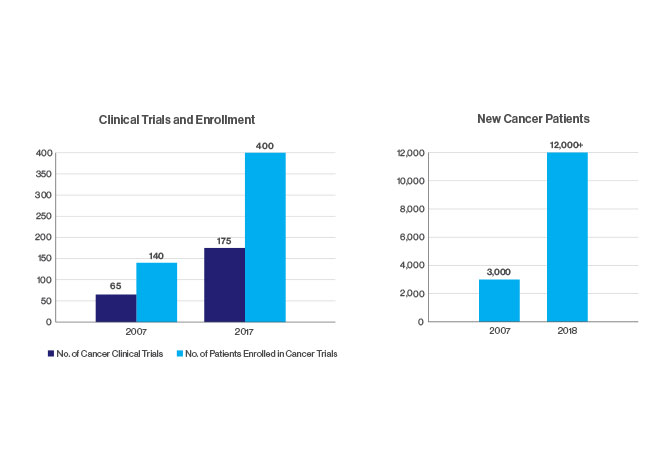
The Tisch Cancer Institute
At The Tisch Cancer Institute, a National Cancer Institute-designated center, our multidisciplinary approach to cancer treatments and clinical breakthroughs may one day put an end to cancer.
The Tisch Cancer Institute
Innovation and transformation energize our programs and reflect The Tisch Cancer Institute’s world-class research. As a National Cancer Institute-designated Cancer Center, we belong to an elite group of U.S. institutions committed to advancing the research and treatment of cancer.
This specialty report highlights:
- Extending lives by putting cancer cells to “sleep”
- Cancer dormancy research that is gaining traction
- Advances in multiple myeloma: Clinical trials explore the potential of CAR T-cell therapy
- Advances in multiple myeloma: Novel sequencing platform that may help determine effective drugs
- Mount Sinai investigates a novel class of drugs for patients with myeloproliferative disease
- Setting new standards in treating neuroendocrine tumors
- Surprising cancer results found among first responders
- Improving the prognosis for melanoma patients
- New studies that provide insight into p53-like bladder cancer
- Consortium begins to investigate the rising incidence of colon cancer among people under age 50
- Message from the Director: Ramon Parsons, MD, PhD
Extending Lives By Putting Cancer Cells To “Sleep”
For more than 20 years, Julio Aguirre-Ghiso, PhD, Professor of Oncological Sciences, Otolaryngology, and Medicine (Hematology and Medical Oncology), has studied the source of metastatic cancer and focused on therapies that target dormant cancer cells before they start growing. His major studies have disrupted the prevailing scientific dogma by revealing that early evolved cancer cells can spread before primary tumors develop and remain dormant for months or even years before spawning aggressive and deadly metastases.
The research has “led us to think that therapies that target the root of metastasis would be really transformative because we would be eradicating the residual disease,” says Dr. Aguirre-Ghiso.
Currently, Dr. Aguirre-Ghiso and William Oh, MD, Deputy Director of The Tisch Cancer Institute, are studying prostate cancer cells that have departed the primary tumor site and sit quietly secluded elsewhere in the body. The cells are not actively dividing but are poised to awaken at some future time, often in bones, a common site for prostate cancer recurrence.
In a clinical trial involving 20 patients, Drs. Aguirre-Ghiso, Oh, and Vaibhav Patel, a clinical fellow in Hematology/Medical Oncology, are testing two commercially available products—5-azacytidine and all-trans retinoic acid (a type of vitamin A)—to determine whether the drug combination can induce tumor dormancy in prostate cancer. In preclinical studies, the products showed an ability to put cancer cells to sleep by regulating their epigenetic signaling, thus making them unable to initiate a relapse or further metastasis.

Another goal of the clinical trial is developing a blood test and biomarkers in order to identify dormant cancer cells. In a recent study published in Breast Cancer Research, Dr. Aguirre-Ghiso and his team revealed that NR2F1, a protein involved in dormancy onset, can pinpoint dormant disseminated cancer cells in breast cancer patients and inform on their prognosis. NR2F1 will be tested as a dormancy biomarker in the prostate cancer clinical trial.
“Prostate cancer is a very heterogeneous disease, so knowing at the outset which patients are more likely to respond to the combination of 5-azacytidine and all-trans retinoic acid would be extremely beneficial,” says Dr. Oh. The targeting of cancer dormancy has prompted the Mount Sinai Health System to form an alliance with a new biotechnology company, a spinoff from the Aguirre-Ghiso lab that could eventually bring to market drugs that directly target dormant cancer cells.
