Departments of Neurology and Neurosurgery
We are dedicated to rapid advances in technology, pioneering research, and multidisciplinary collaboration to set a new standard for team care to dramatically improve the quality of life for patients.
Departments of Neurology and Neurosurgery
The Mount Sinai Health System’s departments of Neurology and Neurosurgery are excited about bringing new advances in neuroscience into clinical practice.
This specialty report highlights:
- A study that shows how artificial intelligence helped detect acute neurological cases earlier
- Advancing the use of novel technology in the OR
- Using vivid displays of vascular and cranial nerve involvement with pathology to engage patients
- Study findings that provide insight into how viruses may play a role in Alzheimer’s disease
- Prominent neuroimaging researcher Trey Hedden, PhD, joins Mount Sinai
- How a long-ago lab discovery by neurologist leads to treatment that received approval in 2018
- Mount Sinai’s pioneering work in Neuro-Otology
Study Shows How Artificial Intelligence Helped Detect Acute Neurological Cases Earlier
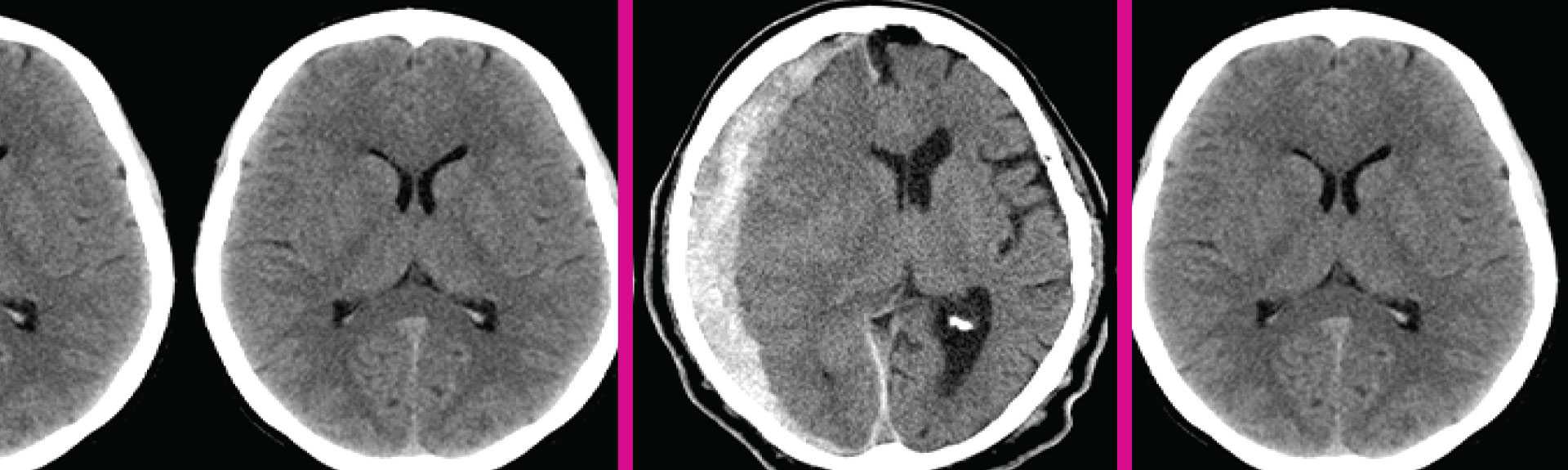
Mount Sinai Health System researchers have designed an automated deep learning neural network that can detect acute neurological diseases such as stroke, hemorrhage, and hydrocephalus within minutes, enabling earlier intervention that can preserve neurological function and improve patient outcomes.
