The Dr. Henry D. Janowitz Division of Gastroenterology
Our mission is excellence in the delivery of patient care, research, and education in diseases of the GI tract.
The Dr. Henry D. Janowitz Division of Gastroenterology
Including GI Surgery
Our mission is excellence in the delivery of patient care, research, and education in diseases of the GI tract.
This specialty report highlights:
- Perfecting transanal surgery and leading the development of minimally invasive surgical approaches for treatment of colon and rectal cancers
- A study looking at whether specific bacteria in the microbiome passed from mothers to their offspring increase the likelihood that their children eventually will develop inflammatory bowel disease (IBD)
- Researchers have discovered that biomarkers in the blood can help identify patients at risk for inflammatory bowel disease
- Two cases in which screening helped catch pancreatic disease early
- Fecal microbiota transplant (FMT) procedures to treat cases of recurrent C. diff infections
Perfecting Transanal Surgery
Minimally Invasive Approach Helps Much-Scarred Patient
Steven Harris, MD, is well acquainted with the risks of surgery, not only as an internal medicine specialist but as someone who has had his own experiences going under the knife.
In 1991, the Indianapolis physician underwent an emergency colectomy with an ileostomy due to severe inflammation of the colon caused by ulcerative colitis. He subsequently spent six weeks in the hospital due to complications such as sepsis and infection. In 2002, Dr. Harris underwent another operation for a small-bowel obstruction caused by adhesions from the previous surgery, and that resulted in a four-week hospital stay.
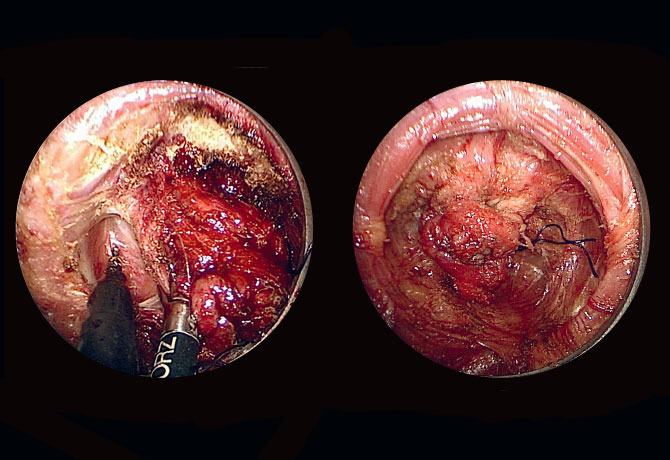
When a routine sigmoidoscopy in summer 2015 revealed mild dysplasia in Dr. Harris’s rectal stump, he knew there was a high risk it could develop into cancer and that he would have to have the stump removed surgically. Dr. Harris, who is now 58, also knew that, given the severity of adhesions in his abdomen, and based on his previous experiences, he wanted to avoid the risk of complications from open surgery, such as ureter damage or the development of more adhesions.
“I started researching my case and came across articles about transanal endoscopic proctectomy,” Dr. Harris says. “There aren’t many doctors who do this procedure. Only three of them are in the United States, and Dr. Patricia Sylla was one of them.”
An associate professor of surgery at The Mount Sinai Hospital, Patricia Sylla, MD, is a recognized leader in the development of minimally invasive surgical approaches for treatment of colon and rectal cancers. In 2009, she collaborated on the first-ever rectal cancer surgery in a human using the natural orifice transluminal endoscopic surgery (NOTES) approach, and she has performed more than 50 such surgeries to date, continually advancing this approach.
Impressed by Dr. Sylla’s credentials, Dr. Harris contacted her in December 2015 to see whether she could remove his rectal stump transanally. Based on his medical records and the fact that he had been classified as having a hostile abdomen, she concluded that he was an ideal candidate for transanal endoscopic proctectomy, but there was a challenge. His MRI revealed that his rectal stump was approximately 15 cm in length, meaning part of it was in his abdomen.
