The Tisch Cancer Institute
At The Tisch Cancer Institute, a National Cancer Institute-designated center, our multidisciplinary approach to cancer treatments and clinical breakthroughs may one day put an end to cancer.
The Tisch Cancer Institute
Innovation and transformation energize our programs and reflect The Tisch Cancer Institute’s world-class research. We are a National Cancer Institute-designated Cancer Center and have joined an elite group of U.S. institutions committed to the research and treatment of cancer.
This specialty report highlights:
- Mount Sinai’s Human Immune Monitoring Core (HIMC), which actively works with clinicians to help characterize the cellular and molecular immune profiles of tumor lesions in their patients
- Our Vaccine and Cell Therapy Laboratory, which supports the development and manufacturing of novel vaccines and cell-based therapies for cancer and infectious diseases
- Novel research into therapies that improve the standard of care for patients who develop acute graft-versus-host disease (GVHD) following bone-marrow transplantation
- Pioneering research collaborations with Celgene and three leading U.S. academic institutions
- Young researchers who bring insights from physics, mathematics, and genomics
Detailed Studies of the Immune System
At the Human Immune Monitoring Core
For years, the endpoint that defined whether a cancer treatment was successful was the patient’s survival. Today, Mount Sinai’s researchers are able to understand how a treatment is working on a cellular and molecular level, with the support of the Human Immune Monitoring Core (HIMC) at the Icahn School of Medicine at Mount Sinai, which also analyzes hundreds of preclinical and patient samples from academic medical centers throughout the New York City area.
The HIMC runs one of the nation’s largest full-service mass cytometry programs, providing researchers and clinicians with the extensive scientific and technical expertise that allows them to leverage a wide range of powerful technology platforms for detailed studies of the immune system.

Working closely with researchers, the HIMC team helps them understand the underlying basis for disease and guides them with the development of new and improved immune therapies.
Joshua Brody, MD, Assistant Professor of Medicine (Hematology and Medical Oncology), used the Core in 2014, when he tested a unique in-situ vaccine for low-grade B-cell lymphoma that was aimed at increasing the number of dendritic cells. Dr. Brody collected blood samples from his patients during their treatments and provided them to the HIMC to perform in-depth analyses of their immune cells. Using a CyTOF2 machine—which is able to analyze 40 biomarkers on millions of individual cells in a single sample and pinpoint those that are particularly interesting— the HIMC team was able to show Dr. Brody that the treatment was working. The team also explained how the treatment was affecting his patients’ dendritic cells and other immune cells.
“We saw the confirmed effect of increased dendritic cells in the patient’s blood,” says Sacha Gnjatic, PhD, Associate Professor, Medicine (Hematology and Medical Oncology), who is also the HIMC’s Co-Director with Miriam Merad, MD, PhD, Professor of Oncological Sciences, and Medicine (Hematology and Medical Oncology).
“It’s hard to make a jigsaw puzzle if you can’t see the pieces,” says Dr. Brody. “The higher resolution of the CyTOF draws a whole picture. It offers us a way of validating the mechanism of these immunotherapies.” In 2017, Dr. Brody will be leading a new in-situ vaccine study that uses the PD-1 checkpoint inhibitor.
What sets the HIMC apart from similar centers around the country is the level of support provided by the Core’s scientific staff, which includes Adeeb Rahman, PhD, Director of Technology Development, and computational scientist El-ad David Amir, PhD. Dr. Amir was a developer of viSNE, a tool that helps researchers map high-dimensional cytometry data onto two dimensions while conserving the high-dimensional structure of the data. The Core plans to purchase a second CyTOF2 machine in coming months to keep up with demand.
Cancer Immunotherapy
Advances at the Vaccine and Cell Therapy Lab
The ability to create a personalized genome vaccine represents a giant leap forward in the evolution of cancer immunology. Under the direction of Nina Bhardwaj, MD, PhD, Director of Immunotherapy and Professor of Hematology and Medical Oncology, The Tisch Cancer Institute is among the few U.S. academic cancer centers with the infrastructure in place to actively pursue this research in its Vaccine and Cell Therapy Laboratory, which opened in 2015.
