
Catherine and Henry J. Gaisman Division of Pulmonary, Critical Care and Sleep Medicine
We provide state-of-the-art consultation, diagnostic testing and clinical and multidisciplinary care to patients with simple and complex respiratory and respiratory-related diseases.
Catherine and Henry J. Gaisman Division of Pulmonary, Critical Care and Sleep Medicine
With the Mount Sinai – National Jewish Health Respiratory Institute, we have created a model for delivering patient-centered, multidisciplinary, state-of-the-art care for patients with simple and complex respiratory diseases.
This specialty report highlights:
- Our smartphone app for asthma, which is shown to ease collection of patients and data
- A multidisciplinary effort that has lowered COPD readmissions
- New guidelines for lung nodules
- Bedside ultrasound that delivers quick results
- Expanded uses for vasodilators
- New insights into sleep disorders
Smartphone App for Asthma Is Shown To Ease Collection of Patients and Data
Digital health research is poised to enter the 21st century, thanks in part to a Mount Sinai study demonstrating that observational clinical trials can be conducted securely and remotely using only a smartphone app.
Published in the April 2017 edition of Nature Biotechnology, the Asthma Mobile Health Study was an innovative collaboration among the Asthma Program at the Mount Sinai – National Jewish Health Respiratory Institute, the Icahn Institute for Genomics and Multiscale Biology, LifeMap Solutions, Apple, and the nonprofit Sage Bionetworks.
Linda Rogers, MD, Associate Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine at Mount Sinai, was a Co-Principal Investigator along with Yu-Feng Chan, MD, Associate Professor of Genetics and Genomic Sciences and of Emergency Medicine, and Eric Schadt, PhD, Dean for Precision Medicine and Jean C. and James W. Crystal Professor of Genetics and Genomic Sciences. Dr. Rogers says the goal was to determine whether it is possible to conduct an observational research study on asthma—including patient recruitment, consent, enrollment, and data collection—all via an app.

“Given that smartphones are the way most people communicate today, we recognize that this technology could be used to obtain real population-based samples with more reach and efficiency,” Dr. Rogers says.
The Asthma Health Application was developed using Apple’s open-source ResearchKit platform and launched through the App Store for iPhone users in March 2015. All told, 7,593 U.S. residents with asthma completed enrollment, providing details about their demographics, comorbidities, and asthma history. Participants then received daily surveys, via text, regarding their symptoms, triggers, and medication adherence, and weekly surveys capturing data on hospital visits and quality of life.
Over the year-and-a-half study—expanded from six months due to its success—participants completed 79,297 daily and 10,969 weekly surveys, with 6,023 participants responding to at least one daily survey and 2,520 participants responding to at least one weekly survey.
The study team was able to observe changes in levels of asthma control in relation to a variety of environmental factors. For example, patients reported more symptoms in regions affected by extreme heat, pollen, and wildfires—a finding that points to the potential for real-time studies of events and their impacts on patients.
Dr. Rogers notes that participants had full control over the data they provided and the features they used, and had the option to allow data from other Apple HealthKit–enabled apps, such as exercise monitoring devices, to populate the app. Furthermore, approximately 88 percent of participants chose to share de-identified data with researchers. The group is currently performing additional analysis on activity data shared by study participants.
“We obtained high-quality, scientifically interpretable data, which we were able to validate using a variety of measures, such as reported peak flows, and could correlate participants’ reported symptoms with separate measurements of local temperatures and pollen levels,” Dr. Rogers says. “Equally important, we were able to do this quickly and efficiently with the use of specially developed digital technology designed to maintain data privacy.”
Ultimately, Dr. Rogers sees opportunities to use the technology to achieve novel insights about asthma epidemiology, reach patients typically underrepresented in traditional studies, and offer them a pragmatic tool to track their asthma symptoms.
“The efficiency of this technology is such that it would no longer take us years to recruit a cross-sectional cohort of this size,” Dr. Rogers says. “We were able to obtain data on thousands of subjects in a matter of months. We also made strides in connecting data from the app with our electronic medical record, which can give us more information to interpret each patient’s asthma control efforts, so this app scratches the surface of what we can potentially do with this technology.”
