The Milton and Carroll Petrie Department of Urology
Our renowned team of physician-scientists is addressing a variety of challenges, including those presented by prostate and kidney cancers.
The Milton and Carroll Petrie Department of Urology
At the Milton and Carroll Petrie Department of Urology at the Icahn School of Medicine at Mount Sinai, our renowned team of physician-scientists is addressing a variety of challenges, including those presented by prostate and kidney cancers. This specialty report highlights:
- A patient who presented with a rare 5 cm renal arterial aneurysm that was removed without complications using a novel approach
- Two genomic studies that may help advance the field toward the goal of truly personalized prostate cancer diagnostics and treatment
- A pregnant patient with a highly rare renal tumor that traveled to her aortic artery just weeks before her due date
- Our team that was the first in the world to perform gold nanoparticle prostate cancer ablation, part of a clinical trial spearheaded by a faculty member uniquely board certified in both urology and interventional radiology
Repairing a 5 cm Kidney Aneurysm
Advanced Imaging and Robotics Aid in Difficult Surgery
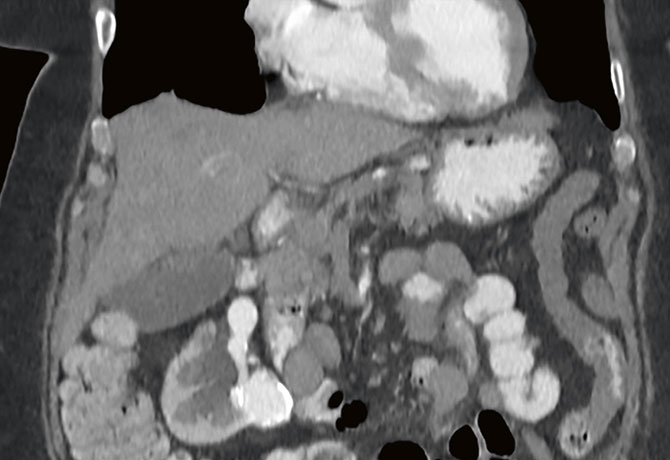
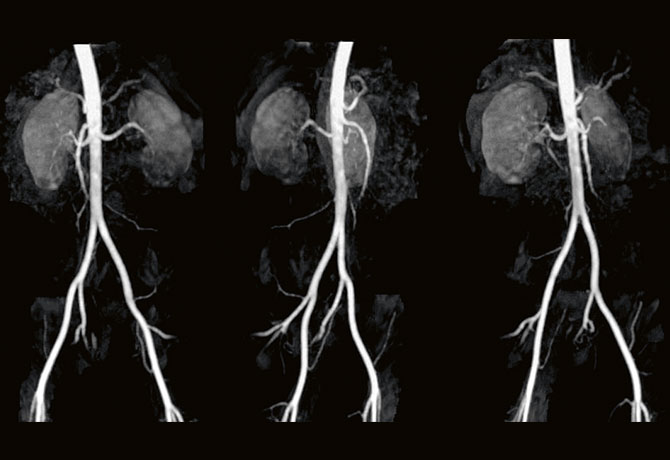
Merely two percent of aneurysms occur in the kidney, and one exceeding 2 cm is exceedingly rare. In August 2016, Ketan K. Badani, MD, Vice Chair of the Department of Urology at the Mount Sinai Health System and Director of the Comprehensive Kidney Cancer Program, saw a female patient, age 65, with a family history of vascular malformations, specifically aneurysms. Her sister had survived a brain aneurysm, and her father had suffered a fatal aneurysm of the femoral artery. The patient’s mass was suspected during physical examination (where a bruit was auscultated in the abdominal region by her primary care physician, Maria Brountzas, MD, of the Mount Sinai Health System), and a vascular 5 cm mass lesion was seen on the right kidney during follow-up ultrasound.
Dr. Badani then ordered a magnetic resonance angiogram at The Mount Sinai Hospital and discovered that the patient had an unusual arterial configuration: she had several small saccular aneurysms (of approximately 1 cm each) throughout her right kidney and a large, 5 cm lower pole aneurysm with aberrant surrounding vasculature. Additionally, she had a duplicated collecting system.
