UCSF QBI and Icahn School of Medicine at Mount Sinai Scientists Report New Preclinical Data Published in Science on Potential Best-in-Class COVID-19 Anti-Viral Treatment: Plitidepsin
- First studies reported on antiviral drug effects against new variant mutant “U.K.” strain of SARS-CoV-2 - Plitidepsin shows potent anti-viral effect in preclinical models of original SARS-CoV-2 as well as the new mutant U.K. strain, and by inhibiting a key host protein, has the potential to have antiviral activity against other future mutations and viruses - Drug targets the biologic pathway prospectively identified and published in Nature months ago from QBI research on SARS CoV-2 which elucidated host proteins and pathways that fuel the survival and virulence of the virus
In a study published online in Science today, scientists at UCSF QBI and the Department of Microbiology at Icahn School of Medicine at Mount Sinai (ISMMS) reported data showing the promise and potential of Aplidin® (plitidepsin), a drug approved by the Australian Regulatory Agency for the treatment of multiple myeloma, against SARS-CoV-2, the virus that causes COVID-19. Specifically, plitidepsin demonstrated antiviral activity that was 27.5-fold more potent against SARS-CoV-2 in vitro than remdesivir, a drug that received FDA emergency use authorization in 2020 for the treatment of COVID-19. In addition, in two preclinical models of COVID-19, plitidepsin showed a 100-fold reduction in viral replication in the lungs and demonstrated an ability to reduce lung inflammation. Furthermore, in a separate publication in bioRxiv, the researchers, in collaboration with Greg Towers and Clare Jolly at University College London in London, England, shared additional in vitro data demonstrating comparable anti-viral activity of plitidepsin in the newly identified b.1.1.7 variant mutant strain of SARS-CoV-2 when compared to the original SARS-CoV-2 strain. Additionally, they found plitidepsin to be ~100 times more potent than remdesivir in human epithelial cells.
The studies were led by the laboratories of Nevan Krogan, Ph.D., Director of the Quantitative Biosciences Institute (QBI) at the School of Pharmacy at UC San Francisco, Senior Investigator at Gladstone Institutes, and Adolfo García-Sastre, Ph.D., Professor in the Department of Microbiology and Director of the Global Health and Emerging Pathogens Institute of ISMMS in New York, among others.
“From the start, our scientific investigation at QBI’s Coronavirus Research Group (QCRG) was to research where and how the SARS-CoV-2 virus was co-opting its human host to thrive, survive and become deadly,” said Dr. Krogan. “That research led us to a biologic pathway, eukaryotic translation machinery, where inhibition of this pathway showed significant antiviral activity in cell culture. One of the promising agents that arose from our screen of drugs that inhibit this pathway was plitidepsin. The preclinical data published today showing increased potency compared to remdesivir, and in conjunction with recent early clinical data showing promise in COVID-19 patients as reported by the drug’s manufacturer, highlight plitidepsin should be further evaluated as a COVID-19 therapy. Additionally, this further validates our prospective and novel approach of looking first at host mechanisms that permit the virus to wreak havoc. By targeting host factors that enable or facilitate disease, we have the potential to have a more far-reaching impact in drug development. In this case, SARS-CoV-2 and its mutant strains, and possibly other viruses that leverage the same pathway may also be susceptible to the same therapeutic agents that inhibit this virus-host interaction.”
The research studies showed that the anti-SARS-CoV-2 activity of plitidepsin was potent in human cells, with an IC90 of 0.88 nM, which was a 27.5-fold more potent antiviral than remdesivir tested in the same cell line. In addition, the antiviral effect of plitidepsin was tested in an established model of human lung cells, which showed that treatment with plitidepsin inhibited SARS-CoV-2 replication with an IC90 of 3.14 nM and a selectivity index of 40.4, suggesting that plitidepsin has potent antiviral activity in primary human lung cells. The researchers also tested the synergy between plitidepsin and remdesivir, given remdesivir’s use as part of the current standard of care for the treatment of COVID-19. In vitro data suggests that plitidepsin has an additive effect with remdesivir and would be a potential candidate to be considered in a combined therapy.
Dr. García-Sastre added, “The ongoing SARS-CoV-2 pandemic has created the immediate need for antiviral therapeutics that can be moved into the clinic urgently. This led us to screen clinically approved drugs with established bioavailability, pharmacokinetics and safety profiles. Our previous study of the SARS-CoV-2 interactome led us to the eEF1A eukaryotic translation pathway as a druggable target with the potential for potent inhibition of SARS-CoV-2. Furthermore, this pathway has been previously described to be an important host factor for the replication of many viral pathogens, including influenza virus and RSV suggesting its panantiviral targeting potential. These studies have only been possible by the combined efforts and expertise of the multiple academic research groups that are co-authors in the published study. We have also been doing these studies in close collaboration with PharmaMar, the company that first isolated the potent eEF1A inhibitor plitidepsin/ Aplidin® from a sea organism.”
