Dean’s Quarterly
The Icahn School of Medicine at Mount Sinai is a national and international leader in biomedical education, research, and patient care.
Dean’s Quarterly
The Dean's Quarterly report features the latest news and research from the Icahn School of Medicine at Mount Sinai.
This report highlights:
- A message from the Dean
- Mount Sinai will help the U.S. military identify exposure to weaponized infectious agents
- Cells associated with treatment-resistant Crohn’s disease are identified for the first time
- Manish Arora, PhD, MPH, receives award for outstanding research
- New treatment for treatment-resistant depression
- The link between calories and inflammation
- Mount Sinai and Sema4 launch five-year asthma study with pharmaceutical giant Sanofi
- Innovative program to give students debt relief
- FDA grants breakthrough device designation to KidneyIntelX™
- Two therapies for treatment-resistant ulcerative colitis
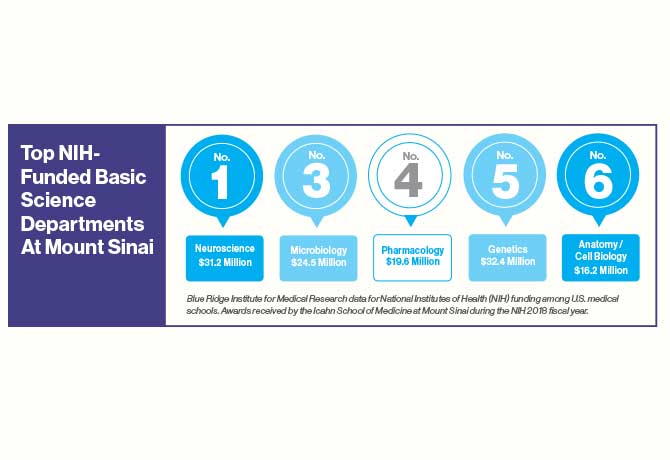
- Top NIH-funded basic science departments at Mount Sinai
Message from the Dean
When the Icahn School of Medicine at Mount Sinai built a supercomputer seven years ago, we became one of the first academic medical centers in the nation to begin harnessing big data in our own facility. Using $4 million in high-end equipment awards from the National Institutes of Health and our own Mount Sinai investments, we have upgraded our supercomputing capabilities several times since then. With more than 1 petaflop of computing power and more than 20 petabytes of storage, our supercomputer has enabled us to generate more than $100 million per year in research resulting in more than 800 peer-reviewed publications.

Today, we are expanding our use of big data by further translating artificial intelligence (AI) into our precision imaging, digital pathology, and genomic health centers. We are launching a clinical genomic screening program for preventive health that can be accessed by patients in our primary care settings. At The Mount Sinai Hospital, we are using machine learning to assist us in identifying patients at risk for cardiopulmonary deterioration, malnutrition, and even falling, so they can benefit from earlier interventions. We expect AI to enhance our medical and scientific discoveries, improve clinical trials, and allow us to bring treatments to market more quickly and efficiently.
Thanks to a generous donation by Mount Sinai trustee Hamilton Evans "Tony" James and his wife, Amabel James, we have announced plans to open a world-class center for AI in 2021. The Hamilton and Amabel James Center for Artificial Intelligence and Human Health will be located on the Icahn School of Medicine campus and will serve as the focal point for high-tech innovation in health care.
In addition, we have established the Hasso Plattner Institute (HPI) for Digital Health at Mount Sinai, with a $15 million gift from the Hasso Plattner Foundation, based in Potsdam, Germany. HPI is recognized for its highly ranked undergraduate and graduate-level computer science programs. Our collaboration will enable us to create digital health solutions for consumers, patients, providers, and health systems and will be housed in our new AI center.
We anticipated the future of machine learning while preparing for our latest capital campaign more than two and a half years ago. After soliciting input from 100 Mount Sinai faculty members and 100 external advisors about where the field of medicine was heading, we created a road map to guide us over the next decade. Today, we are in the midst of carrying out that plan.
This is a watershed moment in health care that calls for experimentation and exploration. Over the next decade, the amount of health data being generated worldwide—from electronic health records, genetic testing, and mobile sensor technology—will make today’s data stream seem small by comparison. Augmented or artificial intelligence can help us harness that data and turn it into knowledge.