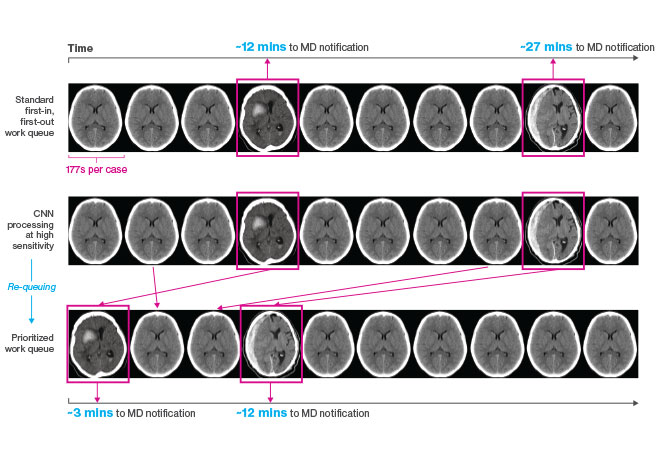
This is the first study to use artificial intelligence (AI) to identify acute neurological disease and thus demonstrate a direct clinical application of AI in a randomized controlled trial. Using 37,236 head CT scans, researchers trained a 3D conventional neural network(3D-CNN) to determine whether an image contained critical or non-critical findings, thus enabling triaging and queuing of radiology workflow for physician review based on the probability of acute neurological illness. Researchers subsequently conducted a randomized, double-blinded, prospective trial to assess the ability and quickness of the automated deep learning neural network to recognize and provide notification of a critical finding compared to results obtained by two radiologists and a neurosurgeon in a simulated clinical environment.
The results of the study, which were published in August 2018 in Nature Medicine, revealed that the 3D-CNN was able to process, interpret, and alert physicians to a critical finding in 1.2 seconds, compared to an average human time of three minutes. Researchers also noted a significant difference in the queue position of urgent versus routine cases in the 3D-CNN and human cohorts, with acute cases appearing earlier in the automated deep learning neural network queue.
“This is a system with the capacity to raise alarms about critical findings in mere seconds, and thus prevent these scans from potentially sitting in a queue for minutes, even hours, before they are assessed by a specialist,” says the study’s lead author, Eric K. Oermann, MD, an Instructor of Neurosurgery at the Icahn School of Medicine at Mount Sinai. “Our trial demonstrated that using artificial intelligence to identify cases of acute neurological illness works, and for that reason we should embrace this technology.”
“We know that rapid response is critical in the treatment of acute neurological illnesses,” says study co-author Joshua B. Bederson, MD, Professor and Chair of the Department of Neurosurgery at the Mount Sinai Health System, and Clinical Director of the Neurosurgery Simulation Core. “Any tools that decrease time to diagnosis may lead to improved patient outcomes.”
The study was conducted using weakly supervised learning approaches, which resulted in lower diagnostic precision for the 3D-CNN platform compared to physicians (0.48 versus 0.85) at a comparable sensitivity (0.79). Researchers are now focusing on re-engineering the platform using strongly supervised learning approaches and novel techniques that will enhance diagnosis accuracy. The expectation is that this work will be completed within the next two years.
“The application of deep learning and computer vision techniques to radiological imaging is a clear imperative for twenty-first century medical care,” says study author Burton Drayer, MD, the Dr. Charles M. and Marilyn Newman Professor and Chair of the Department of Radiology for the Mount Sinai Health System, and Dean for Clinical Affairs at the Icahn School of Medicine at Mount Sinai.
This study was performed by the Mount Sinai AI Consortium (AISINAI), a group of scientists, physicians, and researchers dedicated to developing AI platforms that enhance clinical diagnosis and patient care. AISINAI’s research builds on a paper published in January 2018 in Radiology that details the consortium’s work on natural language processing algorithms capable of identifying clinical concepts in radiology reports for CT scans.