Multidisciplinary Effort Lowers COPD Readmissions
In light of Medicare statistics confirming that 22 percent of chronic obstructive pulmonary disease (COPD) patients will be rehospitalized within 30 days of an acute exacerbation, Mount Sinai has stepped up its efforts to enhance care quality and significantly reduce hospital visits.
Perhaps the most notable undertaking has been t e launch of the COPD Readmission Quality Initiative in 2015, which created a multidisciplinary pathway of patient assessment, education, and care for every COPD admission. David Steiger, MD, an Associate Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine at Mount Sinai, says the goal is to quickly provide appropriate intervention for each exacerbation, whether by prescribing appropriate medication and conducting respiratory evaluations, or offering help with smoking cessation, nutrition counseling, or education on inhaler use and medicines. An effort is also made to follow up with these patients at Mount Sinai’s outpatient facility within 10 days of discharge.

“We established a system-wide program called Meds to Beds, where a local pharmacy delivers prescribed medications to patients while the patient is hospitalized,” Dr. Steiger says. “By providing education on how to use the medicine and making it more convenient to fill the prescription, we aim to improve patients’ compliance with the medication and keep them out of the hospital.”
At The Mount Sinai Hospital, the COPD treatment pathway also includes referring discharged patients to a rapid-follow-up clinic established by Sidney Braman, MD, a Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine.
“We know the most vulnerable period for readmission is within days of a discharge following an exacerbation,” Dr. Braman says. “I help stabilize patients, ensure they are using their medicine correctly, and identify comorbidities, such as coronary disease, high blood pressure, osteoporosis, and diabetes. I then communicate my findings to the primary care doctors so they can help their patients manage their COPD.”
Through bundled initiatives such as these, Dr. Steiger says, Mount Sinai has reduced its readmission rate across its seven campuses over the last two years. “In 2015, it was as high as 22 percent, but now it is in the range of 15 to 17 percent each month at Mount Sinai Beth Israel, which is well below the national average. Based on these results, we have implemented a similar bundle of services for our acute asthma patients to try to decrease asthma readmissions.”
There is a small but significant subset of COPD patients—including patients who are homeless or are poly-substance abusers—who Dr. Steiger says have not experienced a decrease in readmission rates through the bundled pathway.
“We have launched a new program to target these patients, which includes consultation with a psychiatrist and a follow-up appointment within 48 hours of a discharge at an internal medicine clinic that provides daily availability for seeing recently discharged patients considered at high risk for readmission,” he says. “In addition, we are inviting home services to be involved from the beginning to try to support and address these vulnerable patients’ domestic problems.”
Meanwhile, Dr. Braman is implementing new initiatives at The Mount Sinai Hospital to further decrease COPD exacerbations and readmissions. He has established an Emergency Department observation unit that reduced the hospital admission risk for patients with COPD from 79.1 percent to 67.3 percent within weeks of its launch. Dr. Braman is also looking at deploying outpatient coaches and hiring a COPD coordinator to help patients transition from hospital care to management of the disease at home.
“In taking these measures, we can continue to ensure that our patients maintain their health and improve their quality of life,” he says. “Once you’re on a slippery slope of exacerbations, several things happen. Lung function and quality of life decline rapidly, the risk for readmission is much greater, and the rate of mortality is higher. If we can determine more effective therapies to prevent these exacerbations, we can improve patient outcomes.”
New Guidelines for Lung Nodules
Increased use of CT of the chest has resulted in increases in the rate of nodule identification, which rose from 3.9 per 1,000 person-years in 2006 to 6.6 in 2012. Evaluating these nodules has proven challenging for many pulmonary physicians and radiologists, particularly as new data and research continue to reshape accepted approaches to diagnosis and management.
