Mount Sinai Heart
Collaboration, dedication, expertise, and innovation sum up our work at Mount Sinai Heart. Our goal is to help transform the way we practice cardiovascular medicine to improve the health of our patients.
Mount Sinai Heart
Mount Sinai Heart is among the world's leading centers for cardiovascular medicine and advanced diagnostic and therapeutic technologies, successfully treating cardiac conditions from chronic arrhythmias to sudden heart attacks.
This specialty report highlights:
- A clinical trial of a new chemical treatment that extends the life of biological heart valves made from bovine pericardial tissue that is implanted in humans
- A groundbreaking trial testing the efficacy of a new ventricular assist device (VAD) intended to provide hemodynamic support in patients with advanced refractory left ventricular heart failure
- Research into the technological advancement of echocardiology through robotics, machine intelligence and mobile devices
- A first-in-class small-molecule drug that may open a molecular pathway in the treatment of heart failure
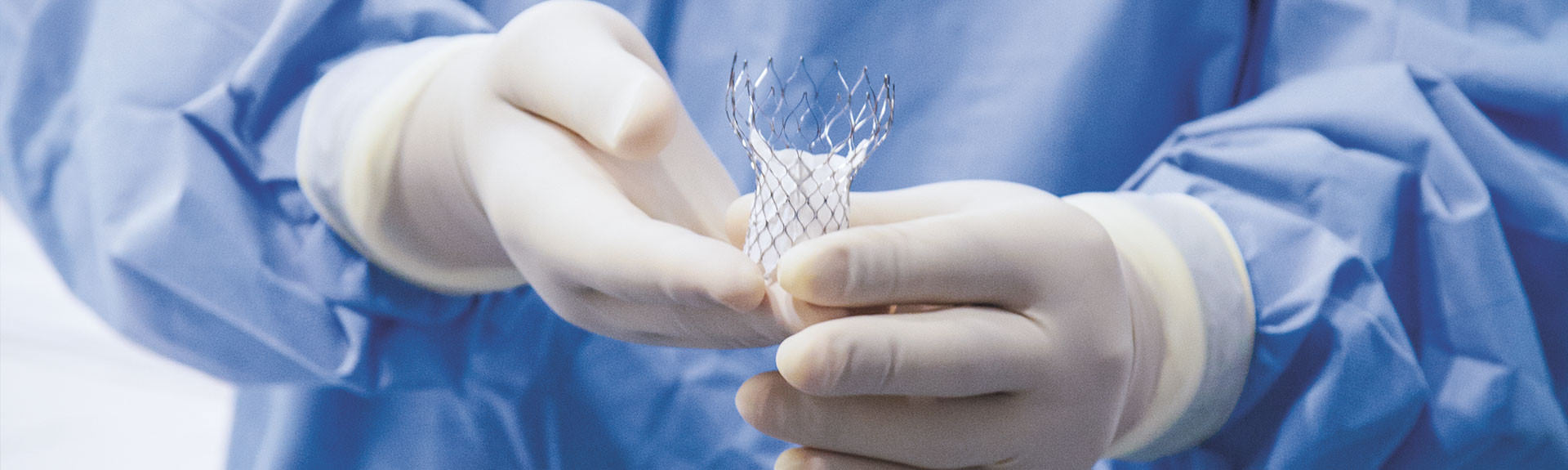
Transcatheter Aortic Valve Replacement Yields Promising Results
The Mount Sinai Hospital’s Dr. Samin K. Sharma Family Foundation Cardiac Catheterization Laboratory in 2015 marked the five-year anniversary of performing the first transcatheter aortic valve replacement (TAVR) with a self-expanding device (called the CoreValve® System) in the United States.

Since then, the Catheterization Laboratory has treated 550 patients with severe aortic stenosis using this minimally invasive technique, achieving a very low stroke complication rate of one percent compared with five to six percent nationally, according to Samin K. Sharma, MD, Director of Clinical and Interventional Cardiology at the Icahn School of Medicine at Mount Sinai.