Mount Sinai Will Help the U.S. Military Identify Exposure to Weaponized Infectious Agents

One of the greatest challenges facing the U.S. military is being able to determine, definitively, whether troops serving in combat zones have been exposed to weaponized infectious agents, chemicals, or radiation, or if suspects have been working with materials used in making weapons of mass destruction. This knowledge would enable the military to respond immediately.
The Icahn School of Medicine at Mount Sinai is now taking an active role in that national security initiative, through a $27.8 million contract that was awarded in June by the Defense Advanced Research Projects Agency (DARPA)—an arm of the U.S. Department of Defense. Under the four-year contract, Mount Sinai researchers and colleagues from industry and other academic institutions will work together to explore new methods of finding molecular signatures in blood that can identify exposures and the time of exposure. They will also develop field-deployable instruments that can perform these sensitive forensic and diagnostic analyses.

“The human body logs exposures in a rich biographical record that we carry around with us in our epigenomes,” says Stuart Sealfon, MD, Professor of Neurology at the Icahn School of Medicine at Mount Sinai, and Principal Investigator on the contract, which is part of DARPA’s new Epigenetic Characterization and Observation (ECHO) program. “The ECHO technology we’re developing will enable us to quickly read someone’s epigenome from a small amount of blood and measure any changes in the cells to accurately predict exposure to hazardous agents or materials.”
Current forensic and diagnostic screening methods require large instruments and are unable to detect previous exposure, according to Dr. Sealfon. The researchers will focus on creating advanced microfluidic instrumentation with a reduced footprint that can be easily deployed and moved around battlefields for the sequencing and analysis of human cells. “This level of portability could enhance the military’s ability to conduct timely surveillance of emerging threats around the world where U.S. troops or our allies are actively engaged,” he says. It could also make a difference by ensuring that medical countermeasures are undertaken when soldiers encounter hazardous nerve agents, such as sarin and VX, or other toxic agents, such as phosgene and chlorine.
Mount Sinai will draw on its considerable experience in the fields of genomics, proteomics, and epigenomics over the course of the research project. In order to detect epigenetic markers in small amounts of blood, the researchers will also leverage their strengths in the rapidly unfolding science of single-cell biology. “My laboratory is an integrated computational and experimental group with cell biologists, molecular biologists, computer scientists, bioinformaticists, physicists, mathematic modelers, and database developers,” Dr. Sealfon says.
“We are also fortunate to have outstanding collaborators within Mount Sinai—including Robert Sebra, PhD, Associate Professor, Department of Genetics and Genomic Sciences, and his lab, and from outside laboratories, since that gives us a broader range of expertise.”
Defense security is only one aspect of the project funded by DARPA. Just as important, according to Dr. Sealfon, is the potential application of ECHO technology in general medicine. “It could be valuable in the field of infectious disease, for example, to quickly and reliably predict if someone has a bacterial or viral infection during the influenza season, giving patients a point-of-care benefit,” he says. Another possible outgrowth of the DARPA work is the development of basic research instruments that could generate epigenetic data on single cells within minutes.
“It’s likely that medical applications from this research program will be realized in a shorter time frame than those on the military side, which are more demanding,” Dr. Sealfon says. “This may result, for example, in the next-generation sequencing technology making many of the genomic analyses we now do much faster, easier, and more accessible.”
Cells Associated with Treatment-Resistant Crohn’s Disease are Identified for the First Time
Precise cell types that correlate with a patient’s resistance to the standard therapy for Crohn’s disease—anti-inflammatory drugs called TNF inhibitors—have been identified for the first time by researchers at the Icahn School of Medicine at Mount Sinai and published in the August 29, 2019, issue of Cell. TNF inhibitors are used in Crohn’s disease to stop inflammation, but as many as 30 percent of patients do not respond to this treatment and end up requiring surgical intervention within 10 years after diagnosis. The new discovery could open the door to identifying biomarkers and tailoring better therapeutic options for these patients.
Mount Sinai researchers used single-cell RNA sequencing and CyTOF technology to examine inflamed and noninflamed small intestine tissue samples as soon as they were removed from Crohn’s disease patients. Looking at the lesions in real time on a single-cell level, the investigators identified the immune cells and the circulating blood cells and their interactions, and mapped a landscape of thousands of cells in the lesion.
