
Department of Ophthalmology
We are continuing to build on the commitment of New York Eye and Ear Infirmary of Mount Sinai (NYEE) and the Mount Sinai Health System to set new standards in ophthalmic clinical care, medical education, and clinical research.
Department of Ophthalmology
Our long-term goal is to advance new diagnostic and therapeutic innovations while continuing our longstanding commitment to providing the highest quality, compassionate, and patient-centric care as well as training and educating the next generation of leaders in eye care and research.
This report highlights:
- Breaking new ground in gene transfer therapy to restore vision
- An i-Doctor transforming the field of ophthalmology
- NYEE using a rare retinal transplant to close a patient’s macular hole
- A clinical trial seeking to boost patient satisfaction by making intravitreal injections less painful
- A new pediatric ophthalmology fellowship program that is broadening NYEE’s patient services
- Faculty news: Promotions
- Faculty news: Appointees
- Message from the System Chair of the Department of Ophthalmology
- Inside Feature: Imaging is a pivotal part of NYEE's rich history
Breaking New Ground in Gene Transfer Therapy to Restore Vision

Fueled by a $2 million grant from the McGraw Family Foundation, the recent arrival of Bo Chen, PhD, and his research lab is giving the Department of Ophthalmology at New York Eye and Ear Infirmary of Mount Sinai (NYEE) a powerful platform to explore the exciting field of retinal regeneration to potentially restore visual function in people with degenerative glaucoma, macular degeneration, and retinitis pigmentosa. The grant is enabling the new lab to expand its work through the purchase of a Beckman ultracentrifuge to make AAV viruses for retinal gene transfer, and a Zeiss confocal microscope to evaluate that process as well as the effects of therapeutic intervention on retinal tissue.
Dr. Chen, Associate Professor of Ophthalmology and Director of the Ocular Stem Cell Program at the Icahn School of Medicine at Mount Sinai, has made considerable progress over the past eight years in highlighting the role of gene therapy. In his previous lab at the Yale School of Medicine, Dr. Chen worked with mouse models to better understand the molecular and cellular pathways in major retinal degenerative diseases. In one major project—for which work continues at the Mount Sinai/NYEE Eye and Vision Research Institute—he has demonstrated how the gene transfer of ß-catenin and three transcription factors can successfully reprogram retinal glial cells into rod photoreceptors.
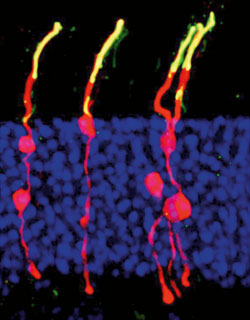
Photoreceptors are light-sensitive cells in the retina in the back of the eye that signal the brain when activated. In mammals, including mice and humans, photoreceptors do not have the ability to regenerate on their own when retina cells are destroyed by diseases like macular degeneration or retinitis pigmentosa, causing irreversible blindness. However, the Müller glial (MG) cells of the zebrafish have regenerative potential whereby they divide and turn into photoreceptors and other retinal neurons, establishing a powerful self-repair mechanism.

To see if MG cells could be programmed to become rod photoreceptors in a living mouse, Dr. Chen induced the cells to divide by injecting ß-catenin into the eyes of mice as a first step and, weeks later, injecting three transcription factors—Otx2, Crx and Nrl. This regenerative process, he found, not only encouraged the newly divided cells to develop into rod photoreceptors, but structurally made them look no different than the real photoreceptors. Would the treatment actually work, though, in restoring vision to mice with congenital blindness? The exciting finding by Dr. Chen was that light responses recorded from retinal ganglion cells—neurons that carry signals from photoreceptors to the brain—and measurements of brain activity showed that the newly formed rods were in fact integrating into the visual pathway circuitry. “Mice who were blind from birth were now able to see light for the first time in their lives following treatment,” he observes.
Dr. Chen’s groundbreaking study, published August 15, 2018, in the online issue of Nature, has received national attention for opening a new pathway for potentially treating in humans retinal degenerative diseases, which currently have no cure, by manipulating our own regenerative capability to self-repair.
“We’ve been able to show through a gene transfer technique using AAV viruses that we could activate resident stem cells—the so-called Müller glial cells—to re-enter the cell cycle to generate a population of stem cells that differentiate into retinal neurons,” Dr. Chen explains. The scientist is now working with Mount Sinai’s patent office to file a provisional patent based on these findings.
As a McGraw Family Vision Researcher, Dr. Chen says he relishes the ability to tap into a rich array of resources at Mount Sinai and NYEE to advance his groundbreaking work. They include a cutting-edge imaging capability under the leadership of Richard Rosen, MD, Vice Chair and Director of Ophthalmic Research at NYEE; the nationally recognized Icahn Institute for Genomics and Multiscale Biology; and NYEE’s considerable pool of patients.
“Dr. Tsai [President of NYEE] has given me a broad mandate to pursue my work in retinal regeneration and neural protection,” emphasizes Dr. Chen, winner of the Pew Scholars in the Biomedical Sciences Award, given to young investigators showing outstanding promise. “These fields represent unlimited opportunities today for labs like mine, and we believe our findings underscore that we are closer than ever to major new therapies for people with severe degenerative eye disease.”

