Hepatitis C Drugs Combined with Remdesivir Show Strong Effectiveness Against COVID-19


A combination of remdesivir, a drug currently approved in the United States for treating COVID-19 patients, and repurposed drugs for hepatitis C virus (HCV) was 10 times more effective at inhibiting SARS-CoV-2, the virus that causes COVID-19.
The combination therapy points a way toward a treatment for unvaccinated people who become infected, as well as for vaccinated people whose immunity has waned, for example due to the emergence of virus variants that escape this immune protection.
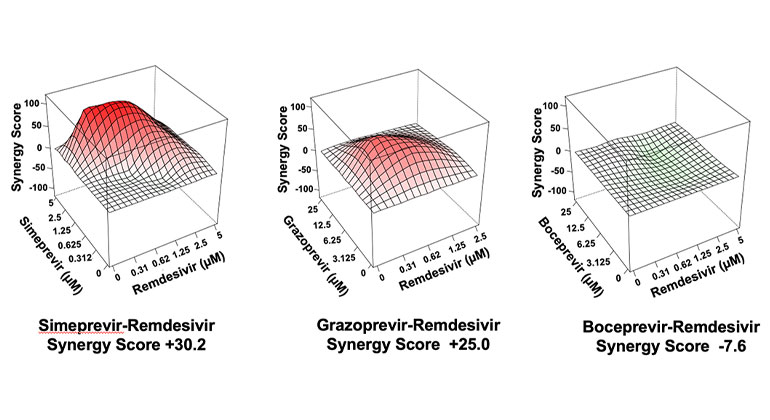
Four HCV drugs—simeprevir, vaniprevir, paritaprevir, and grazoprevir—in combination with remdesivir boosted the efficacy of remdesivir by as much as 10-fold, the researchers reported today in Cell Reports. The research team included scientists from Icahn School of Medicine at Mount Sinai, the University of Texas at Austin, and Rensselaer Polytechnic Institute (RPI).
Remdesivir targets a range of viruses and was originally developed over a decade ago to treat hepatitis C and a cold-like virus called respiratory syncytial virus (RSV). During the Ebola outbreak, Remdesivir was tested in clinical trials and found to be safe and effective for patients. Early in the pandemic, it was seen as a good therapy for COVID-19 but did not live up to its early promise in studies.
The research team performed protein binding and viral replication studies on SARS-CoV-2, the virus that causes COVID-19, using remdesivir and 10 hepatitis C drugs, some of which are already approved by the Food and Drug Administration.
RPI team had previously identified “striking similarity” between protease structures, or enzymes that are essential for coronaviral replication, in SARS-CoV-2 and HCV. The similarity raised the possibility that existing drugs which bind to and block the hepatitis C protease would have the same effect against SARS-CoV-2.
Using a supercomputer to model how drugs bind to viral proteins, the RPI researchers predicted that the 10 HCV drugs could bind snugly to the SARS-CoV-2 Main protease, named Mpro. In addition, they showed that seven of these drugs actually inhibited the SARS-CoV-2 protease. The research team at Icahn Mount Sinai then tested whether these seven drugs would inhibit SARS-CoV-2 virus replication in monkey and human cells grown in culture. In subsequent experiments the researchers were surprised to find that the four HCV drugs inhibited a different SARS-CoV-2 protease, known as PLpro. This observation proved to be very important. When each of the seven HCV drugs were tested in combination with remdesivir, only the four drugs that unexpectedly targeted PLpro boosted the efficacy of remdesivir, by as much as 10-fold.
Adolfo Garcia-Sastre, PhD, one of the authors on the paper, Irene and Dr. Arthur M. Fishberg Professor of Medicine. and Director of the Global Health and Emerging Pathogens Institute at Icahn Mount Sinai, said, “Combined use of remdesivir with PLpro inhibitors for the treatment of COVID-19 could be a game changer for patients with COVID-19 who are not vaccinated. It could also reduce the possibility of selecting SARS-CoV-2-resistant viruses.”
“The identification of PLpro as an antiviral target that has a synergistic effect in combination with remdesivir is a very important finding. We hope this work will encourage the development of specific SARS-CoV-2 PLpro inhibitors for inclusion in combination therapies to produce a highly effective antiviral cocktail that may potentially prevent the rise of resistance mutations,” said Kris White, PhD, Assistant Professor of Microbiology at Icahn Mount Sinai.
“Because these HCV drugs are already approved for use and their potential side effects are known, such a combination therapy could be tested in humans more quickly than for a new drug,” said Robert M. Krug, PhD, Professor Emeritus of Molecular Biosciences at The University of Texas at Austin and co-corresponding author of the paper.
One big drawback of remdesivir is that it must be administered intravenously, limiting its use to patients already admitted to the hospital. “Our goal is to develop a combination of oral drugs that can be administered to outpatients before they are sick enough to require hospitalization,” said Krug. “The HCV drugs that enhance remdesivir’s antiviral activity are oral drugs. It is important to identify oral drugs that inhibit the SARS-CoV-2 polymerase in order to develop an effective outpatient treatment.”
“Nearly 3 million people have died worldwide from COVID-19. There are situations where the vaccine isn’t the best option and it would be helpful to have orally available antivirals,” said Gaetano Montelione, PhD, a member of the Rensselaer Center for Biotechnology and Interdisciplinary Studies (CBIS). “Here we see a promising synergy that, if confirmed through additional research and clinical trials, could provide a new antiviral to combat COVID-19.”
The other participants in this research are Catherine Royer, Theresa A. Ramelot, Thomas B. Acton, and Balasubramanian Harish at RPI; and Romel Rosales, Elena Moreno, Thomas Kehrer, and Lisa Miorin at Icahn Mount Sinai.
This research was supported by the National Institutes of Health, the RPI Center for Computational Innovations, the National Institute of Allergy and Infectious Diseases, the Defense Advanced Research Projects Agency, the Department of Defense, the JPB Foundation and the Open Philanthropy Project.
About the Mount Sinai Health System
Mount Sinai Health System is one of the largest academic medical systems in the New York metro area, with more than 47,000 employees working across seven hospitals, more than 400 outpatient practices, more than 600 research and clinical labs, a school of nursing, and leading schools of medicine and graduate education. Mount Sinai advances health for all people, everywhere, by taking on the most complex health care challenges of our time—discovering and applying new scientific learning and knowledge; developing safer, more effective treatments; educating the next generation of medical leaders and innovators; and supporting local communities by delivering high-quality care to all who need it.
Through the integration of its hospitals, labs, and schools, Mount Sinai offers comprehensive health care from conception through geriatrics, leveraging innovative approaches such as artificial intelligence and informatics while keeping patients’ medical and emotional needs at the center of all treatment. The Health System includes more than 6,400 primary and specialty care physicians and 10 free-standing joint-venture centers throughout the five boroughs of New York City, Westchester, Long Island, and Florida. Hospitals within the System are consistently ranked by Newsweek’s® “The World’s Best Smart Hospitals,” “Best in State Hospitals,” “World’s Best Hospitals,” and “Best Specialty Hospitals” and by U.S. News & World Report's® “Best Hospitals” and “Best Children’s Hospitals.” The Mount Sinai Hospital is on the U.S. News & World Report® “Best Hospitals” Honor Roll for 2025-2026.
For more information, visit https://www.mountsinai.org or find Mount Sinai on Facebook, Instagram, LinkedIn, X, and YouTube.
Scientists Uncover How COVID-19 Variants Outsmart the Immune System
Nov 21, 2025 View All Press Releases