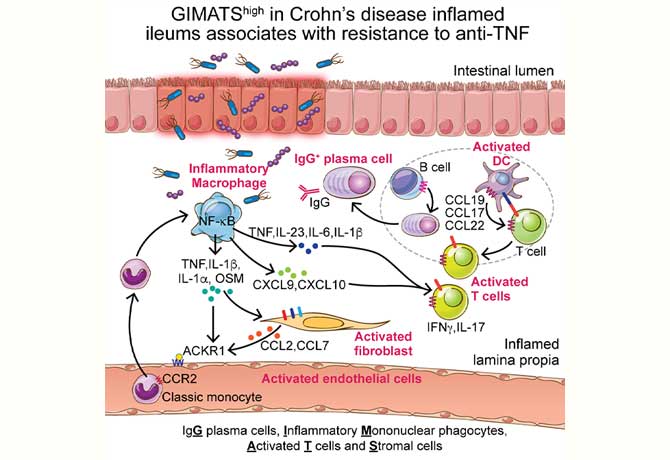
“Single-cell profiling provides unprecedented information on the make-up of the disease,” says co-corresponding author Miriam Merad, MD, PhD, Director of the Precision Immunology Institute and the Human Immune Monitoring Center at the Icahn School of Medicine at Mount Sinai. “This type of analysis will help us understand why patients respond to or resist specific treatment and what else we could be targeting.”
Co-corresponding author Judy H. Cho, MD, Director of The Charles Bronfman Institute for Personalized Medicine, and Ward-Coleman Professor of Translational Genetics and Medicine at the Icahn School of Medicine at Mount Sinai, says, “We designed this study in a way that defines inflammation with unprecedented precision using immunology and computational biology to get a better understanding of this disease.”
According to computational biologist Ephraim Kenigsberg, PhD, Assistant Professor of Genetics and Genomic Sciences at the Icahn School of Medicine at Mount Sinai, and co-corresponding author of the study, “Single-cell analysis revealed different cellular signatures, and when we integrated this with larger data sets, including clinical trials, we were able to make our findings clinically relevant.”
Manish Arora, PhD, MPH, Receives Award for Outstanding Research
Manish Arora, PhD, MPH, an innovative researcher whose work is based on the dynamic interface between the environment and human physiology over a person’s lifetime, recently received the prestigious Outstanding Investigator Award from the National Institute of Environmental Health Sciences (NIEHS). Dr. Arora, the Edith J. Baerwald Professor and Vice Chair of the Department of Environmental Medicine and Public Health at the Icahn School of Medicine at Mount Sinai, will receive $8 million over eight years to complete research into his theory, known as the Biodynamic Interface.
The award is part of the NIEHS’s Revolutionizing Innovative, Visionary Environmental Health Research (RIVER) program, which supports outstanding investigators rather than specific projects and gives the scientists intellectual and administrative freedom.
Dr. Arora plans to use the award to advance his research into developing predictive models that may identify and prevent diseases such as autism, schizophrenia, and amyotrophic lateral sclerosis or ALS, decades before any clinical signs are apparent.
“Understanding the environmental factors in autism, schizophrenia, and ALS could identify specific pathways related to the disease’s pathology and could lead to an early warning system for these and other neurodevelopmental disorders,” he says. “The earlier these diseases are diagnosed or predicted, the earlier people can take advantage of therapies.”
Dr. Arora is known for his use of biomarkers that utilize human baby and permanent teeth to reconstruct the timing of exposure to various harmful chemicals and essential nutrients, and the biological response to those environmental factors. He developed the method of using teeth to reconstruct prenatal and early life environmental exposures and their effects on health, disease, and development.

In a June 28, 2019, study in Cancer Letters, co-author Dr. Arora and first author Lauren M. Petrick, PhD, Assistant Professor, Environmental Medicine and Public Health, were among a team of researchers who found that an abundance of certain fatty acids in newborns was associated with increased risk for acute lymphoblastic leukemia, the most common form of childhood cancer.
The study, using prediagnostic blood collected at birth, found that these lipids were greater in infants who were fed formula rather than colostrum and breast milk and increased with the mother’s prepregnancy body mass index. The study’s authors also included faculty from the University of California, Berkeley; and Karolinska Institute, Stockholm, Sweden.