“I knew he had been traumatized by previous surgery, and he had made it clear he did not want anyone to go through the abdomen, if at all possible,” Dr. Sylla says. “Given the length of the rectal stump, I told him I could not guarantee I could do the whole procedure from the bottom. But I would do my best to minimize any incisions from the top by doing it all laparoscopically, with as little dissection as possible, and he agreed to that.”
As it turned out, Dr. Sylla started the seven-hour procedure, which took place in February 2016, by entering through the abdomen laparoscopically to remove the upper part of the rectum. Although the abdominal scarring she encountered did not appear hostile, it was extensive enough that she spent three and one-half hours performing lysis of adhesions before starting the rectal resection.
“It took a lot of patience to cut down the adhesions, which isn’t easy to do with laparoscopy, because you’re not using your hands to feel the small bowel,” Dr. Sylla explains. “It’s a bit more tedious and meticulous, but the scope has really great magnification, so everything is exposed and easy to see.”
Once Dr. Sylla was satisfied that she had sufficient exposure, she completed the surgery transanally, removing the remainder of the rectum and anus while another surgeon assisted from the top. “For safety reasons, the consensus is that there should be two teams working together—top and bottom—for a transanal endoscopic proctectomy,” Dr. Sylla says. “It effectively reduces the risk of surgical error or organ injury and helps to speed up the operation.”
The final pathology of the rectum demonstrated atypia, indefinite but probably positive for dysplasia. Unlike after his previous surgeries, Dr. Harris was able to leave the hospital in four days with little pain and few side effects. “I would say she saved my life, no doubt about it,” he says.
Although a transanal endoscopic approach can be more time-consuming and somewhat challenging, Dr. Sylla says it is often preferable to an abdominal one for rectal procedures such as proctectomy involving rectal cancer; proctectomy for Crohn’s disease, ulcerative colitis, or other benign conditions; and rectal lesion excision.
“The advantages are incredible, even if you use only the approach for part of the surgery,” Dr. Sylla says. “It’s much less painful; it reduces the risk of infection, scarring, and hernias; and recovery times are shorter. It’s a really great addition to our armamentarium.”
IBD and the Neonatal Microbiome
Studying Link Between Maternal Bacteria and Disease
Diaper-changing is not usually considered one of the joys of parenthood. But Emily, a new mother who has inflammatory bowel disease (IBD), is delighted that her daughter’s diapers might someday help lead to prevention of the disease, thanks to a new Mount Sinai study.
Launched in December 2014, the study, known as MECONIUM for “exploring MEChanisms Of disease traNsmission In Utero through the Microbiome,” is looking at whether specific bacteria in the microbiome passed from mothers to their offspring increase the likelihood that their children eventually will develop IBD.
Inga Peter, PhD, an Associate Professor of Genetics and Genomic Sciences at the Icahn School of Medicine at Mount Sinai, says that this is the first time that researchers have examined the changes in the microbiome in pregnant women with IBD and characterized how those changes may affect the gastrointestinal tract of babies.
“The initial colonization—what the fetus gets in terms of bacteria in utero and immediately after birth—is now believed to play a very crucial role in initiating the baby’s mucosal immune system,” Dr. Peter says.
“Some studies have also shown that the rate of IBD transmission is higher if the mother, rather than the father, has the disease. Based on these facts, our hypothesis is that mothers with IBD pass on some suboptimal composition of bacteria to their babies in utero, putting them at risk of developing IBD or other diseases.”
To test that hypothesis, Dr. Peter and her interdisciplinary team are collecting extensive samples and reproductive-health information from hundreds of families—both with and without a history of IBD. The samples include stool and saliva from everyone in the household; third-trimester vaginal swabs; the placenta, amniotic membranes, umbilical cord blood, and meconium—the newborn’s first fecal discharge—following birth; breast milk; and diapers at regular intervals during the first three years of the baby’s life.
To date, Dr. Peter has observed that pregnant women with IBD have a very different, IBD-prone, microbiome than pregnant women without IBD—a finding that was not unexpected. Furthermore, the microbiome of babies born to mothers with IBD is different from that of the babies’ non-IBD counterparts and resembles that of their mothers, and these differences persist with age. The question now is which specific bacterial strains passed on or under-transmitted by mothers with IBD are the cause of IBD in their offspring.
