Most People Mount a Strong Antibody Response to SARS-CoV-2 That Does Not Decline Rapidly

Florian Krammer, PhD, Professor of Vaccinology at the Icahn School of Medicine at Mount Sinai and senior author of the paper.

Carlos Cordon-Cardo, MD, PhD, Chair of Pathology, Molecular and Cell-Based Medicine and last author of the paper.

Ania Wajnberg, MD, Director of Clinical Antibody Testing at The Mount Sinai Hospital and first author of the paper.
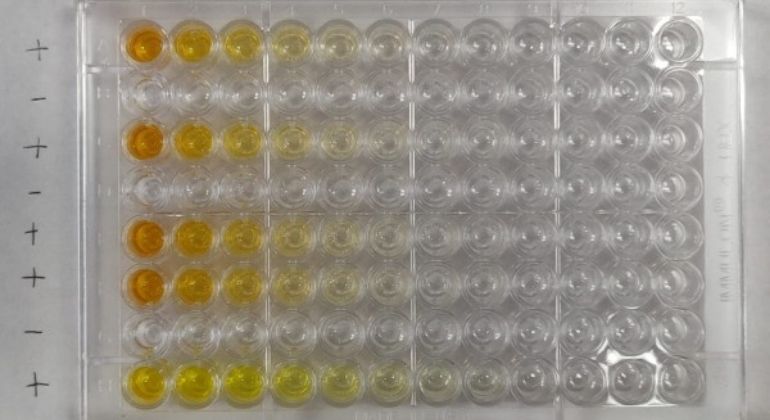
ELISA Plate - the darker the yellow color, the more antibodies in sera. The lighter the color, the less.
Photo Credit: Mount Sinai Health System
The vast majority of individuals infected with mild-to-moderate COVID 19 mount a robust antibody response that is relatively stable for at least five months, according to research conducted at the Icahn School of Medicine at Mount Sinai and published October 28, in the journal Science. Additionally, the research team found that this antibody response correlates with the body’s ability to neutralize (kill) SARS-CoV-2, the virus that causes COVID-19.
“While some reports have come out saying antibodies to this virus go away quickly, we have found just the opposite – that more than 90 percent of people who were mildly or moderately ill produce an antibody response strong enough to neutralize the virus, and the response is maintained for many months,” said Florian Krammer, PhD, Professor of Vaccinology at the Icahn School of Medicine at Mount Sinai and a senior author of the paper. “Uncovering the robustness of the antibody response to SARS-CoV-2, including its longevity and neutralizing effects, is critically important to enabling us to effectively monitor seroprevalence in communities and to determining the duration and levels of antibody that protect us from reinfection. This is essential for effective vaccine development.”
Study findings are based on a dataset of 30,082 individuals, who were screened within the Mount Sinai Health System between March and October, 2020. The antibody test used in this research—an enzyme-linked immunosorbent assay (ELISA)—is based on the virus’s telltale spike protein that contains the machinery that enables it to attach and gain entry into our cells. The ELISA assay was developed, validated, and launched at Mount Sinai by a team of internationally renowned researchers and clinicians. The Mount Sinai antibody test detects the presence or absence of antibodies to SARS-CoV-2 and, importantly, is capable of measuring the titer (level) of antibodies an individual has. The high sensitivity and specificity of this test—meaning that a false negative or false positive is highly unlikely—allowed it to be among the first to receive emergency use authorization from New York State and the U.S. Food and Drug Administration.
In late March, Mount Sinai began to screen individuals for antibodies to SARS-CoV-2 in order to recruit volunteer donors for its convalescent plasma therapy program—one of the first such programs in the nation. The Clinical Laboratories of The Mount Sinai Hospital set up antibody test results using distinct dilutions set at 1:80, 1:160, 1:320, 1:960 or ≥1:2880. The antibody titer score is generated by the number of times the scientist can dilute a patient’s serum and still be able to detect the presence of antibodies. Titers of 1:80 and 1:160 were categorized as low titers; 1:320 moderate; and 1:960 or ≥ 1:2880 were high.
By early October, Mount Sinai had screened 72,401 individuals with a total of 30,082 being positive (defined as detectible antibodies to the spike protein at a titer of 1:80 or higher). Of the 30,082 positive samples, 690 (2.29 percent) had a titer of 1:80; 1453 (4.83 percent) of 1:160; 6765 (22.49 percent) of 1:320; 9564 (31.79 percent) of 1:960; and 11610 (38.60 percent) of 1:2880. Thus, the vast majority of positive individuals had moderate-to-high titers of anti-spike antibodies.
“Our microbiology colleagues generated great science and tools that were brought from the research lab into the clinical laboratory, where we were able to implement robust and compliant diagnostic tests at an unprecedented pace,” said Carlos Cordon-Cardo, MD, PhD, Irene Heinz Given and John LaPorte Given Professor and Chair of Pathology, Molecular and Cell-Based Medicine and last author of the paper. “The tireless efforts of so many have enabled us to uncover knowledge that can help inform COVID-19 policy and aid in vaccine development.”
Determining the neutralizing effects of SARS-CoV-2 is critical to understanding the possible protective effects of the immune response. The research team performed a well-established, quantitative microneutralization assay based on authentic SARS-CoV-2 with 120 samples of known ELISA titers ranging from “negative” to ≥1:2880. They found that approximately 50 percent of sera in the1:80-1:160 titer range had neutralizing activity; 90 percent in the 1:320 range had neutralizing activity; and all sera in the 1:960 to ≥1:2880 range had neutralizing activity.