Changing the Paradigm for Patients with Treatment-Resistant Depression
The U.S. Food and Drug Administration in March, approved SPRAVATO™ (esketamine) CIII nasal spray for the treatment of treatment-resistant depression. Dennis S. Charney, MD, Anne and Joel Ehrenkranz Dean, Icahn School of Medicine at Mount Sinai, is a co-inventor of a method of treatment, which is patented and part of the drug application for SPRAVATO, a product of the Janssen Pharmaceutical Companies of Johnson & Johnson.
“As a researcher, you strive to come up with new treatments for the patient, especially in terms of finding answers to the most debilitating diseases,” says Dr. Charney, who is also President for Academic Affairs, Mount Sinai Health System. “To know that you oversaw the early development of an approach that can make a difference in the lives of countless individuals is extremely rewarding.”
Esketamine represents the first new mechanism of action in decades to treat major depressive disorder. Delivered in the form of a nasal spray, esketamine works differently than the three classes of antidepressants that are currently on the market. The drug works on the N-methyl-D-aspartate receptor, an ionotropic glutamate receptor in the brain. In contrast, widely used antidepressants target different neurotransmitters—serotonin, serotonin and norepinephrine, and norepinephrine and dopamine—and can take weeks, or even months, to work.
An estimated 30 percent of people who are treated for depression do not respond to therapies. Treatment-resistant depression (TRD) is a devastating disease that is associated with greater morbidity, higher health care costs, and various comorbid conditions.
“We commend Dr. Charney and his colleagues for their work in changing the paradigm for patients with treatment-resistant depression,” says Kenneth L. Davis, MD, President and Chief Executive Officer of the Mount Sinai Health System. “Through his commitment to innovation and science, Dr. Charney has inspired countless researchers to leverage new technologies and create discoveries to benefit the lives of patients around the world—while at the same time leading the Icahn School of Medicine to unparalleled growth and high national rankings.”
In his role as Dean of the Icahn School of Medicine, Dr. Charney says that many younger researchers look to him for advice. “I tell them that in science you’ve got to keep trying, to keep pushing,” he says. “There is a lot to be gained by working in a small group that is engaged in the give and take of scientific interaction. Researchers should understand that failure is going to happen, but they can use that failure as a stepping stone to discovery.”
Conflicts of Interest Disclosure: Dr. Charney is named as co-inventor on patents filed by the Icahn School of Medicine at Mount Sinai (ISMMS) relating to the treatment for treatment-resistant depression, suicidal ideation, and other disorders. ISMMS has entered into a licensing agreement with Janssen Pharmaceuticals, Inc., and it has and will receive payments from Janssen under the license agreement related to these patents for the treatment of treatment-resistant depression and suicidal ideation under this agreement. Consistent with the ISMMS Faculty Handbook, Dr. Charney is entitled to a portion of the payments received by the ISMMS. Since SPRAVATO has received regulatory approval for treatment-resistant depression, ISMMS and thus, through the ISMMS, Dr. Charney will be entitled to additional payments, beyond those already received, under the license agreement.
Study Directly Ties Caloric Intake to Inflammation
Researchers at the Icahn School of Medicine at Mount Sinai, using sophisticated, single-cell technology, have isolated the molecular underpinning that explains why short-term fasting and a low-calorie diet are beneficial to people with inflammatory and autoimmune diseases, such as atherosclerosis and rheumatoid arthritis. Until now, the connection between reduced caloric intake and improved health has been a widely held but poorly understood hypothesis that has captured the public’s attention as high-profile celebrities attest to the success of frequent fasting.
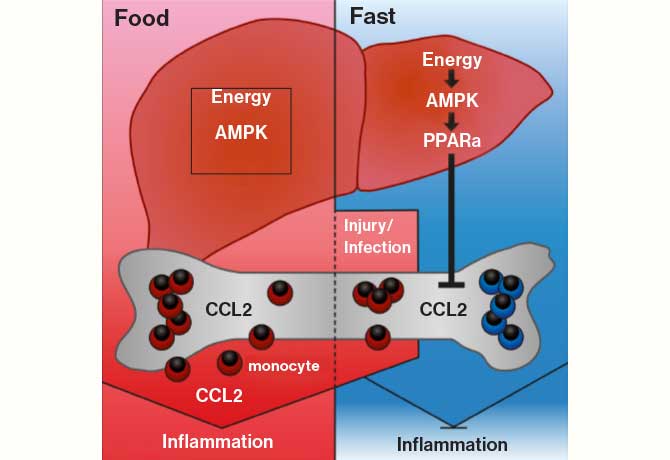
In the new study, published in the August 22, 2019, issue of Cell, the researchers, led by Miriam Merad, MD, PhD, Director of the Precision Immunology Institute at the Icahn School of Medicine at Mount Sinai, were able to describe how dietary intake actually controls the quality and quantity of monocytes—a particular group of white blood cells.
