The Brain Changes Its Rhythm Within Minutes of Therapeutic Stimulation During Deep Brain Stimulation Surgery for Treatment-Resistant Depression
Findings provide a putative physiological biomarker of brain state changes that can predict early antidepressant effects
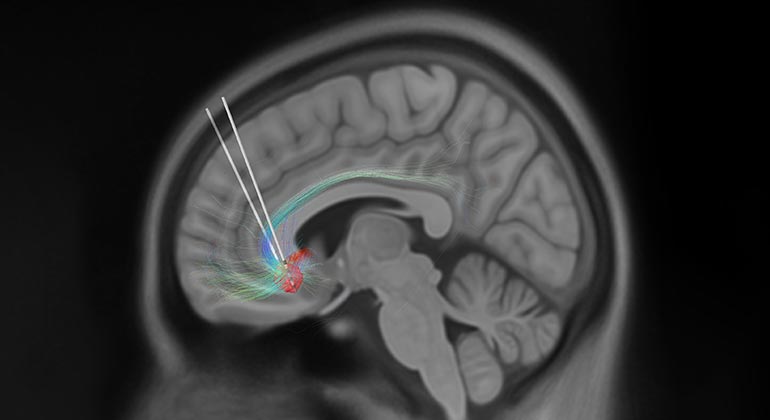
Deep brain stimulation (DBS) has been demonstrated to be an effective treatment for many patients suffering with treatment-resistant depression, but exactly how it works is not known. Scientists worldwide are racing for objective biomarkers of DBS treatment efficacy so that this experimental approach can be optimized, approved and disseminated to those in need. New research published November 3 in Translational Psychiatry, presents new evidence that brief intraoperative exposure to therapeutic stimulation at the time of implantation surgery induces rapid and consistent electrophysiological brain state change – indexed by a decrease in beta power measured at the site of stimulation. These intraoperative brain state changes are seen in individual subjects and are correlated with a significant and sustained decrease in depressive symptoms outside of the operating room without additional stimulation, establishing reduction in beta power as a novel biomarker for DBS treatment optimization.
The data presented are from a collaborative study at Emory University and the Icahn School of Medicine at Mount Sinai. Led by Helen S. Mayberg, MD, Mount Sinai Professor in Neurotherapeutics and Founding Director of the Nash Family Center for Advanced Circuit Therapeutics at Mount Sinai, this research is part of an ongoing National Institutes of Health (NIH) BRAIN Initiative-funded grant.
Deep brain stimulation is approved by the U.S. Food and Drug Administration to treat essential tremor, Parkinson’s disease, epilepsy and obsessive-compulsive disorder. It is a neurosurgical procedure involving placement of a neurostimulator (sometimes referred to as a “brain pacemaker”), which sends high-frequency electrical impulses through implanted electrodes deep in the brain to specific areas responsible for the symptoms of each disorder. While still an experimental treatment, DBS of the subcallosal cingulate (SCC, Area 25), a brain area that has been implicated as playing a major role in depression, has been repeatedly demonstrated as a promising intervention for patients suffering from treatment-resistant depression.
Acute behavioral changes and long-term antidepressant response can be reliably elicited with stimulation of this well-defined, surgically-targeted depression circuit, using individualized neuroimaging guidance. While the clinical effectiveness of DBS over the course of six months of treatment has been repeatedly demonstrated, there are differences in the timeline of recovery across different patients. Understanding the mechanisms of these initial rapid and reproducible behavioral effects and their role in predicting the more critical long-term response trajectories will be key to effective treatment and future study design.
“What we found was that within minutes of stimulation inside the operating room, there was a change in the beta brain rhythm. Patients who showed larger changes then experienced greater relief from their depression in the week after surgery," said Allison C. Waters, PhD, Assistant Professor of Psychiatry, and Neuroscience, at Icahn Mount Sinai and co-first author of the paper. "The beta rhythm is conventionally associated with the brain's determination of whether to stop or keep going with a course of action, which is why neurologists target beta with DBS to treat disorders of movement. We haven't had a clear signal to target with DBS for depression, but now we can speculate as to how the beta signal might function in this context: a release of the brake that generates fatigue and slowness, or interrupting a habitual cycle of negative self-focused thought."
“We were able to leverage machine learning and explainable artificial intelligence approaches to explore unknown (hidden) changes in brain state that would explain the obvious behavioral changes previously observed,” said Mohammad Sendi, MSc, PhD candidate in the Biomedical Engineering Department of Emory University and Georgia Institute of Technology and co-first author of the paper.