This i-Doctor Is Transforming the Field of Ophthalmology

When a New York Eye and Ear Infirmary of Mount Sinai (NYEE) eye surgeon performed a complex anterior segment surgery this June on a patient with cataracts and glaucoma, he relied on three innovative and novel technologies. One was miLOOP™, the first micro-interventional cataract fragmentation device for non-thermal cataract surgery. The second was intraoperative aberrometry, which has uprooted the 50-year-old standard for intraocular lens power determination, and the third was CyPass®, the first non-trabecular micro-stent for glaucoma.
Once the procedure was completed, the surgeon, Tsontcho Ianchulev, MD, could take pride in more than its successful surgical outcome. He had invented and pioneered all three U.S. Food and Drug Administration-approved devices, now becoming standard of care and widely used by ophthalmologists at NYEE. Dr. Ianchulev is a Professor of Ophthalmology at the Icahn School of Medicine at Mount Sinai, but as an entrepreneur, he has reached well beyond the academic bench: Through the years, his technologies have raised close to $200 million in venture financing, and have created more than $1 billion in investor value.
As a physician, inventor, and entrepreneur, Dr. Ianchulev has always had his eye on the big picture—how paradigm-shifting innovations can improve the lives of not just individual patients, but entire populations around the globe. Indeed, he became acutely aware during his early years of medical training at Harvard University of “how much we can accomplish as clinicians if we leave our comfort zone and start to innovate and come up with solutions using our scientific and technocratic minds.”
There is no better window on the “disruptive” mindset of Dr. Ianchulev than miLOOP. Instead of settling for incremental change to the traditional way of doing cataract surgery, he set his sights on a revolutionary new process: an easy-to-use, hand-held, pen-like device that could bridge the yawning gap between developed and developing health care systems and help all to fight the blindness epidemic from cataracts that affects 25 million people worldwide.
“Phacoemulsification has been extremely successful, but the fact is, we’re still doing cataract surgery pretty much the way we did it 50 years ago,” he explains. “It also introduces a lot of undesirable energy and leads to corneal damage and endothelial cell loss, among other things. Also, it has not made a dent outside the developed world because its complexity and cost are incompatible with the developing world.”
Dr. Ianchulev’s path to changing the standard was to borrow from cardiovascular surgery and interventional radiology. He recognized that some of the micro-invasive techniques that have revolutionized those fields in recent years are quite transferable to ophthalmology, particularly cataract surgery, where they have the ability to transform how the procedure is done and potentially reduce blindness around the world. So, he and his engineering team at the company he cofounded, Iantech, developed miLOOP, which allows for energy-free fragmentation of even the hardest cataracts without rupturing the 4-micron-thin capsule enveloping the lens.
Intraoperative aberrometry also started off as a problem in search of a solution. While still a resident, Dr. Ianchulev realized “how imprecise and outdated our existing technology was in figuring out the correct IOL power in cataract surgery.” The flashing light inside his head made him realize that “if you take out the lens before you put in the IOL, you have this privileged state of the eye, this pure optical system from the cornea all the way to the retina. It’s almost like an optical biopsy of the eye that lets you figure out the total optical deficit.” From that insight evolved the first intraoperative biometry in ophthalmology, now known as Optiwave Refractive Analysis (ORA) from Wave Tec.
Today, Dr. Ianchulev is re-inventing one of the oldest ophthalmic devices: the eyedropper. By shrinking inkjet printer technology to a device the size of a smartphone, he can deliver medication to the eye in a precise and targeted way—something that conventional pipette eyedropper technology has not been able to do, resulting in wasting or overdosing the medication.
Dr. Ianchulev is now putting his boundless creative energy and expertise to everyday work at NYEE as Director of the Ophthalmic Innovation and Technology Program. Specifically, he is building a whole ecosystem around innovation—exciting new therapies, technologies, and diagnostic modalities—that can be developed, commercialized, and pushed out to people suffering from eye disease in every corner of the world. To that end, the Ophthalmic Innovation and Technology Program draws vital support from faculty, clinicians, and residents at both NYEE and The Mount Sinai Hospital, as well as from a growing array of academic partners on the outside. And through its collaboration with Mount Sinai Innovation Partners, the program is forging new commercial relationships that cover everything from licensing intellectual property to participating in new entities that can capitalize on our homegrown technology.
“Through our Ophthalmic Innovation and Technology Program, we’re creating an environment where the best and brightest ideas are not just taken out for a spin, but actually brought to life,” Dr. Ianchulev declares. “And more importantly, we’re training our residents and students not only to be excellent clinicians, but also how to staff the front lines of innovation. Our patients need it just as much as their next doctor visit.”

