learning model to predict response to high-dose statins and providing a live webcast that demonstrates therapeutic options for structural heart disease—a series that has been viewed more than 650,000 times in 138 nations. Physician-scientists provided evidence-based guidance in patient care by reviewing the use of PCSK9 inhibitors, participating in the pivotal trial of a new LVAD device, and leading a large study of patients with atrial fibrillation undergoing PCI.
The Mount Sinai Hospital was again among the top 10 in the nation for cardiology and heart surgery, according to U.S. News & World Report, in further recognition of the sustained accomplishment and compassionate patient care at Mount Sinai Heart.