Advancing the Use of Novel Technology in the OR
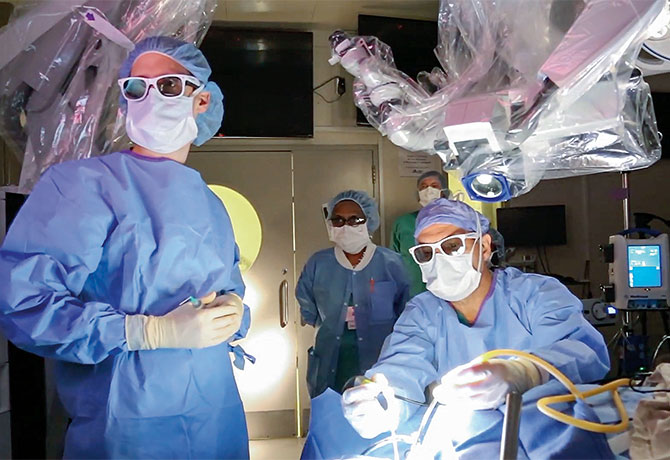
Over the past several years, the Mount Sinai Department of Neurosurgery has established itself as a leader in the development and use of innovative imaging, simulation, and augmented reality technologies to improve patient outcomes and solve neurosurgical challenges. Through novel technologies that were developed in house or were the result of collaborations with industry partners, today’s Mount Sinai neurosurgical patients are able to see 3D representations of their vascular and cranial nerves before surgery, and their surgeons have a vast array of increasingly sophisticated pre- and intraoperative tools to achieve greater outcomes.
The effort is being led by Joshua B. Bederson, MD, Professor and Chair of Neurosurgery for the Mount Sinai Health System and Clinical Director of the Neurosurgery Simulation Core, where there has been considerable investment in next-generation microscopy. Mount Sinai became one of the first health systems nationwide to use the ZEISS KINEVO® 900 microscope, a surgeon-driven robotic visualization system that not only streams 3D optical, navigation, and simulation information in 4K resolution into the microscope’s eyepiece, but also projects those data on large monitors in the operating room. This technology is combined with sophisticated 3D rendering of patient images, intraoperative navigation, and microscope integration. The 3D glasses provide an enhanced visualization of neuroanatomy. Mount Sinai also uses a micro-inspection surgical tool, the ZEISS QEVO®, which is engineered with an angled design, allowing the neurosurgeon to look around complex structures that are out of the microscope’s range.
The result is an augmented virtual reality tool that provides detailed, real-time information about critical structures that enhances a surgeon’s ability to remove difficult tumors with increased safety and efficiency. “Our goal is to integrate multiple information streams for surgeons to use with optical data, navigation, and simulation information all streaming into the surgeon’s viewpoint in a way that is not distracting and helps ensure the safety of the patient,” says Dr. Bederson.
The Mount Sinai Hospital, a U.S. News & World Report Honor Roll hospital, is also one of the first to adopt Modus V™ in the operating room. The cornerstone of Synaptive Medical, Inc.’s BrightMatter™ platform, Modus V is a hands-free, robotically controlled digital microscope that is capable of integrating and projecting navigation and data analytics on larger monitors. Moreover, the technology’s planning platform incorporates diffusion tensor imaging tractography, which enables surgeons to map out eloquent and non-eloquent areas of the brain during the approach phase and the resection using navigation.
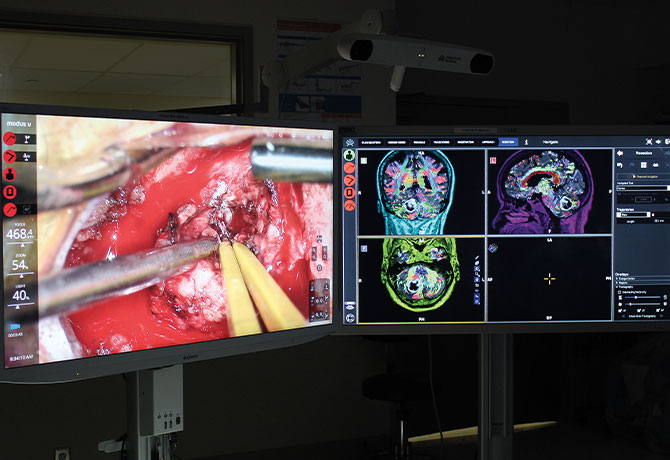
“This is a powerful alternative to traditional microscopy technology,” says Constantinos G. Hadjipanayis, MD, PhD, Chair of Neurosurgery at Mount Sinai Downtown-Union Square and Director of Neurosurgical Oncology for the Mount Sinai Health System. “Using this system during surgery, we can magnify tissue images to delineate tumors, and robotically adjust the arm position to achieve better visualization during tumor removal and avoid important nerve pathways that control movement and speech.”