Dr. Badani requested a consultation with Mount Sinai’s Interventional Radiology team, which can, in most cases, perform selective renal embolization. However, given the size of the lesion, an endophytic component embedding into the kidney, and the unusual arterial anatomy, the patient did not feel comfortable with the risks of that procedure and potential need for emergent surgery. Given her family history and risk of spontaneous rupture of this large lesion, she decided instead to have Dr. Badani perform surgery, because his extensive experience in robotic partial nephrectomy could be utilized to treat her condition.
A robotic approach would allow for optimal operating field magnification and a high level of precision to identify the selective arterial branches feeding the lesion, utilizing both intraoperative ultrasound Doppler and immunofluorescence vision.
During the operation, the main renal artery, followed by the large aberrant branches, was dissected until the large aneurysm and surrounding vasculature were identified. Utilizing near-infrared imaging and immunofluorescence guidance, Dr. Badani sequentially identified each branch artery and clipped only the feeding vessels to the lesion. Then, as in a typical robotic partial nephrectomy scenario, he utilized his FAST (First Assistant Sparing Technique) approach, removing the lesion without any warm ischemia time. Estimated blood loss was less than 25 cc, and the patient was discharged after an overnight stay without compromise to her renal function.
According to Dr. Badani, this surgery is an excellent example of how experience with advanced imaging technologies during robotic partial nephrectomy can be utilized in many different ways in a safe and effective manner. He explains, “During robotic kidney cancer surgery, we routinely utilize a picture-in-picture video display of the operative field and real-time ultrasound to delineate the tumor and margins. Additionally, we toggle between a traditional white-light view of the operative field and a near-infrared view to directly visualize active blood flow and regional ischemia based on selective artery clamping. Both of these techniques, refined from experience with robotic partial nephrectomy, were utilized to treat this renal aneurysm, contributing to an excellent outcome.”

Truly Personalized Prostate Care
Genomic Studies Aim to Refine Diagnostics and Treatment
Two genomic studies under way at the Department of Urology—single-cell genomics profiling and racial disparity profiling—may help advance the field toward the goal of truly personalized prostate cancer diagnostics and treatment.
These studies are among numerous genomic studies being conducted within our Uro-Genomics and Uro-Translation research programs. Many of our initiatives are taking place in collaboration with other prestigious global institutions and Mount Sinai colleagues including Eric Schadt, PhD, Jean C. and James W. Crystal Professor of Genomics, and Kenneth Haines, MD. These collaborations and the work of the Tewari laboratory, led by Shalini Singh, PhD, in conjunction with our large and highly diversified patient cohort, make us uniquely qualified to analyze and understand the molecular drivers of aggressive prostate cancer by understanding tumor heterogeneity.
Single-Cell Genomics-Based Profiling
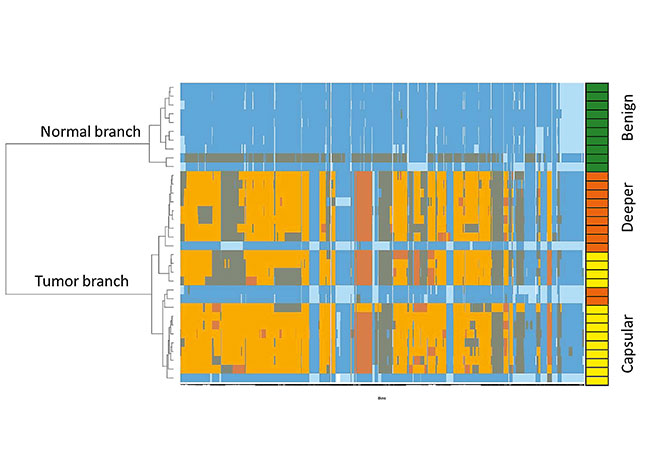
Current risk-stratification approaches for prostate cancer are imperfect. For the past four years, in collaboration with Cold Spring Harbor Laboratory (CSHL), we have dissected the clonal evolution of primary prostate cancer using sparse single nucleus sequencing. This is the first approach of its kind and tackles two major hurdles in diagnosing prostate cancer: intratumor heterogeneity and diagnostic uncertainties in invasive biopsies.