Cancer Dormancy Research Gains Traction
Due to the groundbreaking work of Julio Aguirre-Ghiso, PhD, the field of cancer dormancy research is growing. In June, the American Association for Cancer Research held the first Cancer Dormancy and Residual Disease Conference, which was co-chaired by Dr. Aguirre-Ghiso.
The conference featured more than 300 attendees and 100 research abstracts on dormant disease across multiple cancers.
William Oh, MD, presented the design of his and Dr. Aguirre-Ghiso’s clinical trial, which is one of the first to test this hypothesis directly in patients.
“The fact that we were able to pull together a large and extremely successful conference around this subject shows that we now have a whole new generation of young scientists working and thinking hard about this problem, which may be the most significant output of our work,” says Dr. Aguirre-Ghiso.
Advances in Multiple Myeloma: Clinical Trials Explore the Potential of CAR T-Cell Therapy
Four clinical trials at The Tisch Cancer Institute explore the potential of chimeric antigen receptor CAR T-cell therapy to treat relapsed or refractory multiple myeloma patients.
This therapeutic approach has proven effective in targeting CD19—a biomarker for normal and neoplastic B cells—and has resulted in the U.S. Food and Drug Administration’s (FDA) approval of two CAR T-cell therapies for lymphoma.

Sundar Jagannath, MD, Director of the Multiple Myeloma Program at The Tisch Cancer Institute and a Principal Investigator on some of the clinical trials, says the therapy can be equally effective in attacking B-cell maturation antigens (BCMAs), which have been identified as a target for therapeutic intervention in multiple myeloma. The clinical trials are for patients who have relapsed or refractory disease after exposure to a proteasome inhibitor, an immunomodulator, and D38 antibody therapy. Patients undergo apheresis, their T-cells are sent to a pharmaceutical company for engineering to express the CARs, and they receive their infusion following lymphodepleting chemotherapy.
Two of the clinical trials are currently recruiting patients: a phase I trial that uses two heavy chain-binding domains to enhance binding efficacy; and a phase II trial for CAR T-cell therapy bb2121—the most mature CAR T approach in development for multiple myeloma, which has received an FDA Breakthrough Therapy Designation.
“We know CAR T therapy is life-changing for patients who have received it,” says Deepu Madduri, MD, Assistant Professor of Medicine (Hematology and Medical Oncology) at the Icahn School of Medicine, who also serves as a principal investigator on some of the clinical trials. “The challenge is that the CAR T-cells disappear over time, resulting in a median progression-free survival rate of approximately one year. Thus, the goal now is to develop memory T-cells that deliver long-term positive outcomes to multiple myeloma patients.”
Mount Sinai researchers are also investigating another class of drugs for treating multiple myeloma that are called bispecific antibodies. These “off the shelf” products represent a more cost-effective approach for multiple myeloma treatment because they eliminate the need to collect and engineer the patient’s own T-cells.
“These antibodies attach to both the patient’s T-cells and cancer cells, causing them to come together and enabling the T-cells to kill the cancer cells,” Dr. Madduri says. “Our vast arsenal of therapies are designed to help patients achieve sustained remissions with high quality of life.”
Advances in Multiple Myeloma: Novel Sequencing Platform May Help Determine Effective Drugs
As multiple myeloma progresses and patients exhaust their lines of therapy, there are fewer options for treatment. Relapse is nearly inevitable and median survival stands at around six years. Researchers at The Tisch Cancer Institute have taken steps to improve those odds by developing a novel RNA and DNA sequencing platform that can help determine which drugs work best for late-stage and drug-resistant multiple myeloma patients.

In a clinical trial reported in JCO Precision Oncology in August 2018, the researchers expanded the traditional DNA-based approach by adding RNA sequencing to uncover targets for a broad swath of drugs approved by the U.S. Food and Drug Administration (FDA) that go well beyond those typically used to treat multiple myeloma. The work of these researchers represents the first time genome-wide RNA profiles have been clinically assessed for hematological malignancies.
“Our findings really expand the scope of what patients with advanced multiple myeloma can receive,” says Samir Parekh, MD, Director of Translational Research in Myeloma at The Tisch Cancer Institute of the Icahn School of Medicine at Mount Sinai. “We identified treatment options for each patient by checking different libraries with hundreds of compounds each until we found the best therapeutic drugs.”