Dr. Hadjipanayis says the resulting high-definition images of brain tractography and central nervous system fibers significantly enhance his ability to navigate each patient’s brain during surgery. “It is also ergonomically more comfortable to use because I can visualize the patient’s anatomy on a screen directly in front of me instead of looking down into the microscope. It sets the stage for the next generation of digital image-based neurosurgery.”
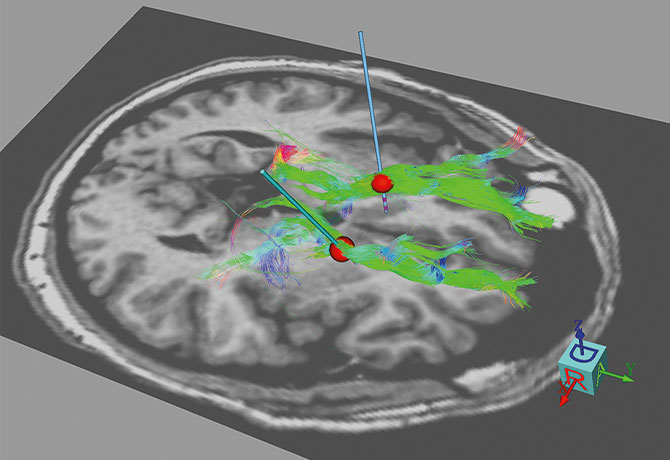
Mount Sinai’s Center for Advanced Circuit Therapeutics, headed by founding Director Helen S. Mayberg, MD, Mount Sinai Professor in Neurotherapeutics, and Professor of Neurosurgery, Neurology, Psychiatry, and Neuroscience, at the Icahn School of Medicine at Mount Sinai, is also enabling advances in tractography-guided targeting to test high-tech multimodal imaging tools that enhance deep brain stimulation (DBS) therapy. One such advance, StimVision, a software tool developed in collaboration with the McIntyre Lab at the Case Western Reserve University School of Medicine, enables real-time visualization of brain tracts and neural computational modeling of volume of tissue activated to facilitate more precise targeting and validation of electrode implantation targets among patients with depression.
“Conventional anatomical imaging does not enable identification of the optimal DBS electrode target—subcallosal cingulate,” says Ki Sueng Choi, PhD, who leads the Center’s Imaging Core and is an Assistant Professor of Radiology, and Neurosurgery, at the Icahn School of Medicine. “Using StimVision, we can visualize the white-matter pathways and remote connections of DBS stimulation settings in real time, enabling us to find and validate the optimal target in the operating room.”
The result has been an increase in clinical efficacy from 43 percent to 80 percent. Based on these results, the goal now is to extend the use of StimVision to patients with obsessive-compulsive disorders and movement disorders. For these efforts, Dr. Mayberg is collaborating closely with the clinical Center for Neuromodulation, led by Brian H. Kopell, MD, Professor of Neurosurgery, Neurology, Neuroscience, and Psychiatry. “We envision that using this technology will result in more precise targeting based on functional and structural connectivity, and enable us to visualize brain anatomical and functional changes with acute and chronic stimulation,” says Dr. Kopell. “Using that information, we will be able to minimize the adverse effects of DBS to the benefit of our patients.”
Constantinos G. Hadjipanayis, MD, PhD, receives compensation as a consultant for Synaptive Medical, Inc.