The process involves three steps: 1) single nucleus sequencing, where tissue or other samples such as fine-needle aspirates are prepared and flow-sorted as single nuclei, which are then subjected to whole genome amplification, library preparation, and sequencing; 2) data processing, in which the results from step 1 are mapped to the human genome and processed informatically, and copy-number profiles are obtained; and 3) data integration and visualization. The figure below shows a combined heatmap and dendrogram showing copy-number losses and gains. We at Mount Sinai have expanded the work we did with CSHL and have been successful in exploring fine-needle aspirates and urine samples as minimally invasive or noninvasive approaches to diagnose prostate cancer at the level of single cells.
Racial Disparity–Based Profiling
Understanding racial disparities in prostate cancer is a critical unmet need. Despite a decline of about 37 percent in mortality during the past 20 years, the disparity has remained unchanged. To understand this disparity, we are carrying out epigenomic profiling of tumors from black men to define the biological drivers of aggressive disease and to advance risk stratification. Recently, the cancer genome atlas (TCGA) project, the largest prostate cancer tumor-profiling study, with 498 samples, was completed. However, black Americans comprised only 11 percent of the total cohort. We carried out a comparative statistical analysis of this data and identified novel differentially expressed genes between black and Caucasian men. The observed differences suggest that prostate cancer in black men may be a different disease subtype, which warrants a more comprehensive profiling study.
A Unique Renal Carcinoma
Tumor Extending Into Vena Cava Removed by a Team Effort
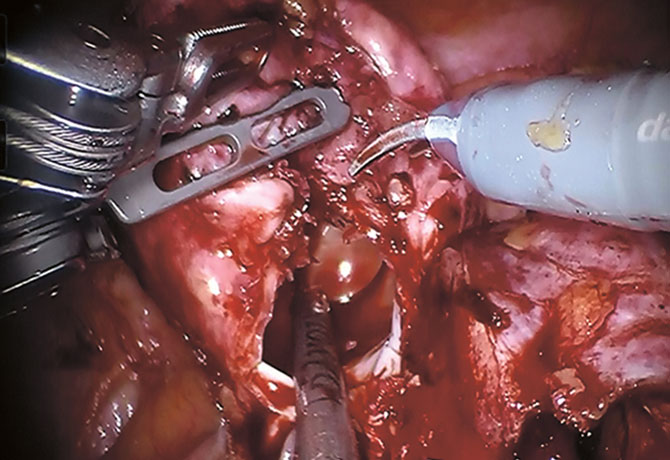
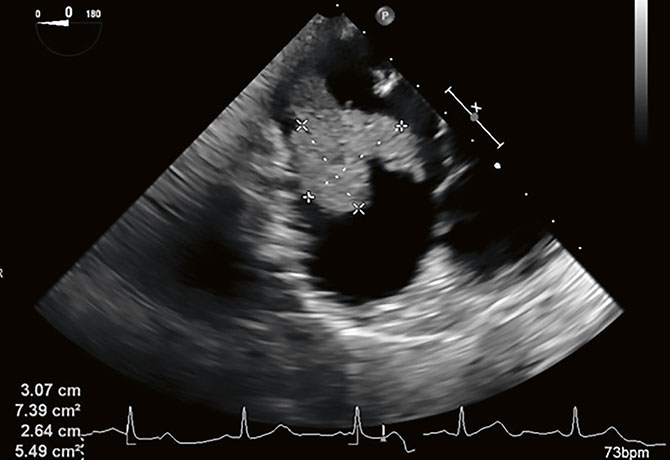
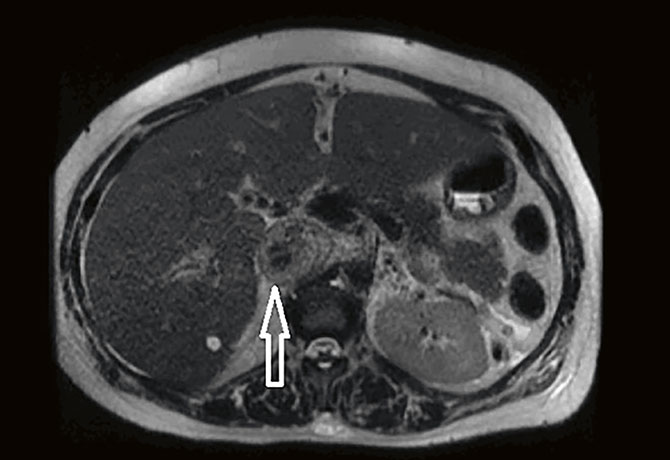
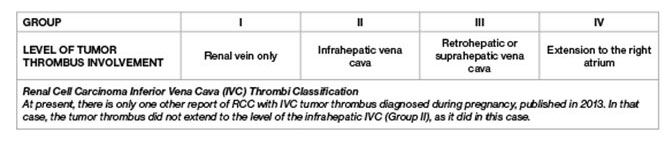
A multidisciplinary surgical and medical team was convened at Mount Sinai to treat a rare case of renal cell carcinoma (RCC) in a pregnant woman in the spring of 2016. Not only is RCC unusual in pregnant women (only 106 such cases had been reported worldwide as of 2015), but our patient had an extension of her tumor into the vena cava up to her right atrium (level IV tumor thrombus), making this a unique case that has never been reported in the literature to date. No management guidelines exist for this situation, but timing of surgery and approach are paramount and must be individualized for successful outcomes.