Now there are new guidelines for assessing incidental lung nodules and molecular biomarkers, which Charles A. Powell, MD, Director of the Mount Sinai – National Jewish Health Respiratory Institute, and Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine), says will enhance the field’s ability to detect, diagnose, and treat lung cancer.
Dr. Powell joined fellow Fleischner Society members in an 18-month initiative to consolidate and revise the Society’s guidelines for monitoring incidental pulmonary nodules detected on CT scans. The Fleischner Society is an international, multidisciplinary medical society for thoracic radiology, dedicated to the diagnosis and treatment of diseases of the chest.
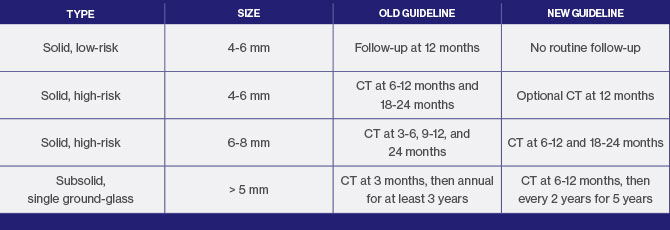
Released in 2017, the new guidelines represent a significant change from recommendations released in 2005 for solid nodules and in 2013 for subsolid nodules, in that they call for a less aggressive approach. The threshold size for ongoing monitoring of low-risk patients with solid nodules has been increased to 6 mm from 4 mm; and for subsolid nodules, monitoring should be less intensive and less frequent, because these tend to be slow-growing cancers. But the most notable change in the revised guidelines is an emphasis on shared decision making with patients for nodule evaluations.
“It has become increasingly evident that it is important to take patient preferences into account in nodule management,” says Dr. Powell, who will assume the role of Fleischner Society President in 2019. “The new guidelines have more latitude so that patients can actively participate with their physicians in decisions regarding frequency for CT scans depending on how concerned they are about their nodules.”
The release of the revised guidelines dovetails with the publication of an American Thoracic Society policy statement on evaluating molecular biomarkers in lung cancer detection to ensure that they meet the standard for clinical use. Developed by an ad hoc steering committee that included Dr. Powell, the statement outlines categories of early-phase research results that should be reported, the evidence necessary to support studying a biomarker’s clinical utility, the outcomes that should be proven, and study designs that can be used to assess outcomes.
“There’s been a continuous effort in recent years to develop, validate, and incorporate biomarkers into clinical practice to improve outcomes in lung diseases,” Dr. Powell says. “As in many other medical fields, biomarkers have been proposed without any certainty that they are clinically useful. This policy statement establishes an expedited process for evaluating and implementing proven biomarkers that will lead to more positive patient outcomes.”
Dr. Powell says that the clinical utility of any biomarker is determined by whether it leads to earlier diagnosis, expedited therapy, fewer aggressive interventions, and fewer lung cancer deaths when compared to the current standard of care. The ultimate goal is to create an online repository of biomarkers that are accepted for clinical use, with full data on how they measure up against the recommendations of the policy statement.
“As medical professionals, we are excited about the prospects of harnessing new knowledge and discoveries that result in earlier diagnosis and better allocation of treatments,” Dr. Powell says. “With this policy statement and the revised nodule guidelines, we have the tools to apply our knowledge in such a way that we continue to improve the balance of benefit to harm in screening and therapy for our patients.”
Bedside Ultrasound Delivers Quick Results
Point-of-care ultrasound (POCUS)—a limited ultrasound performed at the bedside—can aid in risk stratification of patients with acute pulmonary embolism, according to a new Mount Sinai study.
Paru Patrawalla, MD, Assistant Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine at Mount Sinai, conducted the study involving 110 patients with acute pulmonary embolism. They underwent both a goal-directed echocardiogram performed by a Pulmonary and Critical Care Medicine fellow and a formal cardiologist-reviewed echocardiogram to evaluate right ventricular dysfunction, an indicator of increased morbidity and mortality.