“We know in general which bacterial taxa are enriched and which are depleted among mothers with IBD, particularly the major players,” Dr. Peter says. “We’re absolutely sure there are particular bacterial strains that make the difference. By tracking these bacterial strains to see what is present or lacking, we can recommend targets for microbial modification in pregnant women with IBD in order to potentially reduce IBD transmission in utero.”
Mothers like Emily are happy to participate in the study for the promise that it holds. “I think it’s exciting that my daughter’s diapers will be used for research as important as this,” she says.
Advancing IBD Study and Treatment
Biomarkers May Help Detect Disease Years in Advance
Mount Sinai Health System researchers have discovered that biomarkers in the blood can help identify patients who are at risk for inflammatory bowel disease (IBD). For the first time, researchers found that these markers, in the form of antimicrobial antibodies, were present in serum up to six years before diagnosis of IBD and that the higher the numberof antibodies present, the greater the chance of complications like abscesses, strictures, or the need for surgery.
The results of the study were published June 15, 2016, in Alimentary Pharmacology & Therapeutics. Mount Sinai’s study partners included the Naval Medical Research Center, Mayo Clinic, Prometheus Laboratories, and Janssen.
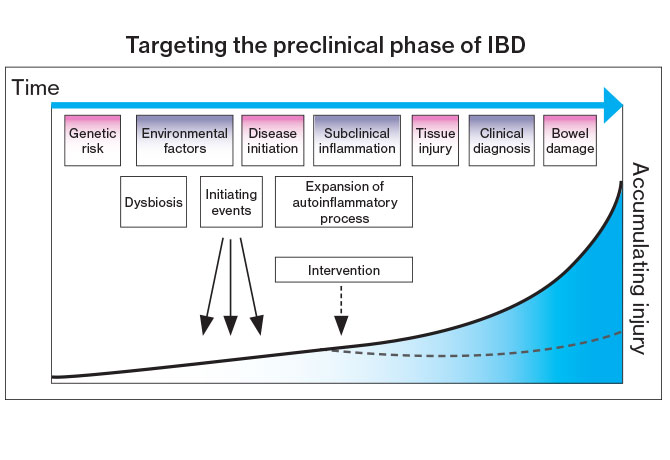
Proposed phases in the evolution of IBD from health to disease. Intervening (dashed arrow) in the preclinical period could truly represent a window of opportunity to reverse or halt the autoinflammatory process and prevent immune dysregulated pathways from progressing into overt disease.
Jean-Frederic Colombel, MD, co-senior author and Co-Director of the Susan and Leonard Feinstein Inflammatory Bowel Disease Clinical Center at Mount Sinai, stresses the importance of early detection of IBD. “IBD is a progressive disease, and by the time it’s diagnosed one-third of patients present with complications,” he says. “Our biomarkers study will hopefully lead to development of a tool to detect the disease before the first clinical manifestations appear so that we can apply preventive strategies.” In the case of a family with children considered at high risk for developing the disease, Dr. Colombel says, these strategies could include changing the diet and manipulating the gut microbiome, host to the largest microbial community in the human body.
Clinical data from the Defense Medical Surveillance System and serum samples from the Department of Defense serum repository made the study possible. From more than seven million members of the armed forces, researchers identified patients with a diagnosis of Crohn’s disease and retrieved up to four pre- and postdiagnostic serum samples from each. That made this study the first to show the progression of these markers using multiple samples at various stages before diagnosis. In the months ahead, investigators will expand the study to look for preclinical biomarkers beyond antimicrobial antibodies, which could include proteomics and infectious agents like viruses.
Pancreatic Cancer and Genetics
In Two Cases, Screening Helped Catch Disease Early
Medical professionals are increasingly recognizing the role that genetics plays in pancreatic cancer. Expert consensus suggests that anyone who has had two first-degree relatives with the disease should be screened for it. Using procedures such as endoscopic ultrasound (EUS), medical centers such as the Mount Sinai Health System are able to better assess troublesome growths to ensure timely resections in patients with a family history of pancreatic cancer.