Dr. Bhardwaj and Philip Friedlander, MD, PhD, Assistant Professor, Medicine (Hematology and Medical Oncology), are currently recruiting patients with squamous cell carcinoma, non-small cell lung cancer, bladder cancer, and other solid tumors for a phase I, proof-of-concept, adjuvant trial that explores the safety of these fully personalized vaccines.

After pathologists extract RNA and DNA from a patient’s tumor, they are sequenced and examined for mutations in potential tumor antigens that could possibly trigger an immune response, efforts undertaken in collaboration with Eric Schadt, PhD, Director of the Icahn Institute for Genomics and Multiscale Biology, and Chair of the Department of Genetics and Genomic Sciences, and Jeffrey Hammerbacher, Assistant Professor, Genetics and Genomic Sciences. Individual vaccines are then created from the mutation-derived tumor antigens that arise as a direct result of the somatic variations that occur during carcinogenesis.
“Ideally, immune therapy produces a controlled response that is specifically targeted toward the population of malignant cells, with minimal risk of adverse reactions and latent, lifelong protection,” says Dr. Friedlander.
Other strategies in cancer immunotherapy are also being advanced at The Tisch Cancer Institute. At the European Society for Medical Oncology meeting in October 2016, Matthew D. Galsky, MD, Director of Genitourinary Medical Oncology, showed that 20 percent of patients with metastatic bladder cancer responded to the immune checkpoint inhibitor nivolumab, and continued to do so at a rate exceeding that of chemotherapy.
In 2016, Dr. Galsky contributed to the clinical development of atezolizumab, another immune checkpoint inhibitor that was the first drug approved for use in bladder cancer in more than 30 years. “These drugs are transformative in patients who respond,” says Dr. Galsky, who is also Professor of Medicine (Hematology and Medical Oncology). “Before this we had nothing. This is the platform from which to build now.”
More than two years ago, Lee Eric Newton, a single father, whose cancer had spread to his brain, joined Dr. Galsky’s clinical trial using atezolizumab. Today, Mr. Newton remains in stable condition. “I’m living a productive lifestyle,” says Mr. Newton. “The ability to run a household, find time to vacation at my brother’s home in Florida—that wasn’t even a consideration when I was receiving chemotherapy.”
Graft-Versus-Host Disease
Enters a New Era in Precision Medicine
Physician-scientists at The Tisch Cancer Institute have been awarded $10 million from the National Cancer Institute to continue their novel research into therapies that improve the standard of care for patients who develop acute graft-versus-host disease (GVHD) following bone-marrow transplantation. Bone-marrow transplantation (BMT) is often successfully used to treat diseases such as leukemia and lymphoma. Acute GVHD, which affects approximately 50 percent of patients, occurs when the donor’s immune cells attack the patient’s tissues, producing potentially fatal results.
Led by world-renowned GVHD researchers James L. Ferrara, MD, DSc, and John E. Levine, MD, the five-year grant will enable the Icahn School of Medicine at Mount Sinai to spearhead a consortium of 20 major medical centers in the United States and Europe that will share patient data.

With Mount Sinai coordinating the information, the researchers plan to examine blood samples from 1,000 BMT patients each year to learn more about the disease and refine new diagnostics and treatments. U.S. participants will include Massachusetts General Hospital, the Mayo Clinic, the Ohio State University Medical Center, and Vanderbilt University Medical Center. Several German academic medical centers, including University Hospital Regensburg, also will be sharing data with Mount Sinai.
“Our grant proposal was very enthusiastically received by the National Cancer Institute, which was impressed with the support that Mount Sinai has provided to run this international consortium,” says Dr. Ferrara, the Ward-Coleman Chair in Cancer Medicine and Professor of Medicine (Hematology and Medical Oncology), Pediatrics, and Oncological Sciences.