Our patient, Brenda T., 37 years old at 29 weeks of gestation, was incidentally found on fetal ultrasound to have a 6.0 x 6.0 cm solid mass in the lower pole of the right kidney. When the patient presented to an outside hospital for shortness of breath, Doppler ultrasound of the bilateral lower extremities demonstrated a right common femoral vein thrombus. Subsequently MRI confirmed the renal mass with tumor thrombus within great vessels and her right atrium. The patient was then referred to Reza Mehrazin, MD, Assistant Professor of Urology at the Icahn School of Medicine at Mount Sinai.
The concerns facing our team were sizable: the potential for fetal and maternal morbidity or mortality, a thromboembolic event (increased by the presence of lupus anticoagulant antibodies, active malignancy, and pregnancy), and intraoperative hemorrhage. Our team consisted of specialists in urologic oncology, cardiothoracic surgery, hepatobiliary surgery, obstetrics, maternal-fetal medicine, cardiothoracic anesthesia, and pulmonary critical care.
They decided to stage the procedures in the best interest of survival for both the patient and her fetus: First, the patient underwent an uncomplicated cesarean delivery at 31 weeks of gestation. Four days postpartum, a team of surgeons led by Dr. Mehrazin performed a right radical nephrectomy and a vena cava and right atrial thrombectomy on cardiopulmonary bypass. On postoperative day eight, the patient was discharged home.
“Management of kidney cancer with tumor extension into major vessels and the heart, not to mention during pregnancy, requires a multidisciplinary and well-coordinated approach to determine the optimal timing and approach to surgery,” Dr. Mehrazin says. “Such cases should be handled only by a team of experts at a facility where treatment decisions are individualized to improve outcomes.”
Using Gold to Take Aim at Tumors
Trial to Test If Nanoparticles Can Help Focus Ablation
Prostate cancer specialists at Mount Sinai have treated their first patient in an exciting new clinical trial of gold nanoparticle tumor ablation, a novel procedure designed to minimize side effects after radical prostatectomy or radiation therapy while still completely eliminating a cancerous tumor.
Martin Feeney, 70, a retired bond salesman, was diagnosed with low-grade, low-volume prostate cancer four years ago. That was distressing for him, but more frightening was the sepsis he developed as a result of a rectal biopsy. Once recovered, Mr. Feeney was advised that he could follow an active surveillance program to monitor his cancer, but he was nervous about the regimen of biopsies required under this treatment plan. He delayed his next biopsy for over three years.