“More and more patients with aortic stenosis can now be treated safely without surgery and its potential complications,” says Dr. Sharma, who is also the Zena and Michael A. Wiener Professor of Medicine. “Once the valve is implanted, there is a very low chance of degeneration. It’s easier, potentially more durable, and longer lasting.”
The five-year data for Dr. Sharma’s patient, Richard Melosh, 92, the first person in the United States to receive TAVR with the CoreValve System
via percutaneous implantation, have been promising. When Mr. Melosh underwent the procedure in 2010, he was a high-risk patient with lung disease, as well as other comorbid conditions.
The retired trucking company executive from New Jersey says he agreed to undergo the procedure because he was not a candidate for surgery and “was beginning to run out of gas.”
Today, Mr. Melosh reports, “I am doing pretty well for a 92-year-old, and this treatment helped me a lot, no question about it. I had a very smooth recovery, thanks to the excellent care I received from Dr. Sharma and his wonderful team, and the support of my loving family.”
Initially, TAVR was used for patients who were not candidates for surgical aortic valve replacement due to serious comorbidities or excessive calcification of the aortic valve. Today, Dr. Sharma would like to expand its use. He is currently involved in a multicenter clinical trial that is testing whether the innovative treatment can be used for all patients with severe aortic stenosis, including those at intermediate or low risk for surgery. Thirty patients at Mount Sinai Heart are enrolled in the Surgical Replacement and Transcatheter Aortic Valve Implantation (SURTAVI) Trial, which is expected to be completed in early 2016.
“Patients will be able to walk better and go home early,” says Annapoorna Kini, MD, Director of the Dr. Samin K. Sharma Family Foundation Cardiac Catheterization Laboratory at The Mount Sinai Hospital, and Professor of Medicine (Cardiology) at the Icahn School of Medicine at Mount Sinai. “This will make it more convenient for the patient, who could be discharged two days after valve replacement. That’s a huge improvement.” The average hospital stay for patients undergoing surgical aortic valve replacement is five days.
The Mount Sinai Hospital continues to offer the Edwards SAPIEN Valve as a second TAVR treatment. It became the first hospital in the New York metropolitan area to offer both TAVR options in 2013.
For more information about Mount Sinai’s SURTAVI trial, please call Melissa Mattimore, ACNP-BC, at 212-241-6431.
Extending the Life of Biological Heart Valves
The efficacy and safety of a new chemical treatment that extends the life of biological heart valves made from bovine pericardial tissue that is implanted in humans is being evaluated in a clinical trial at the Mount Sinai Health System, led by John D. Puskas, MD, Director of Surgical Coronary Revascularization, and Chair of Cardiothoracic Surgery at Mount Sinai Beth Israel.
As the international principal investigator responsible for academic conduct and oversight of the multicenter trial, Dr. Puskas says, “The trial could change the way we think about valve replacement worldwide.”

At its core, the trial tests whether a proprietary chemical treatment applied to the bovine pericardial tissue can help prevent calcification and extend the life of biological aortic and mitral valves. The treatment also allows the valves to be dry while being packaged and sterilized, so that they do not require rinsing before implantation. Heart valves derived from cow hearts typically become calcified after approximately 15 years, leading to structural valve degeneration.
In 2015, 684 patients completed the first phase of the trial, sponsored by Edwards Lifesciences, which examined the use of the chemical treatment in the aortic valve. Now the researchers are completing a second phase of the trial, which is testing the use of the novel chemical preservation process in mitral valves. According to Dr. Puskas, all of the participants will receive valves with the chemical treatment. He expects this phase to be completed in 2017.
“Edwards Lifesciences already makes the most popular biological valve. This technology could make it last longer than any previous biological valve. That would be a new, higher standard of care,” Dr. Puskas says.
To enroll a patient in the trial, please contact Stella Palencia at spalencia@chpnet.org.