The scientists found a direct link between the amount of glucose and protein in the body and the number of circulating monocytes. These white blood cells, likened to military “reserve forces,” respond to inflammatory stimuli and also play a major role in metabolic equilibrium, growth, and other processes. When digestible carbohydrates, protein, and fat were removed from the diet, the investigators found the pool of circulating monocytes decreased. Importantly, the size of the monocyte pool that was circulating in the blood depended upon the amount of carbohydrates ingested. The study profiled 12 healthy, normal-weight volunteers 3 hours after eating and 19 hours after fasting, as well as mice that had fasted for 4 hours.

“Monocytes are highly inflammatory immune cells that can cause serious tissue damage,” says Dr. Merad. “We have seen more of these monocytes in blood circulation as Western-style eating habits have been adopted by increasing numbers of people around the world. Now that we have a better understanding of what is driving this incidence, we can work to treat it more effectively.”
Stefan Jordan, PhD, the study’s first author and a postdoctoral fellow in the Department of Oncological Sciences at the Icahn School of Medicine at Mount Sinai, says, “There is enormous potential in investigating the anti-inflammatory effects of fasting, considering the broad spectrum of diseases that are caused by chronic inflammation. Our discoveries could lay the groundwork for novel treatments in the future.”
Indeed, chronic diseases such as type 2 diabetes, metabolic syndrome, and cardiovascular disease are believed to be mostly caused by chronic inflammation. According to a study by the RAND Corporation, 60 percent of Americans had at least one chronic condition and 42 percent had multiple chronic conditions as of 2014, the last year for which this data was available. In 2018, the Milken Institute released a report that stated the most common chronic diseases cost the U.S. economy more than $1 trillion annually.
Mount Sinai’s study found that short-term fasting did not compromise the body’s ability to protect itself from acute infections or repair tissue, whereas starvation for 48 hours prior to infection did compromise the body’s antimicrobial immunity and ability to repair tissue.
Another finding by Dr. Merad and her team was that by activating a key cellular pathway in the liver, AMPK, they were able to regulate the number of monocytes regardless of caloric intake. “Targeting liver energy sensors could be an innovative strategy for the prevention and treatment of chronic inflammatory and autoimmune diseases,” says Dr. Merad.
Mount Sinai and Sema4 Launch Five-Year Asthma Study with Pharmaceutical Giant Sanofi
Asthma, a chronic disease of the airways of the lungs, is a growing public health problem that now affects 350 million people and results in about 400,000 deaths worldwide each year. Its diagnosis and treatment remain challenging, however, and debilitating symptoms, such as coughing and shortness of breath, are a major cause behind rising health care costs, missed school for children, and loss of productivity and early disability in adults.
Recently, the Mount Sinai Health System and Sema4—a patient-centered predictive health company and a venture of Mount Sinai—joined with Sanofi, one of the world’s largest pharmaceutical companies, to follow 1,200 Mount Sinai patients to gain unprecedented insights into the biological mechanisms and environmental factors implicated in this condition.
The five-year study—the first of its kind—will collect traditional clinical data, such as electronic medical records and clinical samples, including blood samples and nasal brushings, from patients during their doctor appointments.
The data will be analyzed for genomic and transcriptomic information and combined with other data collected using the patient’s mobile phone—environmental data, like air quality and pollen counts, data from the patient’s asthma inhaler, and data from home monitoring of activity and sleep. One of the unique elements of this study is that the research will be incorporated into actual clinical practice, and real-world data using remote devices will be integrated with molecular data.