Martin Abrams made that connection in 2012 when he met with Aimee Lucas, MD, Assistant Professor of Medicine (Gastroenterology) at the Icahn School of Medicine at Mount Sinai (ISMMS), to assess his risk of developing pancreatic cancer. The Rockaway, Queens, resident, now 78, had lost a sister to the disease in 2009, and his brother, who would pass away from pancreatic cancer in July 2013, had just been diagnosed.
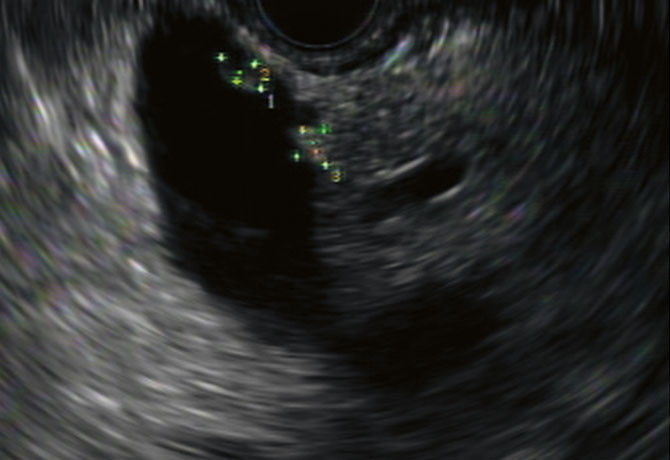
Dr. Lucas recommended that Mr. Abrams undergo an EUS, which was performed in August 2012 by Christopher DiMaio, MD, Associate Professor of Medicine (Gastroenterology) at the ISMMS and Director of Therapeutic Endoscopy at Mount Sinai Hospital. Dr. DiMaio found three cysts that were consistent with an intraductal papillary mucinous neoplasm (IPMN), a type of tumor that grows in the pancreatic ducts, but there were no signs of cancer at that time.
A magnetic resonance cholangiopancreatography (MRCP) exam followed in September, and Dr. DiMaio performed a second EUS in December. That EUS revealed five cysts, one of which had not only grown considerably in size, from 1.9 cm to 3.1 cm, but also contained a nodule, which is considered a high-risk feature. The Mount Sinai multidisciplinary gastrointestinal tumor board recommended that Mr. Abrams undergo surgery, but he decided on further monitoring.
His view began to change in April 2013, when a third EUS revealed the presence of a sixth cyst. In August, Daniel Labow, MD, Associate Professor of Surgery and Director of Surgical Oncology at The Mount Sinai Hospital, performed a distal pancreatectomy. The final pathology found that Mr. Abrams had a branch duct IPMN with high-grade dysplasia.
“This was the best possible timing because we found and removed the cyst right before it developed into cancer,” Dr. DiMaio says. “You don’t want to act too soon, because these lesions grow slowly and there is the possibility that you could remove a low-risk or benign cyst instead.”
Mr. Abrams continues to be monitored through ongoing MRI and EUS exams. He is now encouraging others with a family history of the disease to undergo screening and is advocating for more research on pancreatic cancer.
Like Mr. Abrams, Andrea Feller had two first-degree relatives with pancreatic cancer. But the 59-year-old Brooklyn resident’s case was particularly unusual in that they were both of her parents.
Although environmental factors or chance could have explained that phenomenon, Ms. Feller was referred to Dr. Lucas in September 2015 for a risk assessment. Dr. Lucas ordered genetic testing for Ms. Feller, but the results came back negative.
“We don’t know all of the genes associated with pancreatic cancer, so gene testing at this point is not a sure thing,” Dr. Lucas explains. “Even if your gene test comes back positive, it’s not a guarantee you’re going to get cancer, because the genes aren’t what we call fully penetrant.”
A subsequent MRCP exam, however, revealed a 5 mm branch duct IPMN in Ms. Feller’s pancreas. Given Ms. Feller’s family history, Dr. Lucas recommended an EUS, which Dr. DiMaio performed in April 2016. Although the cyst appeared small and benign to Dr. DiMaio, he detected something else on the EUS that the MRCP did not—a 1.6 cm mass lesion in a different area that was biopsied and demonstrated pancreatic cancer.