Of the evaluable trial participants who received an FDA-approved drug based on a comprehensive profile generated from DNA sequencing and RNA data, 76 percent showed a clinical benefit—a response rate Dr. Parekh calls “a real eye-opener, especially since many of these patients had end-stage myeloma.”
RNA sequencing is viewed by developers as a perfect complement to the move to precision medicine in cancer therapy, where treatment is based on specific genomic alterations observed in a patient’s tumor rather than on the tumor histology or tissue type.
“It’s able to capture more dynamic aspects of unique tumor biology and provide information beyond what’s available with DNA alone,” says Joel Dudley, PhD, Associate Professor of Genetics and Genomic Sciences at the Icahn School of Medicine, who collaborated with Dr. Parekh on the RNA/DNA profiling study.
“Because of our patient volume, we’ve developed a unique understanding of myeloma biology and how to look outside the myeloma toolbox to match DNA mutations and RNA profiles to specific drugs,” says Dr. Parekh.
Mount Sinai Investigates a Novel Class of Drugs For Patients With Myeloproliferative Disease

Researchers in The Tisch Cancer Institute’s Myeloproliferative Neoplasms (MPN) Program have long been intrigued by the anticancer effects of a novel class of drugs, called nutlins, discovered by Roche. These small molecule antagonists work by restoring the tumor suppression activity of the p53 gene—nicknamed the “guardian of the genome”—which, in many cancers, is inhibited by its interaction with MDM2, a phosphoprotein that is often overexpressed in cancer cells.
“When we realized there were agents that could bind to and antagonize the MDM2-p53 interaction, we were anxious to exploit that finding for therapeutic purposes,” says Ronald Hoffman, MD, Professor of Medicine (Hematology and Medical Oncology) at the Icahn School of Medicine at Mount Sinai and a renowned expert in myeloproliferative disorders.
Dr. Hoffman led the pivotal laboratory investigation of MDM2 antagonists either alone or in combination with interferon-alfa, revealing the ability of this class of agent to selectively deplete malignant hematopoietic stem cells in murine models of MPN.
This research paved the way for the significant role The Tisch Cancer Institute currently plays in a series of clinical trials that evaluate the effects of nutlin molecules in patients with MPNs, including polycythemia vera (PV), myelofibrosis (MF), and acute myeloid leukemia (AML).
Since MDM2 is up regulated in each of these blood disorders, “it is a very attractive therapeutic target for us,” says John Mascarenhas, MD, Associate Professor of Medicine (Hematology and Medical Oncology), and Director of the Adult Leukemia Program at the Icahn School of Medicine. As selective small molecule inhibitors, nutlins occupy the p53 binding pocket of MDM2, serving to stabilize p53 and reactivate the normal process of cell apoptosis.
At the annual American Society of Hematology conference in 2017, the Mount Sinai team presented the favorable results of a 12-patient phase I clinical trial of idasanutlin. The drug is a second-generation orally available member of the nutlin family that was tested on patients with refractory PV. Recently, Roche followed up by launching a multi-institution, international phase II study of idasanutlin, in which Mount Sinai serves as a primary investigative site. The multiple nutlin studies have been funded by translational research grants from the Leukemia and Lymphoma Society and the National Cancer Institute (NCI).
“Our hope is that idasanutlin will improve long-term survival in patients with chronic blood cancers,” says Dr. Hoffman, who has also served as President of the American Society of Hematology. He says the mission is urgent because “there are no other agents currently available to improve the longevity of these individuals.”
As part of its extensive research into MPN, Mount Sinai is working with a number of pharmaceutical companies on a molecule that could inhibit the activity of both MDM2 and MDM4, a protein with a structure similar to MDM2. Both have been implicated in cancer cell proliferation.
Still another research initiative involves development of a combination nutlin therapy. “We’re greatly encouraged by what we’ve seen so far of these small molecule inhibitors, and believe they have a lot of potential in the MPN space, though much more research is required,” says Dr. Mascarenhas. The Mount Sinai team, through its longstanding leadership in the Myeloproliferative Neoplasms Research Consortium—an NCI-sponsored program of 11 institutions in the United States and Canada—is helping to advance that work.