Another important and outstanding question in the scientific community is the longevity of the antibody response to the spike protein. To answer that question, the team recalled 121 plasma donors at a variety of titer levels for repeat antibody testing at approximately 3 months and 5 months post-symptom onset. When comparing overall titers, they saw a slight drop from a geometric mean titer (GMT) of 764 to a GMT of 690 from the first to second testing time point and another drop to a GMT of 404 for the last testing time point, indicating that a moderate level of antibody is retained by most people 5 months after symptom-onset. In the higher titer range, they observed a slow decline in titer over time. Interestingly, they saw an initial increase in titer for individuals who had originally tested as having low to moderate titer levels. This is in agreement with earlier observations from their study group that indicate seroconversion in mild COVID-19 cases might take a longer time to mount.
“The serum antibody titer we measured in individuals initially were likely produced by plasmablasts, cells that act as first responders to an invading virus and come together to produce initial bouts of antibodies whose strength soon wanes,” said Ania Wajnberg, MD, Director of Clinical Antibody Testing at the Mount Sinai Hospital and first author of the paper. “The sustained antibody levels that we subsequently observed are likely produced by long-lived plasma cells in the bone marrow. This is similar to what we see in other viruses and likely means they are here to stay. We will continue to follow this group over time to see if these levels remain stable as we suspect and hope they will.”
The Mount Sinai data reveals antibody binding titers to the spike protein correlate significantly with neutralization of SARS-CoV-2 and that the vast majority of individuals with antibody titers of 320 or higher show neutralizing activity in their serum that are stable over a period of at least 3 months with only modest declines at the 5-month time point. Correlates of protection have been established for many different viral infections including influenza, measles, hepatitis A, hepatitis B. These correlates are usually based on a specific level of antibody acquired through vaccination or natural infection that significantly reduces the risk of reinfection. The team will continue following this study cohort over longer intervals of time. Although this cannot provide conclusive evidence that these antibody responses protect from reinfection, the team believes it is very likely that the antibodies will decrease the odds of getting reinfected and may attenuate disease in the case of breakthrough infection. To inform policy for the COVID-19 pandemic and for the benefit of vaccine development, it is imperative to swiftly perform studies to investigate and establish a correlate of protection to SARS-CoV-2. Such investigations are currently being carried out by researchers at the Icahn School of Medicine at Mount Sinai.
This work was partially supported by the National Institute of Allergy and Infectious Diseases Centers of Excellence for Influenza Research and Surveillance, the Collaborative Influenza Vaccine Innovation Center, and the generous support of the JPB Foundation, the Open Philanthropy Project, and other philanthropic donations.Mount Sinai has licensed serological assays to commercial entities and has filed for patent protections for serological assays.
About the Mount Sinai Health System
Mount Sinai Health System is one of the largest academic medical systems in the New York metro area, with more than 47,000 employees working across seven hospitals, more than 400 outpatient practices, more than 600 research and clinical labs, a school of nursing, and leading schools of medicine and graduate education. Mount Sinai advances health for all people, everywhere, by taking on the most complex health care challenges of our time—discovering and applying new scientific learning and knowledge; developing safer, more effective treatments; educating the next generation of medical leaders and innovators; and supporting local communities by delivering high-quality care to all who need it.
Through the integration of its hospitals, labs, and schools, Mount Sinai offers comprehensive health care from conception through geriatrics, leveraging innovative approaches such as artificial intelligence and informatics while keeping patients’ medical and emotional needs at the center of all treatment. The Health System includes more than 6,400 primary and specialty care physicians and 10 free-standing joint-venture centers throughout the five boroughs of New York City, Westchester, Long Island, and Florida. Hospitals within the System are consistently ranked by Newsweek’s® “The World’s Best Smart Hospitals,” “Best in State Hospitals,” “World’s Best Hospitals,” and “Best Specialty Hospitals” and by U.S. News & World Report's® “Best Hospitals” and “Best Children’s Hospitals.” The Mount Sinai Hospital is on the U.S. News & World Report® “Best Hospitals” Honor Roll for 2025-2026.
For more information, visit https://www.mountsinai.org or find Mount Sinai on Facebook, Instagram, LinkedIn, X, and YouTube.