NYEE Uses a Rare Retinal Transplant To Close a Patient’s Macular Hole
When 68-year-old Mary Benjamin came to Ronald Gentile, MD, at New York Eye and Ear Infirmary of Mount Sinai (NYEE), she had a macular hole that three surgeries elsewhere had failed to close. The retinal surgeon quickly recognized the severity of the case. For one thing, Ms. Benjamin’s sight had deteriorated to the point where merely walking down the block became hazardous. “After the macular hole reopened, I could barely count the number of fingers in front of my left eye,” Ms. Benjamin recalls. “And the drops I was taking just weren’t doing the job they were meant to.”
On clinical examination, OCT imaging revealed that the macular hole registered nearly 800 microns, making standard closure using the internal limiting membrane that was previously removed impractical, by Dr. Gentile’s reckoning. Instead, he reached for a surgical technique that had only been performed a handful of times in this country: autologous retinal transplantation. The transplant is used to close the hole and reattach the normal retina around the hole that was detached because of it. “It’s only applicable to the very small number of patients who have failed multiple procedures in attempting to close a very difficult macular hole,” explains Dr. Gentile, Clinical Professor of Ophthalmology at NYEE. “In Mary’s case, we realized it was our best chance to give her something approaching a permanent fix.”
The surgery, which took place in March 2017, began by harvesting an autologous neurosensory free flap from the peripheral retina to place over the macular hole and provide a type of plug for closure. This flap, approximately 0.5 disc diameter larger than the macular opening, was cut using forceps and scissors. Dr. Gentile likens this part of the hour-long procedure to taking a biopsy of an area of the eye that is disease-free, yet not being used for vision.
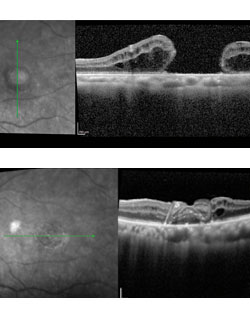
A light laser around the edges of the area marked for grafting, along with diathermy to blood vessels at the edge, was then used by the surgeon to prevent future complications, including retinal detachment. Once the transplanted retinal tissue was placed into and stretched over the macular hole, the eye was filled with perfluorocarbon (PFC) and silicon oil exchange to keep the transplanted tissue firmly in place. Once the transplanted retina is in place, it adheres to the surrounding tissue through what is believed to be migration of glial cells across the patch, which acts like stitching. While Dr. Gentile and some of his colleagues do not believe that the transplanted piece of retina itself is detecting light, the reattachment improves the function of the adjacent neurosensory retina by promoting its reattachment to the retinal pigment epithelium (RPE) to allow the patient to see better.
“The key to the surgery is really getting the transplant into the hole, and ensuring it doesn’t move by applying the right pressure during the PFC silicon oil exchange,” says Dr. Gentile, who has performed two autologous retinal transplants—among the very first cases in New York City. “This involves precise, leading-edge surgery that’s intended only for patients who have exhausted all other forms of treatment.”
For Ms. Benjamin, a retired home health aide who has five grown children and 12 grandchildren, the results were evident within days of the surgery. Her visual acuity improved to 20/200 in her left eye, enough to allow her to resume everyday activities that had previously been off limits. “The thing I notice most is that my vision is now balanced the way it should be between both eyes,” she says, “which allows me to focus on and see things I couldn’t before my repair.”
Ms. Benjamin is hardly out of the woods, though. She has had other complications, including cataracts in both eyes and removal of the silicone oil with gas injection, which required further surgery by Dr. Gentile nine months after the autologous retinal transplant. While Dr. Gentile was candid in explaining to Ms. Benjamin the uncertainties of a procedure still in its embryonic stage, she says she was eager to proceed knowing what the future benefits might be. “There’s certainly some risk to trying anything new,” acknowledges Dr. Gentile, “but we now have an important new procedure in our toolbox, and won’t be afraid to roll it out for patients who previously had no good options.”
A Clinical Trial Seeks to Boost Patient Satisfaction by Making Intravitreal Injections Less Painful