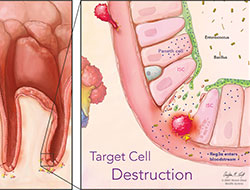
A gastrointestinal crypt is under attack from donor T-cells (red globules), which destroy intestinal stem cells (ISCs) and adjacent Paneth cells that protect them. When the ISCs disintegrate, bacteria (enterococcus, bacillus) from the intestinal lumen enter the bloodstream, causing widespread infection and inflammation. An antimicrobial peptide made by Paneth cells (Reg3a) also enters the bloodstream at the same time, acting as a biomarker of crypt damage. Dr. Ferrara’s $10 million grant is focused on exploring the biological properties of biomarkers such as Reg3a and new clinical trials to treat patients at high risk of severe gastrointestinal GVHD.
Dr. Levine is a Professor of both Medicine and Pediatrics at the Icahn School of Medicine at Mount Sinai. He and Dr. Ferrara have been longtime colleagues in the study of GVHD, first at the University of Michigan and for the past couple of years at Mount Sinai, where their groundbreaking research is now being used to inform patient care.
Early diagnosis is the key to successfully treating GVHD, Dr. Ferrara says. To that end, he and Dr. Levine have discovered and validated a biomarker signature in the blood of two plasma proteins that can predict which patients will develop the most severe disease. They are now testing a new treatment that, if given early enough, should keep the disease from spreading through the patient’s gastrointestinal tract to the rest of the body, which is the normal course that GVHD takes.
With the new protocols in place, Mount Sinai has become the first health system in the world to test patients for early signs of GVHD—usually within the first month after BMT—and to offer them the new treatment. Mount Sinai recently treated its first high-risk patient with positive results.
“This is the beginning of precision medicine for BMT patients,” Dr. Ferrara says.
For the past 40 years, physicians have treated patients with a range of high-dose steroids that have differed in type and dose, depending on the medical center. The large number and variety of centers participating in the consortium will allow clinicians to standardize the treatment of GVHD for the first time.
Pioneering Research Collaboration
With Celgene and Three Research Institutions
The Tisch Cancer Institute at the Icahn School of Medicine at Mount Sinai and three other leading U.S. academic institutions recently established a pioneering research consortium to accelerate the discovery of new treatments for cancer.
In addition, Celgene Corp., a global biopharmaceutical company, paid $50 million to enter into four public-private collaboration agreements with each member of the new consortium for the option of developing and commercializing novel cancer therapeutics arising from their efforts.

Celgene awarded $12.5 million each to Mount Sinai, The Abramson Cancer Center at the University of Pennsylvania, The Herbert Irving Comprehensive Cancer Center at Columbia University Medical Center, and the Sidney Kimmel Comprehensive Cancer Center at Johns Hopkins.
Over the next 10 years, these academic cancer centers intend to present multiple high-impact research programs to Celgene with the goal of developing new lifesaving therapeutics. Subject to Celgene’s decision to opt in and license the resulting technologies, each program has the potential to be valued at hundreds of millions of dollars.
“This is a paradigm-shifting collaboration that further strengthens our innovative ecosystem,” says Bob Hugin, Executive Chairman of Celgene Corp. “We remain firmly committed to driving critical advances in cancer and believe the tremendous expertise of our collaboration partner institutions will be invaluable in identifying new therapies for cancer patients.”
The magnitude of the multi-institutional consortium and agreements will support the rapid delivery of disease-altering treatments to clinicians and ultimately benefit cancer patients, global health care systems, and society. Collectively, the four academic medical centers care for more than 30,000 new cancer patients each year and have nearly 800 faculty members involved in clinical care and basic and clinical cancer research. They are among the 69 institutions designated as Cancer Centers by the National Cancer Institute (NCI), serving as the backbone of the NCI’s research on cancer.
“We have an unprecedented opportunity to take basic cancer research all the way to drug development with one of the most creative pharmaceutical companies,” says Steven J. Burakoff, MD, Director of The Tisch Cancer Institute at the Icahn School of Medicine at Mount Sinai, and the Lillian and Henry M. Stratton Professor of Cancer Medicine. “These agreements are focused on basic and translational research that is conducted at academic institutions that treat a tremendous number of new cancer patients annually.”