Setting New Standards in Treating Neuroendocrine Tumors

Since taking the helm of the Mount Sinai Health System’s Center for Carcinoid and Neuroendocrine Tumors in early 2018, Edward M. Wolin, MD, has been setting new standards in the diagnosis and treatment of the disease. Among his first changes was moving the Center from the Division of Gastroenterology to The Tisch Cancer Institute to encourage multidisciplinary care. He also launched a weekly Neuroendocrine Tumor (NET) Board to develop more personalized treatment plans and initiated a NET Support Group for patients.
Earlier diagnosis and intervention, he says, are the key to achieving positive results. “If you have a patient who presents with a small bowel obstruction, look for the possibility of cancer as opposed to treating the symptoms only. Imaging studies, small bowel endoscopy, and lab studies go a long way toward determining or ruling out a NET and make a huge difference in the lives of patients.”
Dr. Wolin is increasing the Center’s participation in clinical trials. He is the principal investigator of several National Institutes of Health–funded studies, including a randomized phase III trial to determine the efficacy and safety of MLN0128, a dual kinase mTOR inhibitor. MLN0128 has demonstrated promise among patients who have not responded to everolimus and who have tumors that have spread throughout their bodies or that cannot be resected. He is also leading a trial of a liver metastasis therapy administered through the hepatic artery.
A pioneer in NET therapies, such as novel somatostatin analogs, mTOR inhibitors, antiangiogenic drugs, and peptide receptor radiotherapy (PRRT), Dr. Wolin has led the adoption of advanced diagnostic and treatment technologies. PRRT, for example, uses an intravenous injection of Lu 177-DOTATATE, a radiolabeled somatostatin analog, to deliver molecularly targeted radiation to NETs. “Four outpatient infusions of the Lu 177-DOTATATE treatment can control NETs for many years,” says Dr. Wolin, who helped develop the therapy with Lale Kostakoglu, MD, Chief of Nuclear Medicine and Molecular Imaging at Mount Sinai.
The Center was among the first to use a novel PET scan with an imaging isotope, Ga-68-DOTATATE. “There are other promising targeted agents that we are studying, such as cabozantinib. My goal is to develop and test therapies at the Center so that we can offer our patients more options for treatment,” he says.
Surprising Cancer Results Found Among First Responders
More than 17 years after the September 11 attacks, Mount Sinai Health System researchers are seeing higher rates of prostate and thyroid cancer among first responders when compared to other populations. These findings have been borne out in two studies.

Most recently, researchers led by Emanuela Taioli, MD, PhD, Director of the Institute for Translational Epidemiology and Associate Director of The Tisch Cancer Institute, detailed their findings on prostate cancer in a 2018 study in the European Journal of Cancer Prevention. The investigators found that responders who spent the most time working at Ground Zero and had a higher exposure to the toxic dust cloud that formed after the World Trade Center buildings collapsed had the most advanced stages of prostate cancer—stages III and IV, which represent tumor invasion.
In a 2016 study in the American Journal of Industrial Medicine, which was led by Paolo Boffetta, MD, Professor of Medicine (Hematology and Medical Oncology), three cohorts of 9/11 responders—firefighters, sanitation and construction workers, and protective services employees—were found to have modest elevations in thyroid and prostate cancer. Mount Sinai is home to the largest World Trade Center Health Program Clinical Center of Excellence in the New York metropolitan region, and 25,000 patients have consented to participate in research. A biobank of more than 600 cancer samples from first responders has helped lay the groundwork for Mount Sinai’s World Trade Center research findings.
“The results of these studies were not something we anticipated,” says Dr. Taioli. “We expected to see increases in lung cancer given that responders were inhaling considerable amounts of toxic dust, metals, and asbestos. Our hypothesis is that the dust is creating local inflammation in the lungs, which triggers a general inflammatory response that facilitates cancer progression. But we do not know why we are seeing it in these two specific organs and not others.”
Dr. Taioli says continued research and monitoring will eventually confirm whether the responders are at a higher risk for prostate and thyroid cancer. “These cancers are largely preventable, so if we can determine with certainty that exposure is the cause, we can make clinical care recommendations that lead to early intervention and prevention,” she adds.
Improving the Prognosis for Melanoma Patients
A novel epigenetic mechanism that causes resistance to the standard treatment given to melanoma patients with mutations in the BRAF gene has been discovered by Emily Bernstein, PhD, and her team of researchers at the Icahn School of Medicine at Mount Sinai. BRAF mutations are found in roughly half of all melanomas. The researchers also found a biomarker—a protein called IGFBP2—that is associated with poor prognoses in melanoma patients.