Joe Mauro dreaded his injections every month or two at a local ophthalmologist’s office for diabetic retinopathy. “It pricked a little bit when I got the needle, but the big problem was afterwards, when it felt like I had a hot coal in my eye,” recalls the 68-year-old Staten Island resident, who has type 2 diabetes. “And I have a pretty good threshold for pain.”
Intravitreal injections have revolutionized the treatment of retinal disease, from macular degeneration to diabetic retinopathy to other disease forms involving leaky blood vessels or the growth of new ones. What has received far less attention from the medical community, though, is the issue of patient satisfaction with this breakthrough therapy—particularly as to how they react to the very thought and experience of receiving repeat injections in the eye. Indeed, pain can persist for a few days following treatment, and can disrupt and/or even diminish patient compliance.
New York Eye and Ear Infirmary of Mount Sinai (NYEE) has become one of the first institutions to address this matter with a recent study focused on improving the patient experience and reducing pain from intravitreal injections. “When we question patients, they tell us their greatest fear, besides going blind, is pain from the injection,” says Ronald Gentile, MD, Clinical Professor of Ophthalmology at NYEE, and the study’s principal investigator. “So our goal became to put a procedure in place that could actually improve their experience and eliminate any discomfort from pain.”
The clinical trial, which began collecting data in June 2017, enrolled 60 patients who received intravitreal injections of bevacizumab, aflibercept, or triamcinolone acetonide. They were then randomized to receive post-injection one of three treatments: a single drop of topical nepafenac 0.3 percent suspension, an NSAID; a pressure patch for two hours; or a single drop of preservative-free tears (the control group). The results were extremely encouraging for investigators. They showed that the group receiving topical nepafenac had significantly lower pain scores than those in the control group at 6 hours and 24 hours. While the group who received a pressure patch after the injection also showed lower pain scores, the data was not found to be statistically different from those in the control group.
“What we found was an easy way to reduce the patient’s pain after injection with literally just a single drop,” explains Richard Kaplan, MD, a vitreoretinal surgery fellow at NYEE, who worked on the study with Dr. Gentile. “Our hope is that this procedure will not only make patients happier with their treatment, but result in better compliance and thus better outcomes.”
The pain management trial turned Mr. Mauro into a believer—and an NYEE patient. “It was amazing how, after getting a drop in the eye, I had no discomfort at all after the needle,” reports the retired New York City police officer. “No one wants to get a needle in the eye, but now I don’t really have a big problem with it.”
While not a standard procedure, NSAIDs like nepafenac are often used by ophthalmologists to relieve discomfort following a wide range of procedures. The favorable findings of this patient satisfaction study make it more likely than ever that these agents will enjoy an increasingly prominent role at NYEE and other ophthalmic centers.
New Pediatric Ophthalmology Fellowship Program Is Broadening NYEE’s Patient Services
The growing integration of New York Eye and Ear Infirmary of Mount Sinai (NYEE) and Mount Sinai Health System is about to yield another dividend: a joint pediatric ophthalmology fellowship program accredited by the Association of University Professors of Ophthalmology Fellowship Compliance Committee (AUPO FCC). The AUPO FCC-compliant year-long program, which will start in July 2019, will add to both institutions’ nationally recognized roster of specialized training in the fields of cornea and refractive surgery, glaucoma, vitreoretinal surgery and uveitis.
“The Mount Sinai Hospital (MSH) and NYEE both have long histories of excellence in education, and the new pediatric fellowship will really complete our fellowship training programs,” says Douglas Fredrick, MD, newly named Deputy Chair for Education in the Department of Ophthalmology and Chief of Pediatric Ophthalmology for the Mount Sinai Health System. Because fellows take an active role in training residents, the new program will enable NYEE and Mount Sinai to jointly grow their pediatric ophthalmology and strabismus service lines by increasing the number of providers, adds Dr. Fredrick, who will lead that expansion along with creation of a comprehensive pediatric ophthalmology center.
While the new pediatric fellowship will start modestly with a single fellow, Dr. Fredrick envisions the program soon taking under its wings three fellows—two domestic and one international—over the next five years. “International fellows will return to their home countries and help improve the quality of care for both developing and developed nations,” he explains. “It’s also my hope that we’ll be able to establish closer relationships with departments of ophthalmology and training programs in those developing countries.”
Closer to home, the AUPO FCC-approved fellowship program will address a national shortage of pediatric ophthalmologists. “Our goal is to help fill that gap by training highly qualified pediatric ophthalmologists to handle everything from basic to very complex eye disorders in children, as well as strabismus in adults,” says Tamiesha Frempong, MD, who will serve as Director for the new joint fellowship program when it launches next summer.
While MSH does not currently train pediatric ophthalmology fellows, NYEE has a long tradition of excellence in orthoptic, strabismus, and pediatric ophthalmology education. With over 50 years of leadership of the Orthoptics service, Sara Shippman, CO, along with Co-Directors of the Pediatric Ophthalmology and Strabismus division, Steven Rosenberg, MD, and Erin Walsh, MD, work as a team to supervise residents, sub-specialty fellows and attendings to provide pediatric-centered integrated care to children with the broadest spectrum of ocular and vision-threatening conditions.
Under the new fellowship program, the network of training sites will grow beyond NYEE and MSH to include Elmhurst Hospital in New York City’s borough of Queens, giving fellows the chance to treat a spectrum of disorders akin to what might be found in developing countries, according to Dr. Frempong. “These fellows will be part of a multidisciplinary approach to treating patients who could have rare, complicated syndromes and complex eye diseases like orbital tumors,” she notes.
What will set the new pediatric ophthalmology fellowship program apart from others across the country is its unique combination of eye hospital and major academic medical center, stresses Erin Walsh, MD. “Fellowship programs typically have access to one or the other, but rarely do they have both of these resources at their fingertips,” she elaborates. “For fellows, this will translate into exposure to a wide range of pathology, as well as to attendings who can share a wealth of knowledge and experience.”
Faculty News: Promotions

Paul A. Sidoti, MD, Site Chair of Ophthalmology at NYEE
A faculty leader at NYEE for more than 24 years, Dr. Sidoti previously served as Deputy Chair for Clinical Affairs at Mount Sinai Health System and has been instrumental in the growth of NYEE’s national and international reputation as one of the top teaching hospitals for ophthalmology. He received his medical degree from the Albert Einstein College of Medicine. He completed his residency at NYEE and a clinic al/research fellowship in glaucoma at the Doheny Eye Institute/University of Southern California School of Medicine.

Richard B. Rosen, MD, Deputy Chair for Clinical Affairs at Mount Sinai Health System
An internationally renowned researcher and lecturer, Dr. Rosen joined the Department of Ophthalmology at NYEE in 1989. Under his leadership, NYEE established one of the most advanced imaging centers in the country. As Deputy Chair for Clinical Affairs, Dr. Rosen will apply his problem-solving skills to continue to improve clinical operations and patient care delivery. He will continue to serve as Director of Ophthalmic Research and Director of the Retina Service/Retina Fellowship at NYEE. Dr. Rosen earned his MD from the University of Miami School of Medicine, and completed his ophthalmology residency and vitreoretinal fellowship at NYEE.

Robert C. Della Rocca, MD, Chief of Oculoplastic and Reconstructive Surgery at Mount Sinai Health System
Dr. Della Rocca is an international leader in complex cosmetic and reconstructive eyelid, midfacial, and orbital surgery. A distinguished lecturer, editor, and author of five textbooks, Dr. Della Rocca is also a dedicated teacher; over the years, he has mentored 35 fellows in U.S. and over 85 internationally. A resident graduate of NYEE, Dr. Della Rocca was instrumental in growing the Oculoplastic and Reconstructive Surgery division for the past 35 years, ultimately serving as its Director. Dr. Della Rocca is also passionate about mission work, traveling to 15 countries, in the past three decades, as a volunteer surgeon to care for patients from the remote villages.