“We found that fellows performed and interpreted goal-directed echocardiography with high degrees of sensitivity and specificity—83 percent and 88 percent, respectively, for right ventricular size and 71 percent and 94 percent for right ventricular function,” says Dr. Patrawalla, who collaborated with lead author Jason Filopei, MD, Assistant Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine and an attending physician at Mount Sinai Beth Israel.
“When compared to other indicators of right ventricular dysfunction, such as cardiac biomarkers and CT scan results, goal-directed echocardiography performed by fellows achieved higher diagnostic accuracy.”
Equally important, Dr. Patrawalla notes, was the fact that the goal-directed echocardiogram delivered significantly quicker results than a formal echocardiogram process. This, she says, can make all the difference in assessing the heart of a patient with acute pulmonary embolism, given that patients with this condition and right ventricular dysfunction require early triage to a monitored setting and potentially more aggressive therapies. “We determined that point-of-care ultrasound can provide a quick and accurate picture to improve patient outcomes.”
The study, which was published online in Critical Care Medicine on September 26, 2017, was conducted in light of technological advances that have made ultrasonography more compact and portable, enabling physicians in a wide range of specialties to integrate POCUS into patient diagnosis and management where it matters most: at the bedside.
“POCUS gives us snapshots to answer very focused questions about what is going on with our patients’ illnesses, so we can continually re-evaluate their diagnoses and clinical states, and change our course of management accordingly,” Dr. Patrawalla says.
Several POCUS research and quality-improvement projects are underway in the Pulmonary, Critical Care and Sleep Medicine Division. Through one project, led by Young Im Lee, MD, Assistant Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine, researchers are exploring the effects of mechanical ventilation on diaphragmatic function in patients with respiratory failure. Another initiative is investigating the impact of an ultrasonography protocol performed during cardiac arrest.
Additionally, Dr. Patrawalla is continuing her work in evaluating competency-based training in POCUS, including projects to evaluate a longitudinal curriculum in the Pulmonary and Critical Care Medicine fellowship and collaborations with residents, fellows, and other faculty to develop innovative resident training programs across the Mount Sinai Health System.
Dr. Patrawalla says that most of these studies are intended to evaluate and establish protocols for the use of POCUS in patient diagnosis and management. “This mode of ultrasonography is relatively new, and most clinicians do not have a system yet to apply it,” she says. “My hope is that by establishing these protocols, we can encourage more integration of ultrasound across the system and achieve significant improvements in patient care.”
Expanding Uses for Vasodilators
The development of pulmonary vasodilators has transformed pulmonary arterial hypertension (PAH) from a hopeless diagnosis to a treatable disease. But there has been concern among physicians that these vasodilators would be harmful for the other four pulmonary hypertension disease groups identified by the World Health Organization (WHO). As a result, they are not currently FDA-approved for use in these groups. Yet a recent Mount Sinai patient case suggests that there are subsets of patients in WHO groups 2 and 3 who could benefit from this therapy.
In March 2016, Hooman Poor, MD, an Assistant Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) and Cardiology at the Icahn School of Medicine at Mount Sinai, saw Edwin Mourino, a 61-year-old Rockaway Park, Queens, resident who had been recently hospitalized three times, had experienced several fainting spells, and had an overload of 20 liters of fluid due to severe right-sided heart failure. Although Mr. Mourino would technically have been classified as a WHO group 3 pulmonary hypertension patient, Dr. Poor believed that he was a good candidate for aggressive pulmonary vasodilator therapy.

“Because PAH, or WHO group 1, is a disease that primarily involves the blood vessels, medications that help remodel those vessels are very efficacious,” Dr. Poor says. “But PAH is a relatively rare form of pulmonary hypertension. Most of the patients I see are either in WHO group 2 (pulmonary hypertension due to left heart disease) or 3 (pulmonary hypertension due to lung diseases or hypoxia). We have found that there are instances where the disease can cause increases in blood pressure that result in damage to the blood vessels very similar to what we see in WHO group 1. Thus, we believe there are subsets of patients in these groups who may see long-term benefits from receiving these medications.”