“Despite advances in recent years, we still see many patients struggling with asthma, so there is a tremendous need for innovation to reduce the burden of this disease,” says Linda Rogers, MD, Associate Professor of Medicine (Pulmonary, Critical Care and Sleep Medicine) and Clinical Director of the Adult Asthma Program at the Mount Sinai – National Jewish Health Respiratory Institute. Dr. Rogers is the clinical principal investigator of the study, which is a collaboration among the Respiratory Institute, the Icahn Institute of Genomics and Multiscale Biology, Sema4, and Sanofi.
The Respiratory Institute is uniquely positioned to undertake this research. In addition to the large number of asthma patients that the program treats, the Mount Sinai and Sema4 study team have unparalleled capabilities in specimen analysis, data science, and multiscale biological modeling, allowing researchers to gather large amounts of data more rapidly than using more traditional research methods.
Clinical research teams will deploy advanced analytics on this information to better understand how the disease functions, including what triggers asthma attacks and which patient segments are most likely to respond to certain therapies. “This collection of large amounts of multiple types of data is needed to fully understand asthma—a condition researchers now believe is far more complex than was previously understood—and how best to treat patients,” says Tom Neyarapally, Sema4’s Chief Commercial Officer.
Significantly, gathering and analyzing these kinds of data from patients will demonstrate at the molecular level how their bodies are responding to asthma, says Andrew Kasarskis, PhD, Executive Vice President and Chief Data Officer for the Mount Sinai Health System and a co-principal investigator of the study. For example, analysis of a blood sample will show changes in the cellular activity, such as which proteins are being produced, and a nasal swab may reveal important clues about one’s immune response and what is happening in the lungs.
“We will define asthma subtypes clinically, then understand the molecular basis of disease in each subtype in order to discover new therapies and better manage asthma in all our patients,” says Dr. Kasarskis.
Ultimately, adds Erik Lium, PhD, Executive Vice President of Mount Sinai Innovation Partners, “this collaboration may lead to the identification of novel drug targets and the development of groundbreaking therapies to benefit all patients with asthma.”
Innovative Program to Give Students Debt Relief

Medical students with financial need will receive substantial debt relief from a new program at the Icahn School of Medicine at Mount Sinai, which launched during the 2019-2020 academic year. The program, the Enhanced Scholarship Initiative (ESI), enables qualifying students who are enrolled in the medical school for four years to graduate with a maximum total debt of $75,000, compared with the national median of $200,000.
The program is an integral part of the Mount Sinai Health System’s effort to promote equity and diversity in medical education, and well-being among its students. It will enable students to follow their passions in medicine, regardless of their financial circumstances.
The Icahn School of Medicine is one of a small number of medical schools nationwide that have established campaigns to offer debt relief for students. An estimated 40 percent of students attending the School of Medicine—those with “demonstrated need”—will benefit from the program, with aid covering tuition, housing, meals, and other expenses, such as travel and books. Demonstrated need is the difference between the cost of attending the school and the resources that each student and their family have available to pay that cost. Students who qualify will take out a loan of no more than $18,750 per year, funded by a combination of institutional and federal loans.
The rest of these students’ unmet need will be covered by the new scholarship. “We have taken an approach that is both practical and principled,” says David Muller, MD, Dean for Medical Education and the Marietta and Charles C. Morchand Chair in Medical Education at the Icahn School of Medicine at Mount Sinai. “From a practical perspective, we want to cover not just tuition, but all the other costs of education, which can add up to $25,000 - $30,000 a year to a student’s debt over and above tuition. From a principled perspective, we want to channel our resources to students with the greatest need so they can move on to healthy, productive careers.”
It is well established that even the prospect of debt serves as a deterrent to students who are underrepresented in medicine or come from lower-income families, Dr. Muller says. “There are unacceptable disparities among many African American and Latino students whose desire to seek a career in medicine far outstrips their financial wherewithal to do so. This is the kind of statistic we want to change,” says Dr. Muller. He also points out that overwhelming debt has been correlated with an increase in stress, burnout, and depression.
The initiative was made possible by members of the Mount Sinai Boards of Trustees, led by Donald J. Gogel, Chair of the Boards’ Education Committee. “This is a tremendously important moment for our students with financial need and their ability to reduce the debt incurred from attending medical school. For the Mount Sinai Boards of Trustees, this issue is a very important area of focus, almost a moral imperative,” says Mr. Gogel. “We believe that the new scholarships will increase our students’ freedom and flexibility in the choice of where and how they will practice medicine, conduct research, or teach once they graduate.”