David A. Della Rocca, MD, Director of Oculoplastic and Reconstructive Surgery at The Mount Sinai Hospital and Mount Sinai St. Luke’s
A specialist in reconstructive orbital and eyelid surgery with a busy clinical practice, Dr. Della Rocca is committed to being a teacher of residents at NYEE, The Mount Sinai Hospital, and Mount Sinai St. Luke’s. An author of many peer-reviewed articles and chapters, Dr. Della Rocca is a recipient of the 2012 Achievement Award for teaching from the American Academy of Ophthalmology. After earning his medical degree from Creighton University School of Medicine, Dr. Della Rocca completed his ophthalmology residency and fellowship in oculoplastic, reconstructive, and orbital surgery at NYEE.

Harsha S. Reddy, MD, Director of Oculoplastic and Reconstructive Surgery at NYEE and Physician Wellness Champion for Ophthalmology GME at Mount Sinai Health System
Dr. Reddy is an ophthalmic plastic surgeon specializing in correction of age-related changes in the eyelids and reconstruction after facial trauma and tumors. With a passion for teaching, research, and service, Dr. Reddy has written many peer reviewed articles, lectured at international scientific conferences, and led medical outreach in Nepal, Ethiopia, and India. He has received many teaching awards at NYEE and was a finalist for the 2014 Henry Bayliss Outstanding Thesis Award (ASOPRS). Dr. Reddy earned his medical degree at Harvard Medical School, completed an ophthalmology residency at the Doheny Eye Institute and an ASOPRS oculoplastic surgery fellowship at the University of Washington.
Faculty News: Appointees

Louis R. Pasquale, MD, FARVO
An internationally renowned clinician and researcher into primary prevention strategies in open-angle glaucomas, Dr. Pasquale joins the Department as Site Chair of Ophthalmology at The Mount Sinai Hospital and Mount Sinai Queens, as well as System Vice Chair for Translational Ophthalmology Research. In these combined roles, he will seek to achieve excellence in clinical care delivery using the latest technologies for data acquisition and analysis, as well as integrate the department’s translational research efforts into the larger global research network. Dr. Pasquale previously served as Professor of Ophthalmology at Harvard Medical School and Director of the Glaucoma Service at Massachusetts Eye and Ear.

Douglas Fredrick, MD
An academic leader with an international reputation in the treatment of complex strabismus, pediatric cataracts, cortical visual impairment, and modalities to slow the progression of childhood myopia, Dr. Fredrick joins the Department as Deputy Chair for Education and System Chief of Pediatric Ophthalmology. He will work to build upon the Department’s national and international reputation as a premier teaching institution for residents and fellows, in addition to leading the expansion of pediatric ophthalmology services, including the establishment of a comprehensive pediatric ophthalmology center. He previously served as Clinical Professor and Vice Chair of Education and Clinical Affairs in Ophthalmology at Stanford University.

Valerie I. Elmalem, MD
Fellowship trained in both neuro-ophthalmology and in orbital and ophthalmic plastic surgery, Dr. Elmalem joins the Department as Director of Neuro-Ophthalmology at Mount Sinai West. With a special interest in surgical education of ophthalmology residents, Dr. Elmalem’s clinical focus includes diagnosis and management of optic nerve and brain disorders affecting vision, and management of a wide range of eyelid and orbital conditions in adults and children. Recognized by her peers as an outstanding ophthalmologist, Dr. Elmalem has been listed in Super Doctors® New York Rising Stars since 2015. She previously served as Director of Neuro-Ophthalmology at SUNY Downstate Medical Center.

Gareth Lema, MD, PhD
Gareth Lema, MD, PhD joins the Department as Director of Quality, Safety, and Experience at The Mount Sinai Hospital. A fellowship-trained retina specialist with a particular interest in diseases of the retina and posterior uveitis, including diabetic retinopathy, macular degeneration, and ocular trauma, Dr. Lema also holds a PhD in biophysics. He is leaving his positions as a Clinical Assistant Professor of Ophthalmology at the University at Buffalo Jacobs School of Medicine and Director of the Retina Division of the Ira G. Ross Eye Institute.
Message from the System Chair of the Department of Ophthalmology: James C. Tsai, MD, MBA