“That is something we would hope the MRCP would detect,” Dr. Lucas says. “But MRCP may not be as good as EUS at picking up small solid lesions, so either it was there and we could not detect it, or it developed after the MRCP.”
In May 2016, Dr. Labow, the surgeon, performed a Whipple resection. Ms. Feller has since undergone chemotherapy and radiation, and she too is encouraging family and friends with family histories of pancreatic cancer to have risk assessments done at Mount Sinai.
Fecal Matter Aids in C. Diff Cases
Technique Is Also Studied for Other Diseases
Clostridium difficile (C. diff), which once mainly affected older patients in nursing homes and hospitals, has become more widespread, aggressive, and hard to treat with antibiotics. Increasingly, medical centers such as Mount Sinai are using fecal microbiota transplant (FMT) procedures to treat cases of recurrent C. diff infections, and our patient Violet is one of Mount Sinai’s many success stories.
The 69-year-old Manhattan resident began experiencing diarrhea in fall 2012. She had undergone dental work and, because of a previous knee replacement, was prescribed clindamycin, an antibiotic typically given to dental patients with artificial body parts.
Violet underwent tests that initially came back negative for C. diff until a DNA test confirmed the diagnosis. She had been assumed to have ulcerative colitis, in part due to problems such as pyoderma gangrenosum, which caused an ulcerative rash on her lower extremities.
Several rounds of antibiotics followed, including metronidazole and vancomycin, to treat her C. diff, but Violet’s C. diff and diarrhea recurred after each round. This continued until September 2014, when a specialist referred her to Ari Grinspan, MD, an Assistant Professor of Medicine (Gastroenterology) at the Icahn School of Medicine at Mount Sinai and the Director of Gastrointestinal Microbial Therapeutics at The Mount Sinai Hospital.
Dr. Grinspan began performing FMTs at Mount Sinai in 2013 and has completed more than 150 transplants to date. He says the procedure repopulates the colon with healthy bacteria, restoring colonization resistance so that C. diff cannot take hold.
“Violet had already had five C. diff infections, and once you have had more than three episodes, the likelihood that antibiotics alone will cure you drops below 50 percent,” Dr. Grinspan says. “If I give a patient one fecal microbiota transplant, there’s a greater than 90 percent chance that he or she won’t have C. diff again.”
Dr. Grinspan performed the FMT in October 2014. At the time, Violet was taking vancomycin, and he instructed her to stop taking it so it would not interfere with the procedure. Violet also undertook a standard colonoscopy bowel preparation prior to the FMT. After mixing the donor stool with 500 ccs of saline and shaking it, Dr. Grinspan infused the solution into Violet’s cecum and terminal ilium. He also gave her Imodium to prevent diarrhea so the transplant could take hold. Normally, Dr. Grinspan does not recommend testing to confirm eradication of C. diff. But Violet reported increased urgency and loose stools following the procedure, so he performed a sigmoidoscopy followed by stool tests, all of which found no evidence of active C. diff, and Violet has had zero episodes of infection since the procedure.
Although the U.S. Food and Drug Administration restricts the use of FMT to treatment of C. diff infections, Dr. Grinspan says there is active research exploring its utility in a range of diseases, including obesity, diabetes, and autism. He adds that the most promising research involves the role of FMT in treating inflammatory bowel disease, in which he is involved.
“There have been several recent clinical trials that have shown FMT to be effective in treating some patients with ulcerative colitis,” Dr. Grinspan says. “We are studying the microbiome in these patients for post-FMT changes to see whether there are microbial signatures that correspond with disease remission.”
Teaching People Coping Skills for Chronic Illness
Few words convey comfort quite like “home.” It is where we learn the necessary skills to face everyday challenges and a place we can return to whenever we need support.