Both findings appear in the August 24, 2018, issue of Nature Communications, and point to a potential, multipronged approach to preventing drug resistance or reversing it once it has occurred by inhibiting the effects of BRAF mutations and IGFBP2-driven biological pathways.
“The incidence of cutaneous malignant melanoma is rising and its therapeutic management remains challenging,” says Dr. Bernstein, Associate Professor of Oncological Sciences, and Dermatology. “In recent years, there has been extensive therapeutic development to inhibit key biological targets. Although a large proportion of patients with advanced metastatic melanoma who harbor BRAF mutations respond to the standard therapy, known collectively as MAPK inhibitors, subsequent resistance remains a major clinical challenge.”
Dr. Bernstein is conducting further studies on the epigenetic mechanisms altered in cancer, including the modeling of macroH2A knockout mice models for melanoma and breast cancer.
“The H2A family is the largest family of histone variants and it has been largely understudied in cancer,” Dr. Bernstein says. “We know histone variants can promote tumorigenic programs, so we want to target these proteins and prevent them from doing that. But we cannot target the histone on its own because there is no aspect of its structure where we could apply a small molecule that would block its role.”
Instead, Dr. Bernstein is looking at potentially targeting the “histone chaperone–histone variant” interaction that introduces these proteins into the genome. “We are setting up assays and working together with the Drug Discovery Institute at the Icahn School of Medicine to identify small molecules that could block this interaction. We are still in the very early stages, but if this research is successful, it opens the door to collaborating with medicinal chemists on an enhanced molecule that can be tested in animals.” She says the more that researchers delve into the epigenome, the more targets they will discover. “There are many startups and large pharmaceutical companies already moving into this line of drug development, and some treatments are already undergoing clinical trials. It is clear that alteration of the epigenome is a major driver in multiple cancers, and the more we explore that, the better we will understand how it works.”
New Studies Provide Insight Into p53-like Bladder Cancer
Two microRNA biomarkers that can help stratify which patients with aggressive p53-like bladder cancer will have a better or worse prognosis have been uncovered by researchers at the Icahn School of Medicine at Mount Sinai. Until now, the optimal approach to treating p53-like tumors—which are generally resistant to chemotherapy—has been poorly defined and urgently needed.

“Working with our colleagues in Genetics and Genomic Sciences, we applied a computer model that was able to make microRNA much more understandable in terms of its impact on tumor behavior,” says Matthew Galsky, MD, Professor of Medicine (Hematology and Medical Oncology) at The Tisch Cancer Institute at the Icahn School of Medicine. That research—published in the July 2018 issue of Oncogene—identified two specific microRNAs as potential prognostic markers of the p53-like subset that might also have the ability to yield “critical information for personalizing treatment options,” he adds.
The study’s computer model, known as ActMIR, had previously been developed at Mount Sinai and successfully validated microRNA activity in multiple subtypes of breast cancer. Researchers now applied it to bladder cancer genomic data in the National Cancer Institute’s Cancer Genomic Atlas to identify the activities of the two biomarkers of p53-like bladder cancers, miR-106b-5p and miR-532-3p.
“Our computational methods not only provided us with deeper insights into the cellular mechanisms of this elusive type of bladder cancer, but also revealed the potential of microRNAs as therapeutic targets for treating the disease,” says the study’s senior author, Jun Zhu, PhD, Professor of Genetics and Genomic Sciences at Mount Sinai, and Head of Data Sciences at Sema4, a Mount Sinai venture.
Additional discoveries about the sensitivity and resistance of bladder cancer to immunotherapy were published by the research team in the August 2018 issue of Nature Communications.
The team’s goal is to bring their findings to the clinic and establish early-stage clinical trials that will improve patient care. The Tisch Cancer Institute established the Novel Therapeutics Unit almost two years ago in order to accomplish this. To date, more than 15 clinical trials are underway, some involving new drugs and others involving novel combinations of existing drugs. “Our approach is to capitalize on the most promising science originating within Mount Sinai and outside from our industry partners,” says Dr. Galsky.
Consortium Begins to Investigate the Rising Incidence of Colon Cancer Among People Under Age 50

The Mount Sinai Health System in July created a new consortium to determine why an increasing number of U.S. adults under the age of 50 are being diagnosed with colorectal cancer at a time when the incidence is dropping among people over the age of 50.