Erik Lium, PhD, Senior Vice President of Mount Sinai Innovation Partners, who leads Mount Sinai’s development and commercialization division, helped spearhead the collaboration agreement.
“This consortium enables the four participating institutions to leverage their synergistic and individual strengths in research and patient care to develop new treatments for cancer,” he says.
Young Cancer Researchers
Bring Insights From Physics, Mathematics, and Genomics
Talented young researchers in theoretical physics, structural biology, mathematical modeling, and translational genomics—whose training once was considered far afield from translational medicine—are adding their unique insights to the field of cancer and claiming new discoveries.

Benjamin Greenbaum, PhD
Trained as a physicist and quantitative biologist at the Institute of Advanced Study in Princeton, New Jersey, Benjamin Greenbaum, PhD, concedes that he may have been considered the odd man out on a cancer research team just five years ago. Today, he works with clinicians at the Icahn School of Medicine at Mount Sinai to investigate how a little-known group of noncoding RNA molecules can touch off an immune response that encourages tumor growth. He also collaborates with a multidisciplinary team of investigators from institutions such as Princeton University, Massachusetts General Hospital, and Memorial Sloan Kettering Cancer Center to determine whether a synthetic form of vitamin D can render treatment-resistant pancreatic cancer cells vulnerable to a patient’s immune system.
“The sheer amount of data we’re able to collect and the need to find an underlying testable hypothesis from that data have changed the way we conduct research,” says Dr. Greenbaum, Assistant Professor of Medicine (Hematology and Medical Oncology). “That’s a job for which physicists are well trained.”
Dr. Greenbaum’s lab applies the tools used in theoretical physics, mathematics, and computer science to quantify the role of the immune system in several known cancers. “Very little of what I do concerns the physical properties of things,” he explains. “It’s more about applying the methods and skills from my physics training to try to better understand the evolution of tumors and viruses.”
That process involves building mathematical theories that result in predictions that can be rigorously tested in the lab. To perform his groundbreaking research on noncoding RNA molecules, Dr. Greenbaum’s team searched a large database of ncRNAs—the so-called “dark matter” of the genome—for aberrant patterns of nucleotides to see how they differed from the RNA transcribed in cancers. Working with Nina Bhardwaj, MD, PhD, Director of Immunotherapy and Professor of Hematology and Medical Oncology at The Tisch Cancer Institute, he learned that in some cancers, ncRNAs exhibit properties of pathogenic RNA, which causes them to stimulate an innate immune response. “We believe these ncRNAs may play a significant role in mediating immune responses against cancer,” he says, adding that much work remains to describe the precise interactions.
Dr. Greenbaum also is part of a team formed last year with funding from the Entertainment Industry Foundation’s Stand Up to Cancer initiative that is supporting researchers working to find a novel approach to curing pancreatic cancer, a malignancy that will take 42,000 lives in the United States this year. They are researching a synthetic vitamin D that essentially disables tumor defenses against immune-system attack. Executing this project requires massive sequencing of patients’ DNA, T-cells, and tumors. Physicists and quantitative biologists like Dr. Greenbaum are trying to make numerical sense of it all by counting and characterizing T-cells and their targets and using the data to develop models that predict T-cell response.
He says the relationships he has built with clinicians, molecular biologists, immunologists, and virologists within and outside The Tisch Cancer Institute have greatly influenced his work. “It’s a great opportunity because I’m connected to many clinicians and a giant hospital network,” Dr. Greenbaum says. “That keeps me up to date on what’s going on in my field and constantly reminds me of the real impact the work of people around here has on the lives of patients.”

Arvin Dar, PhD
While completing his postdoctoral studies at the University of California, San Francisco, Arvin Dar, PhD, became aware of the power of pure science to help unlock the mysteries of cancer. “I realized there were many opportunities to use basic science and chemistry to test mechanisms and ideas that the pharmaceutical industry would not pursue,” he says. “And so I transitioned from studying basic chemical biology to developing drugs that fight cancer.”