Fast forward to early 2016, when he met Art Rastinehad, DO, Director of Focal Therapy and Interventional Urologic Oncology at the Mount Sinai Health System, and learned he could undergo a biopsy that combined an MRI of the prostate with a new approach pioneered at The Mount Sinai Hospital. Dr. Rastinehad was the first in the world, in 2015, to use the new electromagnetically tracked UroNav fusion biopsy device from Invivo, which helps patients avoid serious infections by enabling the biopsy to be performed through the perineal route rather than via the rectum, a frequent source of infections. Mr. Feeney was enthusiastic to proceed.
This time, the biopsy result was less favorable. Mr. Feeney had a clinically significant Gleason 3 + 4 cancer. He was faced with choosing either surgery or radiation to treat it.
Fortunately for Mr. Feeney, Dr. Rastinehad is also leading the national team investigating a new procedure—gold nanoparticle-directed focal laser ablation—that can treat prostate lesions locally instead of removing or irradiating the entire prostate. This nanotechnology-based option can give patients more confidence that their cancer will be successfully removed without worrying about side effects that can impact their quality of life.
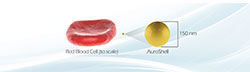
In May, Mr. Feeney was the first man to undergo this type of ablation of his lesion. Gold nanoparticles are trademarked as AuroShell® particles and regulated as a device by the U.S. Food and Drug Administration. They are systemically infused into the blood of the patient and are deposited in hypervascular tumors via leaky capillaries.
The figure below illustrates the construction of the nanoparticle and its size relative to a red blood cell. These particles are activated by near-infrared radiation from a laser, which they absorb and release as heat, destroying the lesions in which they are embedded but not the adjacent nerve tissue or blood vessels. Remaining nanoparticles are removed from the bloodstream via the spleen and are typically gone from the body within a few weeks.
Patients who may benefit from this groundbreaking treatment are those who have one or two focal lesions proven on a targeted biopsy with a Gleason score of 7 or less.
The exciting part of this trial is that we are using information from targeted biopsies to treat prostate cancer in a localized, precise way, and avoiding radical surgery, which can endanger the quality of life. Patients can go back to work the following day; there’s no down time.
Three months afterward, follow-up imaging and biopsy of his prostate show that Mr. Feeney has no sign of cancer and has experienced no side effects. “It’s like magic,” he says. “I had absolutely no recovery time, no bleeding or pain.”

Precise Prostate Surgery
New Technology Saves the Neurovascular Bundle
Accomplishing negative surgical margins while preserving sexual function and continence plays a significant role in determining the success of surgical intervention for prostate cancer. With an increasing number of men being diagnosed at younger ages, achieving this “trifecta goal” is of utmost importance to preserving quality of life. But as cancer control is the foremost guiding principle, eradicating the tumor and preserving the nerves becomes a delicate juggling act for the surgeon.
Researchers under the leadership of Ash Tewari, MBBS, MCh, have previously described that the prostatic neurovascular bundle (NVB) enmeshes the gland in an intricate trizonal hammock of nerves (below). This complex network of nerves increases the surgeon’s opportunities for nerve rescue, but the sites of oncological surveillance increase concomitantly. It is for this reason that real-time analysis of the entire neurovascular tissue-adjacent circumference is the optimal solution to safeguard the maximum number of nerves while achieving comprehensive histological surgical margins.