Using Vivid Displays of Vascular and Cranial Nerve Involvement With Pathology to Engage Patients
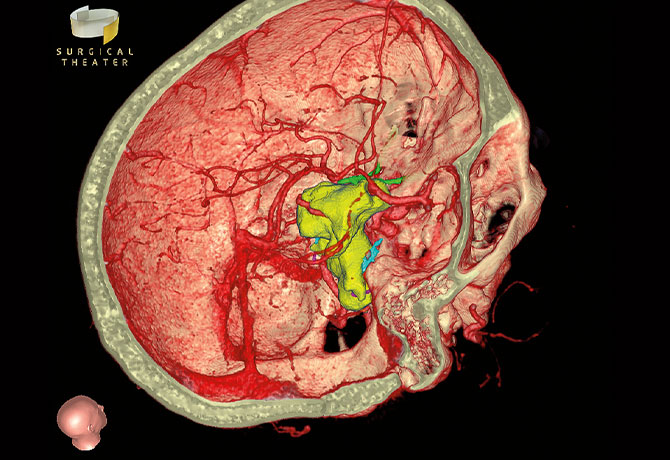
In complex intracranial surgery, the use of simulation has proved to be a valuable resource at The Mount Sinai Hospital, not only for surgeon preparation but also for patient engagement. Initially used to improve communication with patients as they consider a commitment to surgery, simulation has also increased patient confidence in the surgeon, enhanced patient awareness of the condition and upcoming surgery, and increased the likelihood of the patient choosing Mount Sinai for care. Patient-specific or pathology-similar simulations can be used during initial consultation, or pathology-specific simulations may be pulled from a carefully curated database of more than 600 previous simulations during the initial consultation. Patients may interact with images, videos, or virtual reality simulations depending on the level of interest.
In June 2018, 64-year-old patient David Wendrow came to Mount Sinai for a follow-up appointment. He had had a large cerebellopontine angle epidermoid cyst that had been resected twice, first in 2006 by Kalmon D. Post, MD, former Chair of the Department of Neurosurgery, and then in January 2017 by Joshua B. Bederson, MD. Mr. Wendrow had been experiencing a progression of symptoms, including double vision, a slight drooping of the right eyelid, and occasional new left-sided facial numbness. A recurrent expansile growth of 1-4 mm in multiple directions, seen on MRI and CT DICOM images, showed the tumor displacing the brainstem and cranial nerves.
Having had familiarity with advanced digital technologies offered to him before his 2017 surgery, Mr. Wendrow requested a 3D simulation (by Surgical Theater, LLC) of the larger recurrent growth, which provided a patient-specific representation of anatomy using volumetric CTA and MRI images fused together and segmented (see Figure 1). Leslie Schlachter, PA-C, Clinical Director of Neurosurgery at The Mount Sinai Hospital, and the simulation team also created a video that displayed Mr. Wendrow’s tumor from his 2017 surgery along with the residual tumor from postoperative MRI scans and areas of regrowth after his visit in June 2018,
highlighting vascular and cranial nerve involvement. During his simulation consultation visit in July 2018, 3D representations of his pathology and surgical plan were demonstrated on various platforms to Mr. Wendrow. The 3D representation of his anatomy was used to explain the various steps of surgery, including skin incision and craniotomy, and to identify critical structures that may cause possible side effects during the resection phase. Using an HTC VIVE VR headset to experience the patient-specific virtual environment, Mr. Wendrow was able to see the inside of his own head to help him better understand these potential side effects and their relationships with anatomy (see Figure 2).
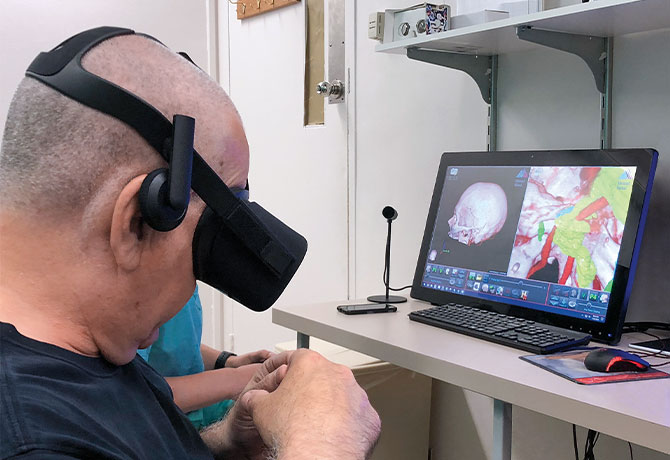
For Mr. Wendrow, the ability to compare growth and see intricate involvement of his recurring epidermoid cyst was especially valuable. Mr. Wendrow is back to baseline and able to return to his routine of going to the gym, driving, and running errands with ease. In other cases, when time is limited or where scan quality is not as great, patients can still benefit from seeing similarly located pathology in order to understand the treatment plan and postoperative outcome and potential side effects or changes.