As Assistant Professor of Oncological Sciences and Pharmacological Sciences at the Icahn School of Medicine at Mount Sinai, Dr. Dar has focused on Ras-dependent cancers. He has won numerous awards and honors in the process, including the prestigious New Innovator Award for High-Risk, High-Reward Research from the National Institutes of Health, and a Young Investigator Prize from the Pershing Square Sohn Cancer Research Alliance.
One in four human cancers involves a mutant form of the Ras oncogene. Dr. Dar and his lab have responded to the challenge of developing drugs for these cancers, which remain largely untreatable, by investigating new targets and strategies for interrupting the Ras pathway. Prominent among them is kinase suppressor of Ras (KSR), a protein that can prevent the overactive, cancer-causing forms of Ras from stimulating other genes that lead to increased cell division.
Through large genetic screens in flies and worms, Dr. Dar’s lab identified mutations in KSR that could suppress the development of Ras-driven tumors. “As it turned out, most of these mutations were co-localized to a hot spot on the protein,” he says. “This identified for us a potential drug-binding pocket and a path forward for developing new compounds to combat Ras-driven cancers.” That discovery was recently published and has since opened unexplored therapeutic opportunities.
Significantly, Dr. Dar’s work goes beyond trying to find the next targets for cancers. “Using biology as our guide,” he says, “we ultimately attempt to execute our ideas through chemicals and small molecules that can become drugs.” Fueling this effort is the convergence of disciplines such as synthetic organic chemistry, structural biology, mass spectrometry, biochemistry, and model organism genetics. “Bringing these technologies to our collaborations and co-mingling with other people who think about cancer in different ways is, for me, the most exciting aspect of being at Mount Sinai. We think it gives us the chance to fill gaps in our knowledge and helps our ability to answer important questions.”
A textbook example is Dr. Dar’s collaboration with Ross Cagan, PhD, Professor, Developmental and Regenerative Biology at the Icahn School of Medicine at Mount Sinai. Using a cancer model built by Dr. Cagan’s lab around the fruit fly Drosophila, the researchers developed an investigational compound, AD80, which, for the first time, precisely targets multiple cancer genes—a process known as polypharmacology. Tested in mouse models, the drug (bearing Dr. Dar’s initials) proved far more effective and less toxic than standard cancer drugs that generally focus on a single target.
Says Dr. Dar of the unusual pairing of fly geneticists and medicinal chemists, “The magic is that we’ve used genetics in the fly to map out the best targets to inhibit and the best targets to avoid. Now we’re excited about the next step—moving into clinical trials.”

Yujin Hoshida, MD, PhD
From his early days as a hepatologist, Yujin Hoshida, MD, PhD, has worked tirelessly to bridge the worlds of scientific discovery and clinical practice. He applied his skills in medicine, cancer genomics, and bioinformatics to develop high-throughput genomics assays and innovative new ways to analyze in the lab archived specimens collected from patients. After Dr. Hoshida joined the Icahn School of Medicine at Mount Sinai in 2012 as an Assistant (now Associate) Professor of Medicine (Liver Diseases), his efforts bore impressive fruit.
He developed the first-ever gene test that can be used to identify patients with cirrhosis who are at highest risk for hepatocellular carcinoma (HCC), the most common type of liver cancer and the third leading cause of cancer mortality worldwide. The test, which he developed in collaboration with Josep Llovet, MD, Professor of Medicine and Director of Mount Sinai’s HCC Program, also helps determine whether a patient diagnosed with HCC is likely to have a recurrence of the cancer after the tumor is removed.
For Dr. Hoshida, who took up cancer genomics in earnest eight years ago while a postdoctoral fellow at the Broad Institute of MIT and Harvard, the discovery of the gene signature is “particularly gratifying because we can now explore ways to use it as both a diagnostic test and an opportunity to discover new drugs that might be effective against HCC.” This development, he adds, could prompt doctors to more closely monitor liver cancer patients who are at greatest risk of tumor recurrence and enable clinicians to identify patients who are good candidates for clinical trials of new agents in both adjuvant and chemopreventive settings.
