The Dr. Henry D. Janowitz Division of Gastroenterology
Our mission is excellence in the delivery of patient care, research, and education in diseases of the GI tract.
The Dr. Henry D. Janowitz Division of Gastroenterology Including GI Surgery
Innovative, translational research that makes a real difference in our patients’ lives continues to be the hallmark of the Mount Sinai Health System’s Division of Gastroenterology.
This specialty report highlights:
- “Resetting” the immune system in Crohn’s disease
- Artery occlusion identifies resectable tumors
- Treating to target in Crohn’s disease
- New tools and therapies for liver cancer
- An app that helps IBD patients manage symptoms
- Teaching fellows how to teach
- Welcoming new guidelines for colorectal screening
- Message from the Chief: Bruce E. Sands, MD, MS
“Resetting” the Immune System in Crohn’s Disease
A Mount Sinai Health System physician is leading an innovative clinical trial to determine if combining stem cell transplantation with drug therapy can lead to better outcomes for thousands of Crohn’s disease patients who do not respond to existing therapies.
Louis Cohen, MD, MS, Assistant Professor of Medicine (Gastroenterology) at the Icahn School of Medicine at Mount Sinai and Principal Investigator of the study, says extensive studies of hematopoietic stem cell transplantation (HCT) in Europe have demonstrated response rates exceeding 50 percent among patients, with complete healing of the bowel. But researchers are not sure why this is the case.
“This clinical study is designed to answer that question,” Dr. Cohen says. “We think that by suppressing the patients’ immune system and giving them back their own stem cells, we are resetting their immune system to a pre-disease state.”
Candidates for this open trial are assessed by a multidisciplinary team for suitability and enrolled in the protocol once disease activity and distribution are confirmed by endoscopy and MRI. They are then weaned off their current medication and undergo stem cell mobilization,
a procedure in which they receive chemotherapy and growth factors to promote stem cell production. The stem cells are then collected using leukapheresis, and, once enough cells have been gathered based on the patient’s weight, participants will undergo HCT, which involves chemotherapy and two to four weeks of hospitalization for regeneration of the immune system.
This clinical trial is unique in that it adds a medication to the protocol following transplantation. Patients will receive eight doses of vedolizumab once their immune systems have regenerated. Dr. Cohen expects this will prevent newly activated immune cells from returning to the gut and restarting the inflammation that characterizes Crohn’s disease, and will lead to better outcomes in cases where the disease returns.
“Studies have shown the disease recurs among most patients who have undergone HCT,” Dr. Cohen says. “In resetting the immune system, we have a better opportunity to control the disease because we are catching it at an earlier stage where it may be more responsive to medicine. By combining vedolizumab with HCT, the goal is to help patients feel better in the short term and maintain positive outcomes over the long term.”
Participants will be monitored for six months following HCT through blood work and stool tests, and then undergo a colonoscopy and an MRI to assess the healing of the bowel. Those who respond to treatments can remain on vedolizumab indefinitely with the approval of their referring physician. Those who do not respond can leave the trial if evidence suggests that the vedolizumab is not working. They will then be prescribed other medications.
Only one patient has undergone combined HCT and vedolizumab therapy to date, but Dr. Cohen says the results have been impressive. “He has been able to cut his trips to the bathroom from approximately 20 per day to 10 per day, or five with one Imodium®. His inflammation is under control, he is able to play soccer with his kids, and he feels healthier than he has in years. We’re very happy with what we’re seeing so far.”
Dr. Cohen plans to enroll up to 50 patients in the clinical trial, at which point he hopes a protocol and program will be in place offering a safer, more efficacious treatment for patients, either through the advent of adjuvant drug therapies or a better understanding of which cell populations to target. “We may be able to develop the protocol in such a way that it is applicable to many more patients than just those who are very sick and unresponsive to current therapies,” Dr. Cohen says.
Physicians who wish to refer patients for enrollment in this clinical trial may do so by emailing crohnstransplant@mssm.edu.