That is the thinking behind the Mount Sinai IBD Home initiative at the Susan and Leonard Feinstein Inflammatory Bowel Disease Clinical Center. Launched in January 2016, IBD Home combines psychosocial, medical, and nutritional expertise to help Mount Sinai’s inflammatory bowel disease (IBD) patients build their capacity to manage this chronic condition.
Laurie Keefer, PhD, a senior faculty member with Medicine (Gastroenterology) at The Mount Sinai Hospital and a Co-Director of IBD Home with Marla Dubinsky, MD, says the initiative is unique in that it emphasizes building patient resilience and self-sufficiency.
“This is about reducing disability, improving relationships, and enhancing quality of life,” Dr. Keefer says. “Given that IBD is often diagnosed in early adulthood, you can imagine how resilience makes a difference in terms of someone reaching his or her full potential. By taking a 360-degree approach to care and skills development, we ensure that every patient we diagnose is set up for success.”
Each week, the multidisciplinary IBD Home team, which includes a child-life specialist, social worker, nutritionists, nurse-practitioners, gastroenterologists, and a psychiatrist,
meets to assess IBD patients, determine who requires additional support to manage their disease, and enroll those patients in IBD Home. Participants are monitored through consultations and self-reporting using mobile applications, and the team intervenes when necessary.
“The types of patients we enroll could include adults who are struggling with pain management or who have sought significant health care assistance in recent months, and
young people who are having difficulty swallowing pills, are afraid of needles, or are missing school due to the disease,” Dr. Keefer says.
Many programs offered through IBD Home are focused on building resilience among adolescents and young adults. “We give them the wings to be self-reliant, but they can always come back to us if they need help with anything.”
Managing IBD With an App
Digital Initiative, Seen as a Model, Gets a Clinical Trial
The Mount Sinai Health System is conducting a two-year clinical trial of a new app that not only brings monitoring and management of inflammatory bowel disease (IBD) into the 21st century but also establishes a model for launching more digital initiatives for patient-reported outcomes.
Developed by Mount Sinai’s AppLab at the Division of Gastroenterology, HealthPROMISE is a HIPAA-compliant, cloud-based app that enables IBD patients to enter biweekly updates on their quality of life and the symptoms they experience, such as number of bowel movements and level of pain. Patients also maintain checklists of key quality metrics determined by physicians, such as colonoscopies, flu shots, and bone-density tests. Physicians and care navigators can access these data through a secure dashboard and intervene when necessary, for example by recommending changes in medication based on the symptoms patients report.
Ashish Atreja, MD, MPH, Assistant Professor of Medicine (Gastroenterology) at the Icahn School of Medicine at Mount Sinai and Director of the AppLab, says HealthPROMISE is a proactive approach to treating chronic ailments like IBD.

“Previously, we had no way of knowing if patients were falling through the cracks or had gone ahead with surgery such as strictureplasty or resection, because often their primary-care physician or the emergency department to which they had been admitted was affiliated with another hospital,” Dr. Atreja notes. “With HealthPROMISE, we can monitor for symptoms and proactively reach out to those patients to potentially prevent surgery, or we can deliver information that puts them in the driver’s seat to improve their health and quality of life.”
More than 300 Mount Sinai IBD patients are participating in the clinical trial, which launched in spring 2015. One group has full access to HealthPROMISE’s reporting, tracking, and educational tools, whereas the control group has only the educational resources.
“In the first five months, we observed that approximately 89.5 percent of patients with full HealthPROMISE access were actively using the app, compared with 27.7 percent of control-group patients,” Dr. Atreja says. “We also saw lower symptom-burden scores among HealthPROMISE users.”
Six IBD centers in the United States and Canada—the Mount Sinai Health System, the University of Pittsburgh Medical Center, the University of Miami, Northwestern University, McGill University, and Baystate Health—have joined to form the HealthPROMISE Consortium, giving their patients access to the app.
Dr. Atreja also is exploring opportunities to use HealthPROMISE as a model to enable more patient-reported outcome initiatives. For example, the app will be used to collect data for the new fecal microbiota transplantation (FMT) National Registry, which promotes research into the gut microbiome and use of FMT.
Division of Gastroenterology
Download the Winter 2017 Specialty Report