Joshua B. Bederson, MD, Professor and Chair of Neurosurgery for the Mount Sinai Health System, owns equity in Surgical Theater, LLC, and has contracted research agreements with Brainlab.
Study Findings Provide Insight Into How Viruses May Play a Role in Alzheimer’s Disease
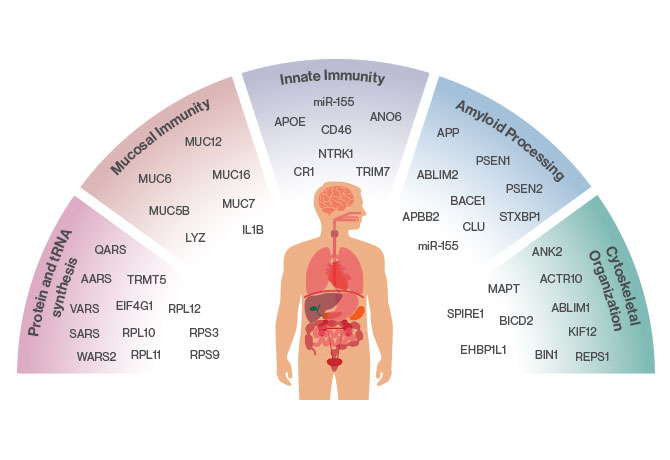
Two strains of human herpesvirus—human herpesvirus 6A (HHV-6A) and human herpesvirus 7 (HHV-7)—are found in the brains of individuals with Alzheimer’s disease at levels up to twice as high as in those without Alzheimer’s, according to findings published June 21, 2018, in Neuron by a team of researchers at the Icahn School of Medicine at Mount Sinai.
It is the first study to use an entirely data-driven approach to examine the impact of viruses on Alzheimer’s and to identify potential disease-associated roles of these particular viruses, which was an unexpected discovery. The researchers had been comparing genetic data in healthy and postmortem Alzheimer’s brains to identify possible new drug targets for Alzheimer’s disease when their complex computations revealed the unusually high levels of the viral genomes.
“This study represents a significant leap forward in our understanding of how viruses may play a role in Alzheimer’s disease,” says the study’s senior author, Joel Dudley, PhD, Mount Sinai Professor in Biomedical Data Science, and the Director of the Institute for Next Generation Healthcare at the Icahn School of Medicine at Mount Sinai. “We were able to not only measure these viruses using computational techniques, but also build out the networks in which these viruses are operating and influencing known Alzheimer’s genes.”
The study was enabled in part by powerful new molecular profiling data released by the National Institute on Aging (NIA) Accelerating Medicines Partnership-Alzheimer’s Disease, a collaboration among industry, government, and nonprofit organizations dedicated to identifying new targets for preventing or treating the disease. Using those data, the team conducted computer mapping of four brain regions from more than 600 samples from the Mount Sinai Brain Bank, which for the last 36 years has been examining postmortem brain tissue from donors for Alzheimer’s-related research.
As the study progressed, the team found the higher levels of HHV-6A and HHV-7 viruses, which were subsequently confirmed using data from brain banks in three other major NIA Alzheimer’s Centers. Researchers also discovered that the viruses appeared to accelerate the development of Alzheimer’s-related plaques and tangles in the brain. “Our team was initially quite skeptical about these findings,” says the study co-senior author, Sam Gandy, MD, PhD, Mount Sinai Professor in Alzheimer’s Research, and the Director of the Center for Cognitive Health and NFL Neurological Care at Mount Sinai. “Gradually, we became convinced that what we were seeing is a real property of Alzheimer’s brains in several internationally recognized brain banks and that we ought to report it, even if we cannot completely explain what these viruses are doing in the brain.”