Artery Occlusion Identifies Resectable Tumors
Every year, approximately 500 patients with newly diagnosed neuroendocrine tumors of the small intestine are referred to The Center for Neuroendocrine and Carcinoid Tumors at The Mount Sinai Hospital. Often asymptomatic and diagnosed at an advanced stage, these tumors typically spread to mesenteric arteries, making resection challenging, if not impossible.
In such cases, “the only therapeutic option traditionally has been medical management of symptoms,” says Celia Divino, MD, the Stanley Edelman, MD Professor of Surgery at the Icahn School of Medicine at Mount Sinai, and Chief of General Surgery at The Mount Sinai Hospital. “We decided to apply our extensive expertise in neuroendocrine tumors to the development of a resection procedure that enables more patients to achieve symptomatic relief and disease-free survival.”
In collaboration with Michael Marin, MD, a Professor and the System Chair of Surgery (Vascular Surgery) at the Icahn School of Medicine, and Robert Lookstein, MD, a Professor of Radiology and Surgery at the Icahn School of Medicine, Dr. Divino developed endovascular occlusion tumor excision (EVOTE). This novel hybrid procedure uses occlusion of mesenteric blood vessels to determine whether neuroendocrine tumor patients are eligible for resection.
“Other medical centers do not have this type of hybrid technique,” Dr. Divino says. “By performing an occlusion, we are able to offer more patients the opportunity to undergo resection after other centers have rejected them as candidates for surgical removal of their tumors. What could be better than that?”
Patients are eligible for EVOTE if they have been diagnosed with a neuroendocrine tumor that is deemed a risk to, or has invaded, their mesenteric nodes, so long as it has not spread to other organs. Each patient undergoes mesenteric angiography, in which a catheter is threaded through an artery to the mesenteric artery that is supplying blood to the tumor. Using a contrast dye, Dr. Divino analyzes the blood flow and then inflates a balloon to occlude the blood vessel.
“If the patient experiences pain, it means that the blood vessel is the main source of blood to the small intestine and we cannot resect it,” Dr. Divino says. “If there is no pain, that means that there is collateral blood supply to the intestine, and we can proceed with coil embolization of the blood vessel and resection. In that way, the occlusion serves as a feasibility test, but it also enables us to limit the amount of intestine we need to remove, which is vital for the patient’s long-term survival.”
Following overnight monitoring, patients undergo resection of the tumor via laparotomy. “With carcinoids, there are several factors that come into play, such as fibrosis (desmoplastic reaction), that complicate surgery,” Dr. Divino says. “We not only have the necessary multidisciplinary expertise to treat these unique tumors at our Center, we also have the guidance of specialists such as gastroenterologist Michelle Kim, MD, and the Director of our Center, oncologist Edward Wolin, MD. That is why we see so many referrals at Mount Sinai.”
More than 20 patients have undergone EVOTE since it was introduced in 2014, and Dr. Divino would like to see it become the standard approach for patients thought to have unresectable neuroendocrine tumors. “This is a highly effective procedure that should be done everywhere,” she says.
Treating to Target in Crohn’s Disease
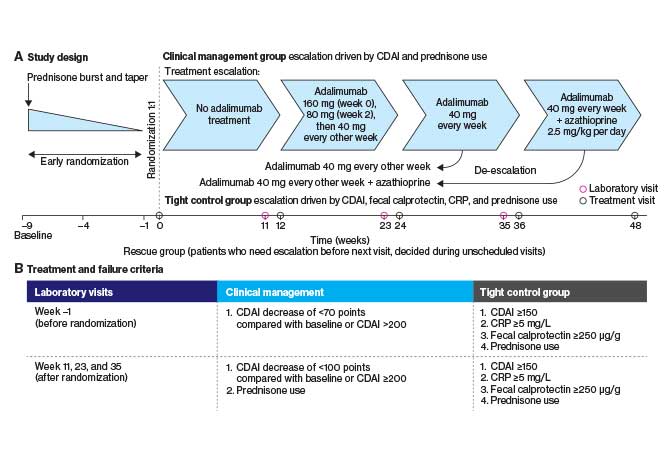
A tight control, treat-to-target approach can lead to improved outcomes for Crohn’s disease patients, particularly when coupled with early intervention, an international clinical trial led by the Mount Sinai Health System has demonstrated.