The Watchman Device: An Important Paradigm Shift in Nonvalvular Atrial Fibrillation
Since leading the first cardiology team in the eastern United States to implant the Watchman™ device in patients with atrial fibrillation in March 2015, Vivek Reddy, MD, Director of Cardiac Arrhythmia Services—along with Srinivas Dukkipati, MD, and Jacob Koruth, MD, Assistant Professors of Medicine (Cardiology)—has performed 80 procedures at The Mount Sinai Hospital.
The Watchman device provides an effective nonpharmacologic alternative for patients with nonvalvular atrial fibrillation (AF) who would otherwise take warfarin (Coumadin) or other anticoagulants to minimize their risk for stroke and systemic embolism. It is designed to close off the left atrial appendage, a blind pouch in the left atrium where stroke-causing blood clots frequently form. Eliminating this structure from the circulation enables patients to stop taking their anticoagulants.
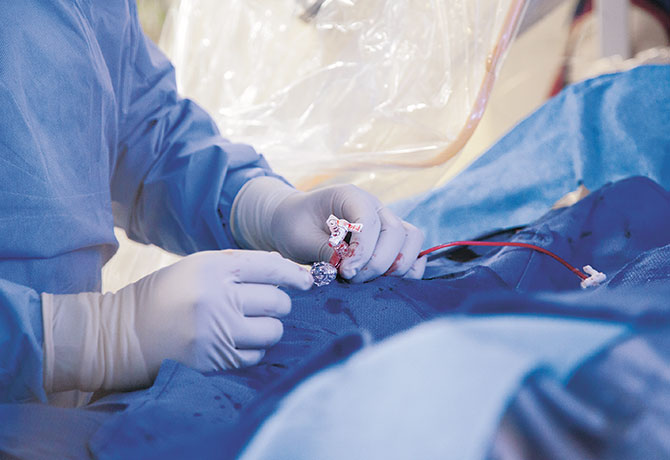
“This is an important paradigm shift in how we think about preventing stroke in AF patients,” says Dr. Reddy. “There are patients for whom long-term blood thinners are not a great option. Watchman successfully eliminates their lifelong need for these blood thinners.”
Eliminating anticoagulant medication is particularly helpful for patients who are at risk for or experience adverse events related to anticoagulation, such as frequent or dangerous bleeding. Elderly patients, particularly those over age 75 who are deemed to be at high risk for falls or need protection from stroke, can benefit from this procedure.
 The device—approved for use by the U.S. Food and Drug Administration in March 2015—is similar in size to a quarter and shaped like a parachute. It is implanted, under anesthesia, into the patient’s heart using a catheter inserted through a vein in the leg. Recovery takes roughly 24 hours, and patients typically spend a night in the hospital.
The device—approved for use by the U.S. Food and Drug Administration in March 2015—is similar in size to a quarter and shaped like a parachute. It is implanted, under anesthesia, into the patient’s heart using a catheter inserted through a vein in the leg. Recovery takes roughly 24 hours, and patients typically spend a night in the hospital.
Dr. Reddy was co-principal investigator for an international clinical trial of Watchman that comprised 707 patients who had nonvalvular atrial fibrillation and at least one additional stroke risk factor, at 59 hospitals in the United States and Europe. The four-year results, published in the November 19, 2014, issue of the Journal of the American Medical Association, found that Watchman patients had a 60 percent reduction in cardiovascular death and a 34 percent reduction in all-cause death. Ninety-two percent of patients who were successfully implanted with Watchman were able to discontinue warfarin at 45 days, and almost all were off the medication at the one-year mark.
Dr. Reddy has served as a consultant to Boston Scientific, Inc., the manufacturer of Watchman.
Technological Breakthroughs Herald the Future of Echocardiology
Robotics, machine intelligence, and miniaturized and mobile devices, “collectively, have the capacity to dramatically rewrite the playbook of how cardiology is practiced,” says Partho P. Sengupta, MD, Director of Cardiac Ultrasound Research at The Mount Sinai Hospital, who is advancing these areas of echocardiology research.
Dr. Sengupta, also Associate Professor of Medicine (Cardiology) at the Icahn School of Medicine at Mount Sinai, investigates technology that optimizes the efficient and rapid detection of cardiovascular disease in patients when it matters most for successful prevention and intervention.