The New York City metropolitan region deserves to have one of the world’s leading centers for ophthalmology and vision research. I am delighted to highlight the many steps that New York Eye and Ear Infirmary of Mount Sinai (NYEE) has taken over the past year to move ever closer to that goal. Rather than rest on our laurels as the oldest specialty hospital in America, founded in 1820, we are focused on extending our longstanding reputation for clinical excellence to that of innovation and development of exciting new treatments and approaches for complex blinding eye diseases.
Recruiting top scientific talent is an integral part of our overall strategy, as evidenced by our recent appointment of Louis Pasquale, MD, FARVO, as Vice Chair of Translational Ophthalmology Research for the Mount Sinai Health System and Site Chair of Ophthalmology at The Mount Sinai Hospital. Dr. Pasquale, who has gained international recognition for his research, will focus on integrating NYEE into the cutting-edge, interdisciplinary clinical and translational research network of the Icahn School of Medicine at Mount Sinai. Our commitment to education complements our research initiatives, which is one of the reasons we have recruited Douglas Fredrick, MD, as Deputy Chair for Education in the Department of Ophthalmology and System Chief of Pediatric Ophthalmology. Among Dr. Fredrick’s priorities will be consolidating and combining the already outstanding ophthalmology residency programs at NYEE and The Mount Sinai Hospital into one of the largest residency programs in the nation. In addition, our pursuit of clinical excellence is highlighted by the appointment of Paul Sidoti, MD, as Site Chair of Ophthalmology at NYEE.
The many accomplishments of our researchers and clinicians this past year underscore the breadth of NYEE’s capabilities—and their enormous potential. For example, a team of scientists led by Bo Chen, PhD, who heads up our Ocular Stem Cell Program, restored vision in congenitally blind mice by activating and reprogramming retinal stem cells. This breakthrough process, described in the August 15, 2018 issue of Nature, could unveil a promising and long-awaited new pathway for treating degenerative retinal disease in humans. Our scientists also developed a revolutionary micro-interventional device, miLOOP™, which has the potential to transform cataract surgery and dramatically reduce blindness around the world. Helping us push the boundaries of discovery in the visual sciences is the recently created Mount Sinai/NYEE Eye and Vision Research Institute and the Ophthalmic Innovation and Technology Program. Together, these programs are helping to ensure we work synergistically with such well-established academic departments and research centers at Mount Sinai as The Friedman Brain Institute, the Icahn Institute for Genomics and Multiscale Biology, and The Black Family Stem Cell Institute. Our research and clinical efforts rest on another powerful pillar with a rich history at NYEE: imaging. We are home to one of the leading imaging laboratories in the world under the direction of Richard Rosen, MD, Vice Chair and Director of Ophthalmic Research at NYEE. As the articles in this Specialty Report illustrate, this extraordinary imaging team, working with the most sophisticated equipment anywhere, is allowing us to explore and understand diseases like glaucoma and macular degeneration at a level we never could before.
As we approach our bicentennial in 2020, it’s important to note that 2018 marks the centennial of The American Journal of Ophthalmology (AJO). A recent article in that journal1 describes how the histories of AJO and NYEE have been inextricably bound. Indeed, in its venerable role as recorder of institutional histories, AJO has consistently documented how a coterie of early ophthalmic hospitals such as NYEE has moved the field of ophthalmology forward. This is a legacy embraced today by our clinicians, scientists, educators, trainees, and staff, all of whom are dedicated to improving and advancing patient care through innovation and collaboration.
1Pathipati AS, Tsai JC. The Influence of Early Ophthalmic Hospitals on the Journal. Am J Ophthalmol. 2018 Aug; 192:xxiii-xxvii.
Imaging Is a Pivotal Part of NYEE’s Rich History