Valerie Parkas, MD, Senior Associate Dean of Admissions and Recruitment, Icahn School of Medicine at Mount Sinai, says the Enhanced Scholarship Initiative will help level the playing field for promising students from a variety of backgrounds: “This a bold move that is grounded in our vision, grounded in our mission, and grounded in our commitment to equity.”
FDA Grants Breakthrough Device Designation to KidneyIntelX™
Renalytix AI plc, a developer of artificial intelligence-enabled clinical diagnostics for kidney disease, was recently granted Breakthrough Device designation by the U.S. Food and Drug Administration (FDA) for its lead diagnostic, KidneyIntelX™.
KidneyIntelX™—being developed in close collaboration with the Mount Sinai Health System—is designed to diagnose and improve clinical management of patients with type 2 diabetes with fast-progressing kidney disease. It will use machine learning algorithms to assess the combination of predictive blood-based biomarkers, including sTNFR1, sTNFR2, and KIM1, in combination with electronic health record information to identify progressive kidney disease.
The FDA’s Breakthrough Devices Program is a voluntary program for certain medical devices and device-led combination products that provide for more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
Early detection and intervention is essential in changing the course of kidney disease, which can go undetected until it is very advanced. Chronic kidney disease is a common disorder and has become a major health concern in the United States, affecting approximately 14 percent of adults.
Two Therapies for Treatment-Resistant Ulcerative Colitis
Two studies in the September 26, 2019, issue of The New England Journal of Medicine validate effective therapies for patients with treatment-resistant ulcerative colitis (UC), a chronic inflammatory disease of the large intestine. Both studies were led by Bruce Sands, MD, the Dr. Burrill B. Crohn Professor of Medicine and Chief of the Dr. Henry D. Janowitz Division of Gastroenterology at the Icahn School of Medicine at Mount Sinai.
The results of the first clinical trial validated ustekinumab as a UC therapy. Ustekinumab has been approved for treating patients with Crohn’s disease. In this phase lll clinical trial, Dr. Sands and his team tested more than 900 patients with moderate-to-severe UC who were unable to tolerate or had an inadequate response to tumor necrosis factor inhibitors. The participants were randomized to receive one of two intravenous induction doses of ustekinumab or placebo. Those who responded to the induction dose were rerandomized at week eight, with 175 participants receiving 90 mg of subcutaneous ustekinumab every twelve weeks, 176 receiving the same amount every eight weeks, and the remainder receiving a placebo.
Researchers found that clinical remission rates at week eight were higher among participants who received intravenous ustekinumab than among those who received a placebo. Among participants who achieved a clinical response to ustekinumab, the clinical remission rates at week 44 were significantly higher among the eight-week and twelve-week ustekinumab cohorts than among the placebo-treated cohort. Equally significant, more patients in the ustekinumab cohorts eliminated corticosteroid use at least 90 days before week 44, and 97 percent of patients in clinical remission were corticosteroid-free at week 44.
The second study was the first ever head-to-head comparison of two biologic therapies for inflammatory bowel disease: vedolizumab and adalimumab. In total, 769 participants with moderate-to-severe UC were recruited for this randomized phase 3b study, with 383 patients receiving 300 mg of vedolizumab intravenously at weeks 0, 2, and 6, then every 8 weeks, and with subcutaneous placebo injections; and 386 receiving placebo intravenously and adalimumab subcutaneously (160 mg week one, 80 mg week two, and then 40 mg every two weeks).
Researchers found that patients who received vedolizumab achieved significantly higher week 52 clinical remission rates than patients who received adalimumab (31.3% versus 22.5%) and endoscopic improvement (39.7% versus 27.7%). The remission rates for both therapies were similar among the 20 percent of participants who had previous exposure to TNF inhibitors.
“Overall, the results were somewhat surprising because the consensus has generally been that anti-TNF therapies are the most effective for ulcerative colitis,” Dr. Sands says. “We also saw faster symptomatic improvement among patients receiving vedolizumab. This study demonstrates that, on average, using vedolizumab to interfere with leukocyte trafficking to the intestinal mucosa is a more effective UC treatment than an anti-TNF.”