 As International Liaison and Chair of the New Technology Task Force at the American Society of Echocardiography (ASE), Dr. Sengupta has led the way in the growth of miniaturized pocket echocardiography for remote assessments.
As International Liaison and Chair of the New Technology Task Force at the American Society of Echocardiography (ASE), Dr. Sengupta has led the way in the growth of miniaturized pocket echocardiography for remote assessments.
He pioneered three international projects with ASE, including the 2012 ASE Global: Focus on India project. For two days, sonographers and cardiologists from the United States traveled to a remote corner of India to perform handheld echocardiograms on more than 1,000 people who were attending a meditation gathering. The patients had already been diagnosed with cardiovascular problems. Their scans were uploaded from the devices to a secure cloud, then downloaded and read by 75 volunteer physicians in the United States, Canada, Europe, and Saudi Arabia.
The physicians supplying the readings emailed full reports to on-site Indian cardiologists, and several seriously ill patients were directly admitted to a nearby hospital.
In Internet-based robotics imaging, Dr. Sengupta has been part of several long-distance echocardiography studies. In Germany in 2014, he performed the first robot-assisted transatlantic ultrasound examination of a patient’s carotid artery in Burlington, Massachusetts, using a low-bandwidth Internet connection. The robotic ultrasound exam took four minutes.
Using the same robotic technology in another study, Dr. Sengupta worked with colleagues at a tertiary hospital in Sweden to perform echocardiography on patients at a rural location 135 miles away. The researchers found they could reduce the time patients waited to receive a consultation with a specialist to 12 days from an average of 86 days, and reduce the diagnostic processing time to 27 days from 114 days. In addition, patients were more satisfied with the remote exam, citing the lack of travel time.
“Ultrasound is the most cost-effective diagnostic tool for cardiovascular evaluations,” Dr. Sengupta says. “But there is often a lack of technical expertise for performing it in remote locations, particularly in the developing world. In fact, only five percent of the world’s population has ready access to ultrasound. This limitation can be overcome by experts using robotic arms from distant locations, reducing the time it takes to obtain a diagnosis and improving outcomes for patients.”
In the area of machine intelligence, Dr. Sengupta has been testing big data applications to see if they can help physicians determine whether a patient has early changes in cardiac function, or can help guide the differentiation of patterns of heart disease that can be difficult to diagnose.
Dr. Sengupta has used machine-based clinical analytics to differentiate an athlete’s heart from one that is enlarged due to cardiomyopathies, and to differentiate the grades of functional abnormalities.
“We have to be cautious,” he says, “but it’s exciting that machine-learning and learning intelligence platforms have the potential to achieve this high level of differentiation, exceeding the human capacity in our pilot studies, so far.”
Dr. Sengupta was recognized as a Top 25 professor of diagnostic medical sonography, vascular ultrasound, and echocardiology in 2015, by the online site MedicalTechnologySchools.com.
Molecular Pathway to Stimulate Failing Hearts
Never has the need been greater for novel targets and treatment strategies for congestive heart failure. In the United States alone, nearly six million people suffer from heart failure; more than a million are hospitalized annually; and about half of those who develop the disorder will die within five years of diagnosis.

Against that backdrop, Roger Hajjar, MD, Director of the Cardiovascular Research Center and the Arthur and Janet C. Ross Professor of Medicine at the Icahn School of Medicine at Mount Sinai, and his research team have opened an intriguing molecular pathway into the treatment of heart failure. It involves a first-in-class small-molecule drug that restores the contractile properties of a failing heart. Specifically, Dr. Hajjar and his team, which includes Chang Won Kho, PhD, and Ahyoung Lee, PhD, discovered that a ubiquitin-like protein known as SUMO1 stimulates the activity of SERCA2a, another protein responsible for the pumping action of the heart muscle. In the models of heart failure they developed, the researchers found reduced levels of both SUMO1 and SERCA2a. Their efforts are focused on the most severe cases of heart failure: patients with few options other than heart transplants or ventricular assist devices.