Groundbreaking work by the Advanced Retinal Imaging Laboratory at New York Eye and Ear Infirmary of Mount Sinai (NYEE) into understanding the microstructure of the human retina rests on a rich historical foundation.
From the very first photographs of a retina in a living organism (a rabbit), taken by Henry Noyes, MD, Executive Director of NYEE in the 1860s, to current efforts to unravel the molecular mystery of retinal disease through high-resolution imaging such as adaptive optics and swept source optical coherence tomography (OCT), New York Eye and Ear Infirmary has been in the forefront.
What Do Retinal Imaging and the Hubble Telescope Share? The Unique Technology of Adaptive Optics
Technology called adaptive optics has allowed mighty telescopes like NASA’s Hubble to reach deep into time and space and send back astonishing images. Now, adaptive optics has been turned inward to allow researchers to investigate the human eye and diseases like glaucoma, diabetic retinopathy, and retinal vein occlusion. “We’ve reversed the direction. Instead of looking at outer space, we’re using adaptive optics to peer into the retina,” explains Yuen Ping Toco Chui, PhD, Assistant Professor of Ophthalmology at the Icahn School of Medicine at Mount Sinai, who is helping this effort through software enhancements that she has developed in the David E. Marrus Adaptive Optics Imaging Laboratory at The Shelley and Steven Einhorn Clinical Research Center of New York Eye and Ear Infirmary of Mount Sinai (NYEE).
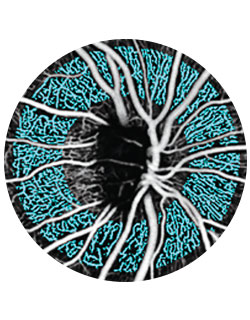
NYEE is one of a handful of sites in the country with adaptive optics scanning laser ophthalmoscopy (AOSLO). This sophisticated imaging technique—part of a translational research collaboration with Medical College of Wisconsin and Stanford University—is allowing researchers and clinicians an unprecedented view of the retinal microvascular structure and blood flow.
Adaptive optics, which produces images that are high resolution and noninvasive, is particularly well suited for retinal imaging. The technology was built into astronomical telescopes by U.S. government scientists nearly 30 years ago to improve image resolution by compensating for distortions caused by atmospheric turbulence. In the case of ocular applications, adaptive optics helps achieve cellular-level imaging by overcoming optical aberrations.
This is accomplished by measuring the distortions of the wavefront of the light reflected from the eye and compensating for them by using a special mirror that corrects or “adapts” to those distortions, essentially eliminating them. Adaptive optics improves the optical transmission of light to allow for resolutions of around 2 microns, 10 times the resolution of optical coherence tomography (OCT) imaging and sufficient to detect and count the individual rods in the photoreceptor mosaic, which are approximately 2.5 microns in diameter.
Dr. Chui, for her part, has made adaptive optics an even more powerful tool by developing software that enables users to quantify the high-resolution images they receive. What motivated her efforts, she explains, was the fact that AO, for all its strengths, does not have a user-friendly interface to help clinicians interpret the reams of data they receive from its images. “The software we’ve developed provides an algorithmic analysis of the image data to help clinicians determine if a patient is likely to have early diabetic retinopathy, say, or glaucoma.” In a study reported in Investigative Ophthalmology & Visual Science in June 2015, Dr. Chui and other NYEE researchers found that microscopic, noninvasive imaging of the fine retinal vasculature using split-detection AOSLO allowed for quantification of lumen and wall thickness. Significantly, they learned that this technique has potential for early detection of diabetic retinopathy, as well as accurate monitoring of progression and treatment response.
Dr. Chui’s work with adaptive optics has produced improvements to another mainstay of NYEE’s advanced imaging toolkit: optical coherence tomography angiography (OCTA). Like AOSLO, the basic OCTA machine lacks the quantitative and qualitative software to enable physicians to know if the images they’re viewing substantially deviate from the norm and, if so, by how much. Dr. Chui addressed that deficiency with software enhancements that allow for precise mapping of the progression of diabetic retinopathy, glaucoma, and other vascular-related diseases.
“We have preliminary data showing our software is able to diagnose preclinical diabetic retinopathy, which is a huge development,” says Dr. Chui. “For the patient, this means avoiding injection of fluorescein dye, using noninvasive imaging that provides a 3-D view of the retinal capillaries.”
OCTA software enhancements from Dr. Chui and her team are also helping to transform the early detection of primary open-angle glaucoma and normal-tension glaucoma. To that end, they are helping physicians identify changes in the retinal nerve fiber layer and its blood supply, the radial peripapillary capillary network, much earlier using motion contrast analysis for red blood cell movement.
“We intend to make the software we’ve developed at NYEE a new addition to our clinical care,” emphasizes Dr. Chui. “And that will help us to better monitor the progress of eye disease, and allow clinicians to intercede at the earliest possible stage to benefit the patient.”
An Unprecedented Look at Acute Solar Retinopathy
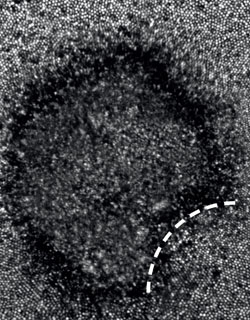
Clinicians at New York Eye and Ear Infirmary of Mount Sinai (NYEE) were on high alert when a 26-year-old woman walked through their doors just days after viewing the solar eclipse of August 2017, complaining of blurred vision and color distortion. Their suspicions were soon confirmed: a case of acute solar retinopathy and photochemical burns, reinforced by images that were unlike any they had ever seen before. Through the use of adaptive optics scanning laser ophthalmoscopy (AOSLO), they were rewarded with a high-resolution, cellular-level view of damage to the patient’s photoreceptor layer.
“We were able to literally see the crescent-shaped spatial damage to a very specific part of her retina,” recalls Avnish Deobhakta, MD, Assistant Professor of Ophthalmology at the Icahn School of Medicine at Mount Sinai and a member of the NYEE clinical team that examined the patient. “We had never been able to see that level of precision before, because imaging modalities like adaptive optics weren’t even available until recently to examine solar retinopathy. We’re hopeful this kind of information will not only help us to better understand the condition, but lead to new therapies for solar retinopathy and other forms of photic injury to the retina.”
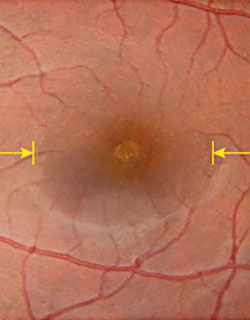
Solar retinopathy is a rare form of retinal injury that results from directly gazing into the sun. In the case of the NYEE patient, damage occurred when the young woman stared at the sun during last year’s eclipse for 21 seconds without proper protective eyewear. Four hours later, she noticed blurred vision, color distortion, and metamorphopsia, which were worse in the left eye, her dominant eye. She also reported seeing a black spot in the center of her left eye.