Just as gratifying to Dr. Hoshida was his decision four years ago to become part of the Icahn School of Medicine’s Liver Cancer program—the only National Cancer Institute–designated liver cancer program in the country—and apply his deep experience in translational genomics research, particularly hepatocellular carcinoma.
“Being able to move my lab to the largest HCC program in the country has been a great opportunity to advance my work,” Dr. Hoshida says. Indeed, the Mount Sinai Health System leads the nation in the number of new HCC patients evaluated annually. Mount Sinai’s international reputation is based on its contributions to the surgical management of the disease, the strength of its liver transplantation program, and its burgeoning clinical research activities.
Dr. Hoshida’s research has gained new impetus through his collaborations with specialists from the divisions of oncology, liver disease, pathology, and radiology, and through his access to vital clinical specimens. In a branch of cancer where molecular indicators have proven elusive, Dr. Hoshida is focused on establishing prognostic biomarkers and generating a panel of genomic datasets representing the population with liver cirrhosis and cancer worldwide. He is the principal investigator of a National Institutes of Health–funded project that seeks to generate genomic profiles of 2,000 patients with cirrhosis linked to hepatitis B and C, alcohol abuse, and nonalcoholic fatty liver disease.
In addition, Dr. Hoshida is actively engaged with other researchers in Mount Sinai’s Liver Diseases program. They include Amaya Lujambio, PhD, who is using sophisticated mouse models to learn how genetic alterations in cancer cells contribute to tumorigenesis and what role cellular senescence plays in initiating liver cancer. Above all, he credits his ongoing relationship with Scott Friedman, MD, Chief of the Division of Liver Diseases, who “has been extremely supportive and allowed my lab to really grow at Mount Sinai.”
Message from the Director: Steven J. Burakoff, MD
The Tisch Cancer Institute treats approximately 10,000 new patients each year and experienced 62 percent growth in ambulatory visits between 2012 and 2015. We attribute this growth to the quality of our specialists who are on the frontlines of cancer prevention and control, early detection, and cancer research. These were among the strengths cited by the National Cancer Institute (NCI) in 2015, when we became an NCI-designated Cancer Center and joined an elite group of U.S. institutions committed to the research and treatment of cancer.

In addition to conducting innovative clinical trials and using new therapies for patients with metastatic bladder cancer, lymphoma, multiple myeloma, melanoma, and head and neck cancers, we provide early-screening programs in lung and skin cancer. Our NCI-designated program in liver cancer continues to grow and save lives.
At the same time, our ability to treat patients with comorbidities is enhanced by the collaborative care Mount Sinai provides. In working with our colleagues in the nation’s most highly ranked programs in geriatrics, cardiology, gastroenterology, endocrinology, nephrology, and neurology, we handle the entire spectrum of health care services that so many cancer patients require.
The field of cancer is moving quickly, and The Tisch Cancer Institute has been an early adopter. Mount Sinai’s Human Immune Monitoring Core (HIMC)—among the largest facilities of its kind on the East Coast—actively works with clinicians to help characterize the cellular and molecular immune profiles of tumor lesions in their patients. The Core houses a CyTOF2 Mass Cytometer—with another expected shortly—that enables physicians to identify immune predictors of response to treatments and novel immune targets of cancer in preparation for developing immune-based combination therapeutic strategies.
Our Vaccine and Cell Therapy Laboratory, built in accordance with current Good Manufacturing Practice as required by the U.S. Food and Drug Administration, supports the development and manufacturing of novel vaccines and cell-based therapies for cancer and infectious diseases. Plans call for the Laboratory to create personalized genome vaccines modeled from mutations that are specific to the malignant cells within the patient’s tumor.
Innovation and transformation energize our programs and reflect The Tisch Cancer Institute’s world-class research. In this Director’s Report, we introduce you to promising young investigators and those with decades of experience behind them, who are breaking down the complexities of cancer in order to reach our goal of improving as many of our patients’ lives as possible. This, after all, is the true measure of our success.
The Tisch Cancer Institute
Download the Winter 2017 Specialty Report