Dr. Poor says the decision to prescribe vasodilators to a WHO group 2 or 3 patient is based mainly on the results of a right-heart catheterization to determine the blood pressure and flow in the lungs and an echocardiogram to assess the degree of strain on the right side of the heart. In Mr. Mourino’s case, his average blood pressure was very elevated, and his cardiac output was very low. An echocardiogram confirmed that the right side of his heart was dilated and dysfunctional, a sign that the blood vessels leading out of that side were narrowed.
Based on these results, Dr. Poor determined that Mr. Mourino was a suitable candidate for pulmonary vasodilator treatment and administered a continuous subcutaneous infusion of Remodulin® (trepostinil), a prostacyclin-class therapy that remodels and dilates the blood vessels of the lungs to increase blood flow. Despite an initial improvement in Mr. Mourino’s exercise tolerance, his pulmonary hypertension progressed to a point where he experienced dizziness and shortness of breath, and he required hospitalization. Dr. Poor switched Mr. Mourino to a continuous IV infusion of Veletri® (epoprostenol), and Mr. Mourino improved to the point where he was discharged.
“I often administer off-label vasodilators for a few weeks to see how the patients do,” Dr. Poor says. “Some patients’ symptoms worsen after a few days, and they are taken off that therapy. Many experience no change in symptoms. But some, like Mr. Mourino, respond and continue on the medication.”
Dr. Poor continues to look for new approaches to treat pulmonary hypertension patients. He expanded his practice and sees patients at both The Mount Sinai Hospital and Mount Sinai Beth Israel, where, together with Ramachandra Reddy, MD, an Associate Professor of Cardiovascular Surgery at the Icahn School of Medicine, he has launched a chronic thromboembolic pulmonary hypertension surgery program for WHO group 4 patients. He is also expanding the clinical trial base for WHO group 2 and 3 patients. One current multicenter study explores the efficacy of inhaled delivery of therapies for WHO group 3 patients, which would avoid complications such as worsened oxygen levels, which can occur with oral or intravenous delivery among WHO group 3 patients.
“WHO groups 2 and 3 are the largest population of pulmonary hypertension patients, and there are no FDA-approved therapies for them,” Dr. Poor says. “If we can identify patients within those groups who will benefit from medications that are already available and proven, we can help improve their quality of life just as we have done with group 1 patients.”
New Insights Into Sleep Disorders
The Mount Sinai Health System continues to make significant advances in studying and understanding sleep disorders to help patients sleep better at night and improve their cardiac, pulmonary, and mental health.
Neomi Shah, MD, Associate Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine at Mount Sinai, is recruiting* patients with obstructive sleep apnea for a study that will assess the effects of continuous positive airway pressure (CPAP) therapy on coronary and carotid plaques using dual-tracer PET/CT imaging, looking at both inflammation and microcalcification.
The study expands on a previous pilot clinical trial in which Dr. Shah determined that patients who adhered to CPAP therapy for at least four hours per night for 70 percent of the study’s duration experienced a significant improvement in atherosclerotic plaques in the carotid arteries. Fludeoxyglucose PET scans of right and left carotid plaque inflammation conducted before and after CPAP use averaging three to six months revealed an average decrease in target-to-background (TBR) mean by 6.1 and 3.8, respectively, and an average decrease in TBR max by 5.4 percent and 5.2 percent, respectively. The five participants in the trial—four of them male—had a median age of 41, had underlying cardiovascular disease risk factors, and were CPAP- and statin-naïve at the time of recruitment.