Although the study does not prove a direct causal role between these herpesviruses and Alzheimer’s disease, or indicate what activates the viruses, the findings do lend credence to the hypothesis that these herpesviruses may trigger progression of dementia in Alzheimer’s. This work also opens new avenues for the identification of subpopulations and biomarkers, which would not only make it easier to diagnose and determine a person’s risk of developing Alzheimer’s, but also possibly create opportunities to use existing antiviral drugs and drugs that stop the brain’s immune cells from responding to these viruses.
Prominent Neuroimaging Researcher Trey Hedden, PhD, Joins Mount Sinai
Trey Hedden, PhD, a leader in neuroimaging research, has joined the Icahn School of Medicine at Mount Sinai as Associate Professor of Neurology, Radiology, and Neuroscience, and Director of Neuroimaging and Biomarker Research in Aging and Alzheimer’s Disease.

Dr. Hedden’s laboratory focuses on integrating multiple brain markers to help build a comprehensive picture of the relationship between brain function and cognition during aging and neurodegenerative disease. His published work, appearing in more than 75 primary research articles, examines behavioral and neuroimaging measures of age-related changes in memory and executive control processes. Dr. Hedden’s current research integrates measures of functional MRI with PET markers of tau and amyloid accumulation, PET markers of dopamine dysfunction, PET markers of glucose metabolism, and MRI markers of white matter to target potential preclinical Alzheimer’s-related neuropathology in otherwise normal older individuals.
Before joining Mount Sinai, Dr. Hedden was Associate Professor of Radiology at Harvard Medical School and an investigator for the Athinoula A. Martinos Center for Biomedical Imaging at Massachusetts General Hospital.
How a Long-Ago Lab Discovery by Neurologist Leads To Treatment That Received Approval in 2018
Foundational work by Stuart Sealfon, MD, that was performed two decades ago at the Icahn School of Medicine at Mount Sinai for a new oral non-opioid medication to help manage pain associated with endometriosis recently received U.S. Food and Drug Administration (FDA) approval.
The drug, Orilissa™, is the first oral regimen that specifically helps to ease the moderate to severe pain that accompanies endometriosis, a condition where the tissue that forms in the lining of the uterus continues to grow outside the uterus.

The disorder, which affects roughly one in ten women of reproductive age, negatively impacts quality of life, since the excess tissue growth is often accompanied by pain during menstruation, intercourse, or urination.
“Orilissa is a drug that resulted from the basic research we conducted at Mount Sinai, and it will help millions of women,” says Dr. Sealfon, Sarah B. and Seth M. Glickenhaus Professor and Chair Emeritus of the Department of Neurology. “At Mount Sinai, we discovered how to clone the drug target that was needed to develop this new drug.”
Indeed, as a young researcher more than two decades ago, Dr. Sealfon led the Mount Sinai team that cloned the gonadotropin-releasing hormone receptor (GnRHR) and genetically engineered host cells that express GnRHR. Gonadotropin-releasing hormone (GnRH), which is secreted by the hypothalamus, plays a key role in controlling reproduction and acts via its receptor GnRHR.
The cloning procedure and primary structure of the receptor were described in two studies authored by Dr. Sealfon in 1992 and 1993, which were published in Molecular Endocrinology and Molecular and Cellular Endocrinology, respectively.
The research provided a better understanding of the complex interplay of hypothalamic, pituitary, and gonadal hormones, which underlie pharmacotherapy and the reproductive system. At the time, Dr. Sealfon says, a career development grant provided him with the funding he needed to conduct his research. Two U.S. patents in 1998 and 1999 assigned these inventions to Mount Sinai.