“We’re quite excited about the discovery of this pathway,” acknowledges Dr. Hajjar, widely known for his extensive work in developing treatments for heart failure using gene therapy. “We now have a small molecule for activating SUMO1 in heart failure, and we’re prepared to go forward with preclinical and, eventually, clinical testing.” That candidate, he adds, could also figure in the pursuit of other cardiovascular-disease targets, including pulmonary hypertension, diastolic dysfunction, and perhaps neurovascular disorders.
In a field where research failures are common, the results so far for SUMO1 activators have been encouraging. In early-stage animal trials, the lead compound N106 induced a 25 to 30 percent increase in heart contractility and improved survival rates with ongoing treatment. This technology represents a significant step beyond beta blockers and ACE inhibitors, the current standard of care.
“It’s been an amazing journey for us thanks to all the resources Mount Sinai has put at our disposal,” Dr. Hajjar emphasizes. “It feels as though we’re part of a small pharmaceutical company with access to an incredible range of in-house experts.” Dr. Hajjar’s team also includes Robert DeVita, PhD, Director of Medicinal Chemistry for the Experimental Therapeutics Institute, and Roberto Sanchez, PhD, Associate Professor of Structural and Chemical Biology.
In keeping with its mission to ensure that its discoveries and innovations are translated into health care products and services, Mount Sinai has produced cutting-edge work in a wide range of laboratories. These advances include:
- The discovery of novel small molecules for treating lysosomal storage diseases (LSDs), a group of conditions caused by the most common genetic disorder in humans. In preclinical trials, these compounds have been shown to reverse the toxic lipid buildup in patient cells, which can severely damage tissue and organs.
- Development of a family of noninvasive assays, called FractalDx, to monitor kidney function both before and after transplantation to predict the likelihood of fibrosis and graft loss in transplant recipients.
- A new therapeutic approach to brain tumors that delivers autologous stem cells to the disease site at the time of tumor resection, bypassing the blood-brain barrier. These stem cells have been encoded with a tumor-cell-killer gene which is activated once the tumor returns, which is 95 percent of the time.
Message from the Director: Valentin Fuster, MD, PhD
An improved transcatheter aortic valve suitable for all patients with severe aortic stenosis; an effective, nonpharmacologic alternative for people with nonvalvular atrial fibrillation; and a new ventricular assist device to support patients with advanced, refractory left ventricular heart failure—these are some of the latest advances at Mount Sinai Heart.
 This 2016 issue of the Heart Report also includes our evaluation of a new chemical treatment that could extend the life of heart valves derived from bovine pericardial tissue that is implanted in humans.
This 2016 issue of the Heart Report also includes our evaluation of a new chemical treatment that could extend the life of heart valves derived from bovine pericardial tissue that is implanted in humans.
Collaboration, dedication, expertise, and innovation sum up our work at Mount Sinai Heart. Our goal is to help transform the way we practice cardiovascular medicine to improve the health of our patients.
To that end, I am pleased to share news about the New York State Department of Health record that we recently set for percutaneous coronary interventions (PCI). The Mount Sinai Hospital in 2012 became the only hospital in the state ever to be awarded a two-star safety rating for PCI in two categories in a single year, for mortality rates in all cases and in nonemergency cases that were significantly below the state average. Mount Sinai consistently has the largest number of PCI cases in New York State.
Under David H. Adams, MD, Marie-Josée and Henry R. Kravis Professor and Chair of the Department of Cardiothoracic Surgery, Mount Sinai has set national benchmarks with greater than 99 percent degenerative mitral valve repair rates. Dr. Adams is co-inventor of two mitral valve annuloplasty repair rings and inventor of a tricuspid annuloplasty ring.
To promote good health, we have launched a new project in Harlem involving 600 preschoolers who attend Head Start programs and their parents or caregivers. The program will use playful methods to teach children the importance of healthy eating, maintaining physical activity, and understanding emotions. The parents and caregivers will receive health-related family activities to reinforce the connection between home and school. If the project is successful, it will be expanded throughout New York City’s preschools and possibly serve as a national model.