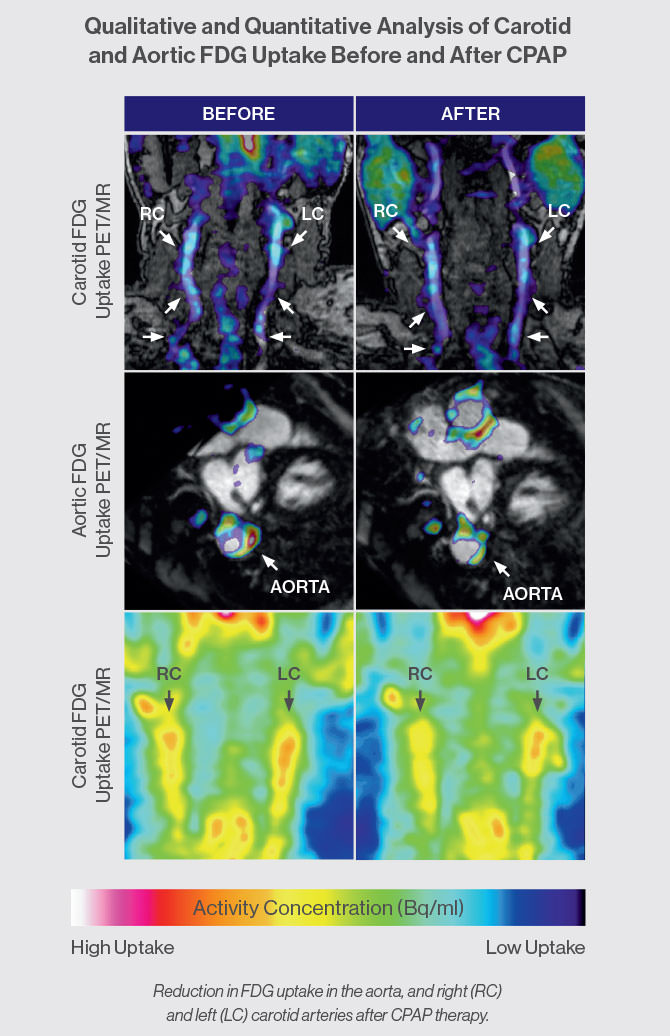
“Although our sample was small, CPAP therapy appears to reduce inflammation to a degree similar to that reported with statins alone,” Dr. Shah says. “Because we can apply state-of-the-art vascular imaging techniques to image and quantify intraplaque inflammation through our collaboration with the Mount Sinai Translational and Molecular Imaging Institute, we are uniquely positioned to understand some of the mechanistic aspects of how CPAP therapy affects arterial vessels and atherosclerosis, and thus determine the future risk of heart attack or stroke.”
At the same time, Andrew Varga, MD, PhD, an Assistant Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine, has been exploring the impact of CPAP patients removing their masks during REM sleep. When 18 CPAP users were tested before and after sleep on a 3D spatial navigation test, Dr. Varga discovered that under usual conditions when their breathing was normal during sleep, next-morning performance improved by 30 percent. However, when sleep apnea was induced during REM in these same users, to mimic the effects of mask removal, the benefit of sleep was completely erased, and subjects were actually 5 percent slower at finding a target in a computer-generated 3D spatial maze the next morning.
“This finding could have serious real-world implications, because many CPAP users consciously or unconsciously remove their masks during the night,” Dr. Varga says. “That leaves their REM sleep untreated and thus results in a deleterious effect on spatial memory.”
Dr. Varga has also found an association between increased apnea severity among older patients and a longitudinal increase in amyloid load in the brain, suggesting a significant increase in the risk of their developing Alzheimer’s disease. Based on these findings, which have been accepted for publication by the American Journal of Respiratory and Critical Care Medicine, he is currently recruiting† patients for another study to explore the impacts of sleep apnea induction during slow-wave sleep on both spatial memory and production of beta amyloid.
“Just because sleep apnea is associated with high levels of beta amyloid at cross section does not mean that it actually causes this accumulation of beta amyloid to happen,” Dr. Varga says. “If inducing apnea leads to an increase of this nature, that would suggest that sleep apnea is really the causal agent in elevating beta amyloid levels.”
As Drs. Shah and Varga advance our understanding of CPAP therapy, Omar Burschtin, MD, an Associate Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) at the Icahn School of Medicine, is helping to innovate home sleep-study technology.