Three days later, retinal scanning began at NYEE’s David E. Marrus Adaptive Optics Imaging Laboratory, using a combination of optical coherence tomography angiography (OCTA), en face OCT, and AOSLO. While OCTA was normal in both eyes and en face OCT showed reflectivity changes, adaptive optics scanning was able to produce high-definition images of foveal cone photoreceptor mosaic disturbance.
“New York Eye and Ear Infirmary is one of a few imaging centers in the country able to marry AO with other advanced technologies like OCT angiography and en face OCT,” Dr. Deobhakta says. “That unique capability opens the door to further research and investigations into diseases we weren’t able to characterize before.”
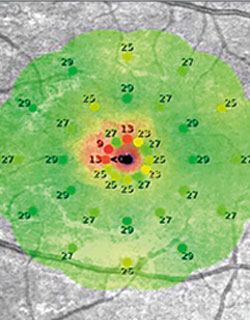
Nowhere is that more evident than with solar retinopathy. While past studies conducted after solar eclipses have traced the mechanism of injury to photochemical toxicity, and localized it to the foveal photoreceptor outer segments and the retinal pigment epithelium, researchers at NYEE were able to drill down even deeper this time, as described in the December 7, 2017, online edition of JAMA Ophthalmology.
Specifically, they used a custom-made AOSLO device with both confocal and split-detector optics to image the cone photoreceptor mosaic at the fovea. What they discovered in the patient’s right eye was a small region of hyporeflectivity, indicating the presence of non-wavelength guiding photoreceptors, and disruption of the photoreceptor structure with a few enlarged inner segments. In the left eye, the advanced imaging showed a large area with ambiguous reflectivity due to abnormal and non-wavelength guiding photoreceptors, as well as sparse, intact inner segments within the area of photoreceptor disruption.
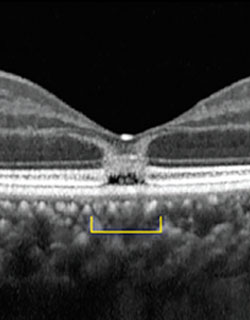
These findings, Dr. Deobhakta notes, could provide the scientific community with fresh insights on how photoreceptors die in a host of diseases involving the central part of the retina, including macular degeneration. And that knowledge, in turn, could provide valuable clues about how to effectively treat them.
For NYEE’s latest solar retinopathy patient, a medical breakthrough able to reverse the permanent blind spot etched on the retina of her left eye—making everyday activities like reading and TV-watching a challenge—couldn’t come soon enough. “At least we now have a much better understanding of the reason behind her injury,” acknowledges Dr. Deobhakta, “and that represents a huge step forward for patients.”
Targeting Macular Degeneration Through State-of-the-Art Imaging
Advanced imaging modalities are enabling researchers at New York Eye and Ear Infirmary of Mount Sinai (NYEE) to peer into the retina in ways they never could before, pinpointing vascular defects and blood flow abnormalities that could provide clues to the biologic footprint of macular degeneration. “With their higher and higher resolutions, these technologies are allowing us to more accurately measure the circulation and the thickness of the layers of the retina,” explains R. Theodore Smith, MD, PhD, Director of Biomolecular Retinal Imaging at NYEE and Professor of Ophthalmology and Neuroscience at the Icahn School of Medicine at Mount Sinai. “And that’s opening up to us a new world of research in terms of previously unexplored ocular tissue.”
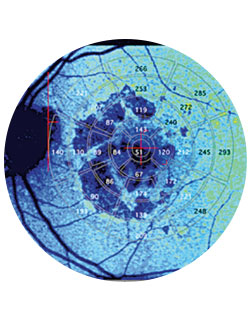
Despite the critical role of the choroid in supplying blood to the outer third of the neuroretina and the retinal pigment epithelium, details about choroidal circulation have remained largely unknown due to poor resolution and reproducibility of previous imaging techniques, including indocyanine green angiography and ultrasound. Imaging of the choroid has improved dramatically, however, with the advent of spectral domain optical coherence tomography (SD-OCT) and enhanced depth imaging SD-OCT (EDI SD-OCT), according to Dr. Smith. Further improvements are being made through swept source optical coherence tomography (SS-OCT) and OCT angiography. Thanks to these state-of-the-art techniques, Dr. Smith’s lab has visualized (Fig 1) segments of the choroid down to the capillary level, and found that individuals with coronary artery disease have a thinner macular choroid, something that could be a biomarker for CAD, as well as for identifying patients with increased risk of outer retinal disease.
Multimodal advanced imaging, including autofluorescence imaging, also figured heavily in another study designed to show the association between geographic atrophy, the advanced dry form of age-related macular degeneration (AMD), and renal impairment in people with AMD. While the link between AMD and chronic kidney disease is well known, Dr. Smith and his team were the first to investigate its biological pathogenesis by looking at blood supply to the outer portion of the retina—the choroidal circulation, which can be impacted by the same type of vascular dysfunction as the kidneys, both being end-arterial organs. The researchers found that individuals with AMD and decreased renal function are more likely to present with geographic atrophy at some point during their disease.
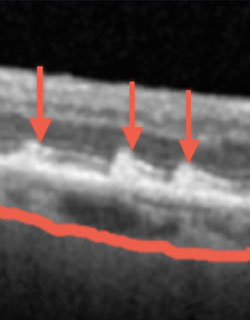
“What we have to recognize is that this part of macular degeneration is a systemic vascular disease, and that patients are going to have to be treated accordingly,” says Dr. Smith, who joined NYEE in September 2017 from NYU Langone Medical Center. “This means they should be controlled with regard to their cholesterol levels, blood pressure, and cardiac status, which to some extent takes it out of the eye clinic. We can still be involved, though, by providing the right antioxidant supplements and other treatments beneficial to macular health.”
Dr. Smith’s investigative work into macular degeneration has also benefited from sophisticated hyperspectral autofluorescence imaging. This technology, first used by scientists to examine the surface of the Earth from space and more recently adapted to the field of medicine, is enabling NYEE researchers to capture, measure, and analyze spectral emissions of light from the retinal pigment epithelium (RPE) to better understand that layer’s chemical composition and its possible role in macular health. NYEE is one of the leading research laboratories in the country to deploy hyperspectral autofluorescence imaging in the investigation of human eye tissue.
A major finding from the team’s hyperspectral imaging was that it detected underneath the RPE a very distinct spectral signature for drusen, and drusen precursors, in the macula. “This has potentially major diagnostic and prognostic import,” advises Dr. Smith, “because if we can identify early warning signs in patients who might be progressing to macular degeneration, we can start treatment sooner.”