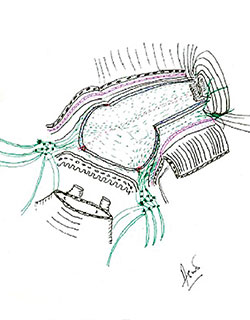
Credit: Ash Tewari, MBBS, MCh
At Mount Sinai, we utilize the NeuroSAFE (neurovascular structure–adjacent frozen section examination) technique of intraoperative frozen section analysis for all patients who are appropriate candidates (more than 70 percent). The technique is used in addition to our athermal, traction-free, risk-stratified, graded nerve-sparing approach, which is now the standard of care at our institution. NeuroSAFE, developed by our collaborators at the University of Hamburg, allows real-time histological assessment of surgical margins for the absence or presence of cancer intraoperatively, and helps in performing a nerve-sparing procedure without compromising oncological safety. Investigators at the Icahn School of Medicine at Mount Sinai have extended NeuroSAFE to sphincter-safe evaluation at the apex, which allows for improved continence preservation. Systematic NeuroSAFE has been reported to significantly increase nerve-sparing frequency and to reduce positive surgical margins. Also, patients with NeuroSAFE-detected positive margins were converted to prognostically more favorable negative surgical margins.
Our team is also developing an MRI-guided intraoperative frozen section technique and sphincter-safe approaches to expand opportunities for nerve sparing and continence preservation.
Immunotherapy, Early?
Seeking to Treat Tumors Before They Proliferate
The first immunotherapeutic agent approved by the U.S. Food and Drug Administration for any cancer was for prostate cancer. Yet, most of the agents for prostate cancer being tested at this time are seen as treatment alternatives for high-risk patients. Novel strategies are needed for patients with less advanced disease to prevent tumors from proliferating and/or resisting the immune system.
Based on patients’ response to immunotherapeutic agents to date, Mount Sinai Department of Urology researchers, including Nina Bhardwaj, MD, PhD, Matt Galsky, MD, Sujit Nair, PhD, and Principal Investigator Ash Tewari, MBBS, MCh, see tremendous potential in immunotherapy for early intervention for treatment of cancer patients with localized, aggressive disease and oligometastasis in addition to metastasis. The potential benefits of this focus are profound: eliminating the need for invasive treatment such as surgery or the ravages of radiation or chemotherapy, and in all cases, preserving quality of life, including urinary and sexual function.
Our Uro-immuno-oncology program focuses on employing novel and high-throughput methods for the development of immunomodulatory agents for cancer, assessing the efficacy of such agents in combination therapy, advancing immunotherapy for early and clinically localized aggressive cancers, and understanding the impact of immunotherapy in racial disparity of cancer.
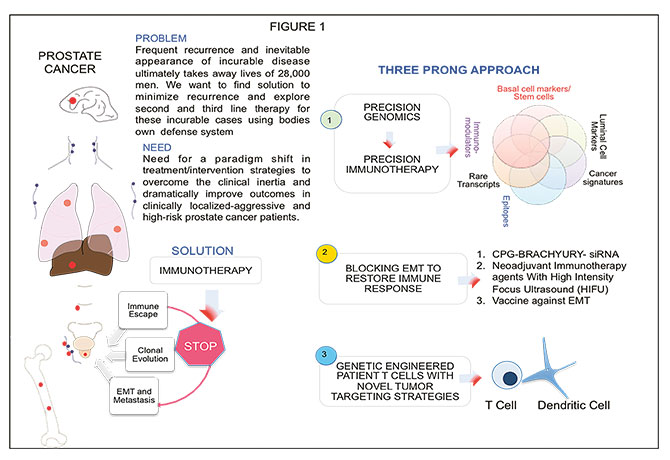
Our approach is three-pronged (see figure below):
- Employing precision genomics as the foundation on which (precision) immunotherapy solutions are designed. Our ongoing studies are focused on identifying cancer-favorable genomic alterations, gene-regulatory networks, and pathways of tumor tolerance. Studies will enable identification of unique therapeutic targets including neoantigens for peptide/antibody-based vaccines for clinical intervention and immunotherapy. One study target is CpG-oligodeoxynucleotide (ODN), a TLR-9 agonist coupled with siRNA for STAT3 or BRACHYURY (see figure below).
- Exploring avenues to enhance the therapeutic benefits of high-intensity focus ultrasound (HIFU) by employing novel combination strategies of HIFU with other anti-cancer techniques.
- Genetically engineering patient T cells with novel tumor-targeting strategies.