Dr. Burschtin is consulting with the medical technology company Itamar Medical on a next-generation version of WatchPAT— an easy-to-use watch-like portable diagnostic device designed to be worn on the wrist. The upgrade will expand the appliance’s capacity beyond measuring obstructive and central sleep apnea and their relationships to sleep, wake, and sleep stages. In addition to having a traditional pulse oximeter with a Peripheral Arterial Tone (PAT®) signal, the upgraded version, once it has received regulatory approval, will give physicians more channels for gathering a range of physiologic data to enable diagnosis of other sleep and neurological conditions, all without sacrificing ease of use.

“This device has the potential to record various elements based on your clinical suspicions without sending all this complexity to all patients all the time and while keeping patient satisfaction and study completion rates high,” Dr. Burschtin says. “Patients can add electrodes to their legs to confirm movement disorders, to the jaw area to confirm bruxism, or to the forehead for EEG recording to provide insights into the impact of other conditions, including depression and anxiety, and the effects of medications on brain activity.”
Dr. Burschtin says the quality, reliability, and versatility of the new WatchPAT is such that it approximates a limited polysomnography study, enabling physicians and health care providers to use it for clinical-setting assessments.
“For those patients admitted to the hospital with more critical conditions, such as respiratory failure or severe obstructive sleep apnea with serious comorbid medical illness, we can do a bedside test, arrive at an early diagnosis, and obtain an apnea-hypopnea index or degree of sustained hypoxemia to ensure patients are discharged sooner with CPAP or noninvasive ventilation therapy devices,” Dr. Burschtin says. “This device is a great tool for making a diagnosis and also for measuring the efficacy of durable medical equipment like CPAP or noninvasive ventilators at home.”
* Patients can enroll in this study by contacting Dr. Shah’s research coordinator at 212-241-6898.
† Patients with severe sleep apnea who use CPAP regularly can enroll in this study by contacting Dr. Varga’s research coordinator at 212-241-3884.
Message from the Chief: Charles A. Powell, MD
Here in the Division of Pulmonary, Critical Care and Sleep Medicine at Mount Sinai, 2017 was a year of progress in all of our academic and clinical efforts.
 Linda Rogers, MD, and her colleagues conducted a potentially paradigm-changing clinical trial of a smartphone app for asthma, obtaining data on thousands of subjects in a matter of months, and making strides in correlating this data both with electronic medical records and environmental factors, to better understand the subjects’ symptoms, triggers, and medication adherence. The keystone manuscript was published this year inNature Biotechnology.
Linda Rogers, MD, and her colleagues conducted a potentially paradigm-changing clinical trial of a smartphone app for asthma, obtaining data on thousands of subjects in a matter of months, and making strides in correlating this data both with electronic medical records and environmental factors, to better understand the subjects’ symptoms, triggers, and medication adherence. The keystone manuscript was published this year inNature Biotechnology.
Our COPD Readmission Quality Initiative at Mount Sinai Beth Israel, under the direction of David Steiger, MD, has created a multidisciplinary pathway of patient assessment, education, and care for every COPD admission, with the goal of quickly providing appropriate intervention for each exacerbation.
The Mount Sinai Integrative Sleep Center continues to make important strides in understanding the benefits and challenges of continuous positive airway pressure as a treatment for sleep apnea, while in Critical Care, we recently demonstrated that point-of-care ultrasound can aid in risk stratification of patients with acute pulmonary embolism.
Our faculty at the Icahn School of Medicine at Mount Sinai—now celebrating its 50th anniversary—published more than 75 papers in 2016–17 and presented in major symposia, from the American Thoracic Society and American College of Chest Physicians to the Chinese International Society for Respiratory Disease.
In early 2018, the Mount Sinai – National Jewish Health Respiratory Institute will open a new location at Mount Sinai-Union Square, including all of the features that have led to the Institute’s success at The Mount Sinai Hospital. We welcome out-of-town referrals and provide comprehensive multiday visits for the evaluation and treatment of complex and undiagnosed respiratory diseases.
As always, I’d be happy to answer any questions about our work via email at charles.powell@mssm.edu. @CAP_MSNJHRI