Called the ColoRectal Cancer in Adults at Young Onset (CRAYON) initiative, the group will include experts from the New York metropolitan area’s leading academic medical centers—Columbia University Medical Center, NYU Langone Medical Center, Weill Cornell Medicine, Montefiore Medical Center, Memorial Sloan Kettering Cancer Center, and Northwell Health.
“It used to be that approximately 7 percent of colorectal cancer would affect people younger than age 50,” says Steven H. Itzkowitz, MD, Professor of Medicine (Gastroenterology) and Oncological Sciences at the Icahn School of Medicine at Mount Sinai. “Now it’s approximately 10 percent. The rate of this rise is worrisome, as is the fact that there is a predominance of rectal cancers among this demographic, which can invade adjacent organs and require different treatment, at least initially. No one knows why these trends are happening, which is why I decided to create this consortium.”
While the drop in colorectal cancer among people over age 50 has been attributed to the detection and early removal of precancerous polyps due to increased screening, the reasons for the rise among younger people are uncertain. The numbers “may reflect an increased sedentary lifestyle and a higher prevalence of obesity and/or unfavorable dietary patterns in children and young adults,” according to the American Cancer Society.
Although each participating academic medical center has a legacy of innovation in cancer research that would support individual studies, Lina H. Jandorf, Research Professor of Population Health Science and Policy, and Oncological Sciences at the Icahn School of Medicine, says the CRAYON consortium would create the critical mass necessary for a more in-depth approach to exploring rising incidence rates.
“This is such a difficult question to tackle that no one has really been able to take it on,” Ms. Jandorf says. “By pooling our considerable resources and knowledge, we can do this thoughtfully and precisely but also with the necessary critical mass of patients that could potentially enable us to determine which populations are at greater risk for colorectal cancer.”
Dr. Itzkowitz says CRAYON members have met several times to set goals, develop research projects, and explore funding opportunities. The plan is to launch a prospective case-control study to identify risk factors that increase the likelihood that a patient will develop colorectal cancer.
“We want to see if there are differences among people who present with an early-onset colon or colorectal cancer based on factors such as age or gender,” he says. “We are currently formulating controls for such a study and identifying experts we can bring on board to help us determine the risk factors we want to look at and the biospecimens we want to gather. It is very early stages, but we are putting the fundamentals in place to make this happen.”
Dr. Itzkowitz says the work could take years, “but if we are successful, we will identify high-risk populations who will benefit from earlier interventions and screenings.”
Message from the Director: Ramon Parsons, MD, PhD
We begin 2019 by highlighting the new goals in precision medicine that we have set for ourselves and reflecting on our successes over the past 10 years since The Tisch Cancer Institute was established at The Mount Sinai Hospital, a U.S. News & World Report Honor Roll hospital.

Today, we are a National Cancer Institute (NCI)–designated cancer center and a principal member of the NCI’s Cancer Moonshot initiative.
We have created early cancer detection and prevention programs in lung, breast, gastrointestinal, liver, and prostate cancer, and established Mount Sinai as a national center of excellence in immunology and drug discovery. More than 200 of our faculty are engaged in cancer research. The NCI has commended us on the strength of our community-based programs and our commitment to population health.
Patient care is at the center of our mission. Mount Sinai’s footprint as a health system has increased exponentially to the point where The Tisch Cancer Institute now has more than 300,000 annual patient visits. The sheer size and scope of our cancer care have enabled us to generate large and diverse datasets and biospecimens. This, in combination with our capabilities in data gathering and analysis, tumor profiling, and clinical translation, is helping us fulfill our goal of discovering and delivering breakthrough cancer treatments.
With this in mind, The Tisch Cancer Institute held an inaugural Cancer Precision Medicine Retreat at the New York Academy of Medicine in the spring of 2018. We invited our Mount Sinai colleagues from the Icahn Institute for Genomics and Multiscale Biology and the Department of Genetics and Genomic Sciences to join us for discussions on the best ways to develop new initiatives and form collaborations by leveraging our size and strength. By drawing upon Mount Sinai’s agile, entrepreneurial culture, we expect to be able to advance cancer therapeutics and deliver the best outcomes to our patients at a quicker pace than ever before.
The articles included in this annual publication summarize our latest discoveries and developments. We expect there will be plenty more to follow throughout 2019.