Overseen by Jean-Frederic Colombel, MD, Professor of Medicine (Gastroenterology) at the Icahn School of Medicine at Mount Sinai, the trial, known as CALM, compared endoscopic and clinical outcomes among patients who received treatment based on symptoms and patients who received the same treatments based on symptoms and two inflammation biomarkers: fecal calprotectin and C-reactive protein (CRP).
In total, 244 patients were recruited by 74 hospitals in 22 countries for this phase III trial. The criteria were limited to Crohn’s disease patients between the ages of 18 and 75 with a median disease duration of one year, no history of immunomodulator or biologic use, a Crohn’s disease
endoscopic index of severity greater than six, and a Crohn’s disease activity index (CDAI) of 150-450. Patients were randomized on a one-to-one basis following eight weeks of prednisone induction therapy, but some were randomized earlier based on their symptoms.
Participants who met the failure criteria one week before randomization—a CDAI decrease of <70 points compared with baseline or a CDAI of >200 for the clinical management group, and a CDAI ≥150 points or a CRP of ≥5 mg/L or a fecal calprotectin ≥250 μg/g and prednisone therapy for the tight control group—received 160 mg adalimumab at week zero, 80 mg at week two and 40 mg every other week thereafter.
At 12 weeks, patients who met the failure criteria—a CDAI decrease of <100 points compared with baseline or a CDAI ≥200, plus prednisone use, for the clinical management group, and a CDAI ≥150, a CRP ≥5 mg/L, a fecal calprotectin ≥250 μg/g, and prednisone use for the tight control group—were also escalated to 40 mg adalimumab every other week.
Patients who continued to meet these failure criteria were further escalated to 40 mg adalimumab each week at week 24, and to 40 mg adalimumab each week and 2.5 mg/kg azathioprine daily at week 36. Participants who did not meet these criteria remained on their previously prescribed treatments. At 24 and 36 weeks after randomization, patients whose symptoms and biomarkers decreased were deescalated from weekly adalimumab to every other week but their azathioprine intake remained unchanged.
The clinical trial results, which were published in the December 23, 2017, edition of The Lancet, revealed that 46 percent of tight control patients reached the primary endpoint of mucosal healing with an absence of deep ulcers 48 weeks after randomization, compared to 37 percent of clinical management group patients. Although the overall rate of adverse events among tight control patients was higher (86 percent) than that of the clinical management group (82 percent), the rate of serious adverse events was lower among tight control patients.
“This is the first prospective study to demonstrate that following patients based on both clinical symptoms and objective biomarkers of disease activity is leading to better outcomes,” Dr. Colombel says. “Mount Sinai was an early adopter of a three-pronged approach focused on early intervention, treat-to-target, and tight control. This clinical trial served as proof of concept and demonstrates that we are leading the way in proposing strategies that block the progression of this disease.”
New Tools and Therapies for Liver Cancer

Death rates for almost all types of cancer have gone down in recent years, but one exception is liver cancer, an aggressive malignancy that often goes undiagnosed until its final stages. Deaths from the cancer have spiked sharply, especially in the United States, where they have increased 43 percent, from 7.2 deaths per 100,000 people in 2000 to 10.3 deaths per 100,000 people in 2016, according to a recent report by the U.S. Centers for Disease Control and Prevention.
Josep Llovet, MD, Co-Director of Mount Sinai’s Liver Cancer Program, says that newly diagnosed cases of liver cancer have also risen “spectacularly in the United States,” and the World Health Organization has projected the incidence of the cancer to exceed a million new cases globally by 2025.
At Mount Sinai, Dr. Llovet and his colleagues—a multidisciplinary group of surgeons, hepatologists, oncologists, pathologists, and other specialists—are working to develop new diagnostic tools and therapies that could help increase patients’ survival rates by identifying the disease earlier and treating it more effectively.