The oral application of Orilissa—also known by its generic name, elagolix—enables women to dial down the reproductive system. The dose-dependent drug suppresses the luteinizing hormone and the follicle-stimulating hormone, which leads to decreased blood concentrations of estradiol and progesterone. This reduces the growth of excess tissue, or lesions that form on the ovaries, fallopian tubes, or areas near the uterus, including the bowel and bladder, that characterize endometriosis and cause pain. The drug was released by AbbVie, a global pharmaceutical company, in cooperation with Neurocrine Biosciences, Inc.
Endometriosis is considered one of the most common gynecologic disorders in the United States, but women can sometimes go years before having the laparoscopic procedure needed to render a proper diagnosis. In addition to the use of oral contraceptives, treatments have included nonsteroidal anti-inflammatory drugs and opioids. In more extensive cases, women may undergo surgical procedures, including a hysterectomy.
In two phase III clinical trials, Orilissa has been shown to be helpful in the treatment of uterine fibroids as well. Fibroids are common benign tumors that cause bleeding or pain in millions of women, and for which there are, currently, limited nonsurgical treatment options.
In the years since his initial discovery, Dr. Sealfon’s lab has continued to study GnRH receptor-mediated gonadotropin regulation and has helped guide future work in the field.
Mount Sinai’s Pioneering Work in Neuro-Otology
Joanna C. Jen, MD, PhD, a physician-scientist with a special interest in the genetic and physiological basis of neurological disorders affecting eye movement control, balance, and coordination, recently joined the Icahn School of Medicine at Mount Sinai as Chief of the Division of Neuro-Otology and Neurogenetics in the Department of Neurology. Dr. Jen, who also has been named Dr. Morris B. Bender Professor of Neurology, and Professor of Neurosurgery, and Otolaryngology, aims to create a comprehensive multidisciplinary clinical care and research program that spans the population health approach, from front-line evaluation and management of dizziness and imbalance, to precision medicine-based diagnosis and treatment for rare disorders of cerebellar maldevelopment and degeneration.

Dr. Jen will build upon the groundbreaking work of her predecessors—the late Morris B. Bender, MD, a pioneer in the neurology of the ocular motor system and a former Chair of Neurology at Mount Sinai, and Bernard Cohen, MD. An internationally renowned scientist and clinician who helped launch the new field of vestibular research, Dr. Cohen and his collaborators were the first to characterize the semicircular canal-induced eye movements in many species, including monkeys and humans. He also helped discover a special neural mechanism in the brainstem, known as the velocity storage integrator, which integrates linear acceleration from the semicircular canals and acts as a neural gyroscope to orient eye movements to gravity and the spatial vertical.
Dizziness and imbalance are common symptoms with many different causes that pose a diagnostic challenge. Dr. Jen will oversee the close collaborative efforts between neuro-otology and programs in otolaryngology, neuro-ophthalmology, headache, and physical therapy throughout the Mount Sinai Health System to facilitate the successful management of patients with complex brain and ear diseases. The Human Balance Laboratory established by Dr. Cohen and directed by Mingjia Dai, PhD, from 1992 until his recent passing, will continue to provide exceptionally rigorous testing of vestibular and ocular motor function. It was under Dr. Dai that the lab developed the first effective treatment for mal de débarquement syndrome (MdDS), which, in collaboration with Sergei Yakushin, PhD, has since treated more than 500 MdDS patients.
Mount Sinai is the only center in New York State that is actively engaged in National Institutes of Health-funded multicenter clinical trials to study new treatment for vestibular migraine, as well as novel approaches in the diagnostic algorithm for dizziness in the emergency room. Philanthropic support for research in neurogenetic disorders and dizziness including MdDS is growing. Dr. Cohen, who will continue his research at Mount Sinai as the Dr. Morris B. Bender Professor Emeritus, has generously provided funding not only for the first fellow in neuro-otology to begin in 2019-2020, but also for research into the pathophysiologic mechanisms underlying MdDS.

Department Chairs
Barbara G. Vickrey, MD, MPH
Henry P. and Georgette Goldschmidt
Professor and Chair of Neurology
Mount Sinai Health System
Joshua B. Bederson, MD
Professor and Chair of Neurosurgery
Mount Sinai Health System
Clinical Director of the Neurosurgery
Simulation Core