Eight treatment-resistant depression patients underwent electrophysiological recording in the operating room during their DBS lead implantation surgeries. Using patient-specific tractography models prior to surgery, investigators identified the “optimal” target within the SCC for lead placement. Stimulation was then delivered in the operating room over the course of an hour while local field potentials (LFPs)—electrical signals between neurons deep in the brain—were simultaneously recorded. A machine learning classification method was subsequently used to discriminate between intracranial LFPs recorded at baseline (stimulation-naïve) and after the first exposure to stimulation inside the operating room. Spectral inputs (theta, 4-8Hz; alpha, 9-121Hz; beta, 13-30Hz) to the model were then evaluated for importance to classifier success and tested as predictors of the antidepressant response. A decline in depression scores by 45.6 percent was observed after one week and this early antidepressant response correlated with a decrease in the SCC LFP beta power, which most contributed to classifier success.
“We generally think of depression treatment as taking weeks to months to show stable and meaningful changes in core clinical features of the illness,” said Dr. Mayberg. “This study shows reproducible and consistent changes in a brain readout over the first minutes of optimized stimulation in the operating room in individual patients. This provides new mechanistic understanding of the 'depression switch' that moves a patient from a state of sustained mental pain and immobility to relief and the renewed capacity to move and engage.”
“This research provides individuals who have struggled with depression a sense of hope through advancements in existing technology,” said John Ngai, PhD, Director of the NIH BRAIN Initiative. “We are making immense strides in better understanding debilitating brain and mental health conditions through these discoveries and look forward to seeing how deep-brain stimulation will continue to improve people’s lives.”
Studies that continue to track these brain state biomarkers during ongoing DBS therapy are ongoing at the Nash Family Center for Advanced Therapeutics at Mount Sinai West in New York. Phase II of this 5-year NIH BRAIN Initiative grant is currently recruiting new subjects.
For more information and/or to be screened for the active study, please contact: Jaimie Gowatsky at jaimie.gowatsky@mssm.edu. Additional information about the active study can be found here.
Dr. Mayberg receives consulting and licensing fees from Abbot Neurostimulation.
About the Nash Family Center for Advanced Circuit Therapeutics at Mount Sinai
The Nash Family Center for Advanced Circuit Therapeutics at Mount Sinai (C-ACT) is a transdisciplinary program co-localized with the Center for Neuromodulation at Mount Sinai West. C-ACT develops and tests new brain-tuning strategies to accelerate the delivery of state-of-the-art individualized surgical treatments for patients with advanced neuropsychiatric disorders—be it those where brain stimulation therapies are already approved for clinical use (e.g., DBS for Parkinson’s disease, epilepsy, OCD and pain) or more experimental applications such as depression, dementia, schizophrenia, eating disorders and addiction. C-ACT brings together neurosurgeons, neurologists, psychiatrists and psychologists with neuroimaging specialists, electrophysiologists, data scientists, engineers and basic neuroscientists to address unmet clinical need through research and to rethink the way neuromodulation treatments are developed, personalized and delivered to patients. Agnostic to classical neurological and psychiatric categories, which are partly arbitrary, C-ACT provides a new model of patient-focused research with the common goal of restoring mood, motor and cognitive function by correcting circuit abnormalities using targeted neuromodulation.
About the Mount Sinai Health System
Mount Sinai Health System is one of the largest academic medical systems in the New York metro area, with 48,000 employees working across seven hospitals, more than 400 outpatient practices, more than 600 research and clinical labs, a school of nursing, and a leading school of medicine and graduate education. Mount Sinai advances health for all people, everywhere, by taking on the most complex health care challenges of our time—discovering and applying new scientific learning and knowledge; developing safer, more effective treatments; educating the next generation of medical leaders and innovators; and supporting local communities by delivering high-quality care to all who need it.
Through the integration of its hospitals, labs, and schools, Mount Sinai offers comprehensive health care solutions from birth through geriatrics, leveraging innovative approaches such as artificial intelligence and informatics while keeping patients’ medical and emotional needs at the center of all treatment. The Health System includes approximately 9,000 primary and specialty care physicians and 11 free-standing joint-venture centers throughout the five boroughs of New York City, Westchester, Long Island, and Florida. Hospitals within the System are consistently ranked by Newsweek’s® “The World’s Best Smart Hospitals, Best in State Hospitals, World Best Hospitals and Best Specialty Hospitals” and by U.S. News & World Report's® “Best Hospitals” and “Best Children’s Hospitals.” The Mount Sinai Hospital is on the U.S. News & World Report® “Best Hospitals” Honor Roll for 2024-2025.
For more information, visit https://www.mountsinai.org or find Mount Sinai on Facebook, Instagram, LinkedIn, X, and YouTube.

Two Leading Mount Sinai Brain Scientists Elected to the National Academy of Sciences
May 05, 2022 View All Press Releases