Multiple factors that vary by geography can put people at risk for liver cancer. In Asia and sub-Saharan Africa, the primary risk factor is hepatitis B. “Many liver cancer patients at Mount Sinai are of Asian descent, a high-risk population because of the prevalence of hepatitis B infection in Asia,” Dr. Llovet says.
In the United States, the upsurge can be largely accounted for by two groups. Baby boomers who were infected with hepatitis C in the 1980s or 1990s are now developing cancer, a trend that is likely to trail off now that effective treatment for hepatitis C is available. More worrisome is the growing number of Americans who have fatty deposits in the liver as a result of obesity, raising their cancer risk.
The Mount Sinai program is the largest of its kind and is part of The Tisch Cancer Institute, which is unusual among National Cancer Institute—designated cancer centers in having a specific program for liver cancer. The program includes a tissue bank, has 24 experts on staff, and has taken the lead in five international clinical trials.
The tissue bank, which now contains 3,000 samples that include all types of liver malignancies, has allowed the program’s research team to work on classifying tumors into distinct molecular subtypes based on the genes expressed by the cancer, giving researchers targets for potentially effective new therapies.
The last few years have seen a revolution in the management of hepatocellular carcinoma (HCC), the most common type of liver cancer, accounting for about 75 percent of cases. The first systemic therapy to improve survival time for patients with late-stage HCC, sorafenib, was approved for use in 2007. Other new drugs have followed.
Still, one of the biggest obstacles to successful treatment remains the fact that in most patients, liver cancer is far advanced by the time it is diagnosed. Augusto Villanueva, MD, PhD, an Assistant Professor of Medicine at the Icahn School of Medicine at Mount Sinai and a member of the Liver Cancer Program team, is hoping to address that problem by developing a liquid biopsy that is capable of detecting tumors in their early stages, when they produce few obvious symptoms. The biopsy method would use blood components like DNA and circulating tumor cells to identify the cancer in higher-risk patients.
“The idea would be to take a blood sample from a patient, analyze these components to see if traces of tumor were in the blood, and say, ‘This patient is very likely to have a tumor,’ so we can detect it earlier and potentially cure it,” Dr. Villanueva says.
He says the technology for the liquid biopsy is in place, but, “We need to validate that it works in actual patients that are at risk for liver cancer.” He adds, “I think we can have a liquid biopsy—based biomarker for early liver cancer detection within the next five years.”
App Helps IBD Patients Manage Symptoms
Thousands of inflammatory bowel disease (IBD) patients will be able to better manage the symptoms and anxiety commonly associated with the disease, thanks to a new app developed at the Mount Sinai Health System.
Designed by Mount Sinai’s AppLab at the Division of Gastroenterology, HealthPROMISE is an innovative, cloud-based, patient-reported outcome tool that facilitates decision making among IBD patients and their care providers. Patients track their symptoms, quality of life, and 17 quality metrics that range from flu shots and pneumonia vaccines to inflammation control, and physicians and care navigators can access and analyze the data in real time to suggest interventions that improve outcomes.
Ashish Atreja, MD, MPH, Associate Professor of Medicine (Gastroenterology) at the Icahn School of Medicine at Mount Sinai and Director of the AppLab, says HealthPROMISE improves the quality of care for IBD patients and has the potential to reduce the anxiety often associated with IBD by giving patients clear, detailed information on how well they are managing their disease.
“For the first time, patients can see how they are doing with the knowledge that, if their situation changes, they can get the right care at the right time to help them get back on track and keep the disease under control,” Dr. Atreja says.
A two-year clinical trial involving 320 Mount Sinai IBD patients demonstrated HealthPROMISE’s efficacy. Dr. Atreja says the 162 patients randomized into the intervention arm with access to the app achieved a significant increase in quality metrics, rising from 50 percent at baseline to 88 percent. Quality metrics among control-arm patients who only had access to education resources rose to 65 percent.