“Plitidepsin is an extremely potent inhibitor of SARS-CoV-2, but its most important strength is that it targets a host protein rather than a viral protein,” said Kris White, Ph.D., Assistant Professor in the Department of Microbiology of Icahn School of Medicine at Mount Sinai (ISMMS), who was a corresponding author on the Science paper. “This means that if plitidepsin is successful in the treatment of COVID-19, the SARS-CoV-2 virus will be unable to gain resistance against it through mutation, which is a major concern with the spread of the new U.K. and South African variants.”
About QBI: The Quantitative Biosciences Institute (QBI) is a University of California organized research unit reporting through the UCSF School of Pharmacy. QBI fosters collaborations across the biomedical and the physical sciences, seeking quantitative methods to address pressing problems in biology and biomedicine. Motivated by problems of human disease, QBI is committed to investigating fundamental biological mechanisms, because ultimately solutions to many diseases have been revealed by unexpected discoveries in the basic sciences. Learn more at qbi.ucsf.edu.
About UCSF: The University of California, San Francisco (UCSF) is exclusively focused on the health sciences and is dedicated to promoting health worldwide through advanced biomedical research, graduate-level education in the life sciences and health professions, and excellence in patient care. UCSF Health, which serves as UCSF’s primary academic medical center, includes topranked specialty hospitals and other clinical programs, and has affiliations throughout the Bay Area. Learn more at ucsf.edu or see our Fact Sheet.
About Gladstone Institutes: To ensure our work does the greatest good, Gladstone Institutes focuses on conditions with profound medical, economic, and social impact—unsolved diseases. Gladstone is an independent, nonprofit life science research organization that uses visionary science and technology to overcome disease. It has an academic affiliation with UC San Francisco. Learn more at gladstone.org.
Authorship and funding: This work was funded by grants from the National Institute of Mental Health and the National Institute of Allergy and Infectious Diseases, both part of the National Institutes of Health; the Defense Advanced Research Projects Agency; the Center for Research for Influenza Pathogenesis; the Centers of Excellence for Influenza Research and Surveillance of the National Institute of Allergy and Infectious Diseases; the Centers of Excellence for Integrative Biology of Emerging Infectious Diseases of the Agence Nationale de la Recherche (France); F. Hoffmann-LaRoche AG; Vir Biotechnology, Centre for Integrative Biological Signalling Studies (CIBSS), European Research Council (ERC) and QCRG philanthropic donors. Shokat is a Howard Hughes Medical Institute investigator. A complete list of authors and full funding information is available in the Science paper.
About the Mount Sinai Health System
Mount Sinai Health System is one of the largest academic medical systems in the New York metro area, with more than 47,000 employees working across seven hospitals, more than 400 outpatient practices, more than 600 research and clinical labs, a school of nursing, and leading schools of medicine and graduate education. Mount Sinai advances health for all people, everywhere, by taking on the most complex health care challenges of our time—discovering and applying new scientific learning and knowledge; developing safer, more effective treatments; educating the next generation of medical leaders and innovators; and supporting local communities by delivering high-quality care to all who need it.
Through the integration of its hospitals, labs, and schools, Mount Sinai offers comprehensive health care from conception through geriatrics, leveraging innovative approaches such as artificial intelligence and informatics while keeping patients’ medical and emotional needs at the center of all treatment. The Health System includes more than 6,400 primary and specialty care physicians and 10 free-standing joint-venture centers throughout the five boroughs of New York City, Westchester, Long Island, and Florida. Hospitals within the System are consistently ranked by Newsweek’s® “The World’s Best Smart Hospitals,” “Best in State Hospitals,” “World’s Best Hospitals,” and “Best Specialty Hospitals” and by U.S. News & World Report's® “Best Hospitals” and “Best Children’s Hospitals.” The Mount Sinai Hospital is on the U.S. News & World Report® “Best Hospitals” Honor Roll for 2025-2026.
For more information, visit https://www.mountsinai.org or find Mount Sinai on Facebook, Instagram, LinkedIn, X, and YouTube.
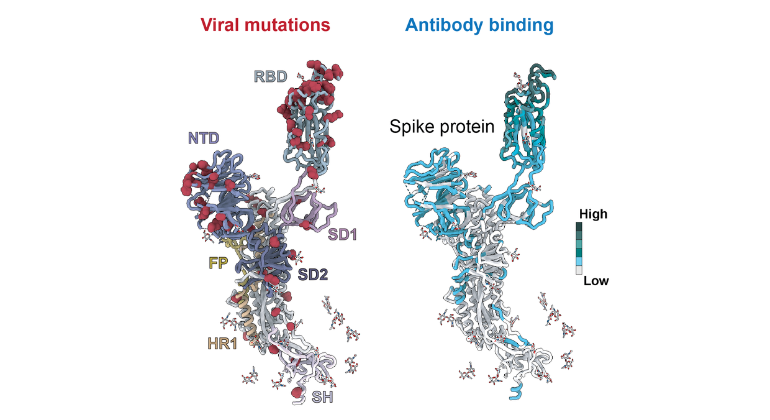
Scientists Uncover How COVID-19 Variants Outsmart the Immune System
Nov 21, 2025 View All Press ReleasesPfizer assures that anticovid pills are also effective against omicron
Jan 18, 2022 View All Press Releases