3 Kidney Cancer Studies
Initiatives Focus on Biomarkers and Stem Cells
The Department of Urology at Mount Sinai is conducting three major research initiatives, under the leadership of Ketan K. Badani, MD, Director of the Comprehensive Kidney Cancer Program, that have the potential to shape the direction of kidney cancer diagnosis and treatment.
Biomarker Discovery of Renal Cell Carcinoma
Genomic studies have identified more than eight distinct kidney cancer genes, impacting therapeutic protocols. Because there are so many varieties of kidney cancer, the challenge is to comprehensively understand its genetic basis and disease pathways. We are developing preclinical models to test drug efficacy.
During surgery, we are collecting tumor tissue, benign kidney tissue, blood, and urine for our biorepository. We are uniquely positioned to conduct this study based on strong collaboration with physician-scientists from Mount Sinai’s renowned Departments of Pathology, Medical Oncology, and Genomics. All patients being treated for a renal mass by Dr. Badani are eligible to enroll.
Urine Biomarkers as Predictors of Acute Kidney Injury After Surgery
Our program will be examining the risk of acute kidney injury after kidney cancer surgery, through a novel urine marker panel. The results will have a significant impact on treatment decision making and patient counseling and, importantly, inform the choice of partial nephrectomy versus total nephrectomy.
In this study, our team will utilize information from each of these proteins and develop an algorithm based on the various levels of expression of all of the proteins.
Stem Cell Regeneration of Kidney Tissue Subsequent to Partial Nephrectomy
A new randomized clinical trial directed by Dr. Badani and his team at Mount Sinai is launching in 2017. This study will examine the use of amniotic stem cells to help regenerate kidney tissue during and after partial nephrectomy to restore normal kidney function. We will enroll patients who are undergoing robotic surgery for a kidney mass.
Message from the Chair: Ash Tewari, MBBS, MCh

We are pleased to update you on the exciting work taking place at the Milton and Carroll Petrie Department of Urology at the Icahn School of Medicine at Mount Sinai. In our laboratories and our clinics, our renowned team of physician-scientists is addressing the challenges presented by urologic cancers. The number of prostate cancer cases diagnosed in the United States continues to decline, but racial disparity and the apparent increase in aggressive disease means we need to stay focused on the origins and progression of this cancer. Therefore, the goal of our first-of-its-kind, integrative, epigenomic analysis of the prostate tumor and its microenvironment is to determine the biology driving the disparity between black and Caucasian men with prostate cancer. Our new single-cell profiling approach is designed to refine risk stratification for prostate cancer across all demographics.
We are proud to present stories about challenging and complex cases referred to the Mount Sinai team. You will read about a pregnant patient with a highly rare renal tumor that traveled to her aortic artery just weeks before her due date and a patient who presented with a rare 5 cm renal arterial aneurysm that was removed without complications using a novel approach. Our team was also the first in the world to perform gold nanoparticle prostate cancer ablation, as part of a clinical trial spearheaded by a faculty member who is uniquely board certified in both urology and interventional radiology.
Our department is experiencing tremendous growth across the Mount Sinai system. I am delighted to welcome to our faculty several renowned physicians. At Mount Sinai St. Luke’s, Robert Valenzuela, MD, is a leader in complex prosthetic urologic surgery, and Gerald Hoke, MD, MPH, is highly regarded for his work in men’s and community health. Nikhil Waingankar, MD, a specialist in kidney cancer, joins our Mount Sinai Queens team. Steven Kaplan, MD, heads up our Men’s Health Program in Midtown Manhattan. And Vannita Simma-Chiang, MD, is new to our Elmhurst faculty.
Finally, we are expanding our footprint at Mount Sinai Downtown. Our newly renovated site will include state-of-the art procedure rooms, a fluoroscopy suite, conference rooms, waiting rooms, exam rooms, physician offices, and a resident/fellow office.
Ash Tewari, MBBS, MCh, is the Kyung Hyun Kim, MD, Chair in Urology of The Milton and Carroll Petrie Department of Urology.
Department of Urology
Download the Winter 2017 Specialty Report