HealthPROMISE is one of several innovative digital medicine projects designed and developed by AppLab. Launched by Dr. Atreja in 2012, AppLab has become a model for medical centers in creating and using technological platforms to advance research and enhance patient outcomes.
“We were one of the first health systems in the country to implement a dedicated app lab,” Dr. Atreja says. “We have developed a mature team and processes for app design, development, and integration with electronic health records, which give us a competitive edge in this field. AppLab has also facilitated our talent recruitment efforts, which will enable us to build on our legacy of innovation in patient-centered apps and analytics.”
HealthPROMISE is being made available to other health providers and payers through the creation of RX.Health, a spinoff venture that is helping health systems embrace digital medicine through its RXUniverse prescription platform. Dr. Atreja’s goal is to expand collaborations with IBD centers nationwide and use RXUniverse to prescribe HealthPROMISE to the centers’ patients, resulting in a digital transformation network of up to 15,000 patients using the app, and supporting IBD home- and value-based programs. He has also forged a partnership with Crohn’s and Colitis of Canada and five Canadian IBD centers of excellence to deliver HealthPROMISE to their patients.
Dr. Atreja is now exploring opportunities to adapt HealthPROMISE for use in other specialties. He sees great potential for this technology to reach populations that have traditionally been underserved in terms of health care delivery, and he is looking at adding tools such as chatbot, artificial intelligence, and voice interface to facilitate those connections.
“Since the majority of underserved patients have access to smartphones, we believe this technology will serve as a digital bridge for communicating with patients and engaging them in their health care,” he says.
Teaching Fellows How to Teach

While completing his gastroenterology fellowship at The Mount Sinai Hospital, Kamron Pourmand, MD, saw a need for more opportunities to develop his teaching skills and prepare for independent practice.
“During my internal medicine training, there was this whole concept of graded autonomy. Senior residents ran the rounds and led the instruction of interns and medical students, with the attending physician serving more in an advisory role,” says Dr. Pourmand, who is now the Chief Fellow of the Hospital’s Division of Liver Diseases.
“I gained the confidence and skills needed to teach internal medicine to junior trainees, but there was nothing comparable to that in my gastroenterology fellowship. I thought, ‘How can we expect fellows to effectively teach gastroenterology-related medical reasoning and technical procedures such as endoscopy if we never put them in the hot seat during training?’”
Dr. Pourmand subsequently developed the Fellow as Teacher (FAT) rotation, an innovative curriculum that equips third-year inpatient gastroenterology fellows with the necessary knowledge and skills to teach gastroenterology practices and procedures to junior trainees. During the two-week program, these fellows effectively serve as co-attendings, leading the instruction of first- and second-year fellows during inpatient consults and endoscopic procedures. An attending physician is always present, providing real-time assistance where required and feedback on teaching abilities.
“The goal of FAT is to improve teaching skills and ease the difficult transition to independent practice,” Dr. Pourmand says. “Giving fellows a greater degree of autonomy and responsibility will likely reduce the pressure that often accompanies making decisions on their own or teaching trainees once they graduate.”
Dr. Pourmand, who developed FAT under the mentorship of Brijen Shah, MD, an Associate Professor of Medicine (Gastroenterology) and of Medical Education at the Icahn School of Medicine at Mount Sinai, is not aware of any other gastroenterology training program that has integrated a comparable teaching-focused curriculum. “After reviewing the literature, I could not find any programs that enable third-year fellows to act as attending physicians under direct supervision for the purposes of gaining teaching skills,” he says. “Everyone at Mount Sinai, from the fellows to supervising physicians to the program leadership, recognized the value of FAT and supported me in making it a reality.”
FAT was officially integrated into the inpatient gastroenterology program at The Mount Sinai Hospital in March 2018 following several successful pilot trials. Dr. Pourmand led instructional sessions on teaching skills, providing effective feedback, and procedural teaching for senior fellows before launch. He then joined his three third-year co-fellows as a program participant. “I surveyed the other fellows before and after the program and it was clear that they felt better prepared to act as inpatient consult supervisors and train junior fellows in endoscopy as a result of the curriculum,” he says.
Dr. Pourmand has made a few enhancements to FAT for its second year, such as empowering attending physicians to incorporate more of their own teaching approaches alongside the senior fellows. He is also developing a formal syllabus for the curriculum that includes an outpatient module that enables senior fellows to precept junior fellows in the clinic setting, and has plans to launch it throughout the Mount Sinai Health System. Eventually, he sees the curriculum expanding to other health systems nationwide.
“Ultimately, I hope that FAT equips fellows with teaching techniques they can use after graduation and inspires others to develop more efficient and effective approaches to learning, because that will benefit gastroenterology trainees and patients alike,” Dr. Pourmand says.
Welcoming New Guidelines For Colorectal Screening
Gastroenterologists at The Mount Sinai Hospital—widely recognized as innovators in enhancing access to colorectal cancer screening—are welcoming American Cancer Society (ACS) guidelines that recommend that people with an average risk for colon cancer start screening at age 45, not 50.
“I am very supportive of this new recommendation and the science behind it,” says David Greenwald, MD, a Professor of Medicine (Gastroenterology) at the Icahn School of Medicine at Mount Sinai. “The statistical modeling that formed the basis for this recommendation is sound; anything we can do to prevent colon cancer, which is a largely preventable disease, is worth the effort.”
The ACS guidelines follow the 2017 release of statistics indicating that, from 2000 to 2013, colorectal cancer incidence rates increased 22 percent among adults under the age of 50. Although other organizations have yet to revise their guidelines in light of the new statistics, Dr. Greenwald, who also serves as Director of Clinical Gastroenterology and Endoscopy at The Mount Sinai Hospital, expects the changed ACS guidelines may have a major impact on the average age of screening.
“We know many people aren’t screened at 50, but instead wait until age 53 or 54 or later due to procrastination, because they fear the procedure or the results,” he says. “Changing the guidelines will help to start the conversation at an earlier age, encouraging more people to be evaluated. Symptoms such as rectal bleeding should always be evaluated at any age.”
Dr. Greenwald also commends the new guidelines for providing a menu of screening options for patients to consider instead of recommending one specific test for all patients. These include stool-based tests and visual exams such as colonoscopy, sigmoidoscopy, and CT colonography. “All of these tests are appropriate, but ultimately the best test is the one that gets done,” he says.
Message from the Chief: Bruce E. Sands, MD, MS
Innovative, translational research that makes a real difference in our patients’ lives continued to be the hallmark of the Mount Sinai Health System’s Division of Gastroenterology in 2018.

This was exemplified by the clinical trial combining stem cell transplantation with drug therapy for Crohn’s disease, led by Louis Cohen, MD, MS. By “resetting” the immune system while preventing the newly activated immune cells from returning to the gut, Dr. Cohen was able to dramatically improve the health of the first patient in this study, and new protocols for safer and more efficacious treatments are in development.
Jean-Frederic Colombel, MD, PhD, and colleagues showed in a randomized controlled trial that a treat-to-target approach incorporating inflammatory biomarkers produces better outcomes in Crohn’s disease than treating based on clinical symptoms alone.
Our pioneering work with smartphone apps for managing inflammatory bowel disease continues. The latest, HealthPROMISE, achieved a significant increase in quality metrics in a clinical trial comparing it to standard educational materials. It is now being made available to other health care providers and payers via the Mount Sinai spinoff Rx.HEALTH.
Our colleagues in the Division of Liver Diseases are developing new diagnostic tools and therapies to cope with a worldwide explosion in liver cancer, and in GI surgery we are pioneering a method to resect advanced neuroendocrine tumors of the small intestine.
Our innovations in medical education are exemplified by the work of Kamron Pourmand, MD, who developed the “Fellow as Teacher” rotation at The Mount Sinai Hospital, nationally ranked by U.S. News & World Report. This program helps young gastroenterologists learn the skills to teach others about the field.
In short, we continue to advance and innovate on many fronts, and are looking forward to further progress in 2019.