The Precision Immunology Institute at Mount Sinai
The Precision Immunology Institute at Mount Sinai integrates disease-focused immunological research programs across 42 labs and two leading-edge technology centers.
The Precision Immunology Institute at Mount Sinai
The Precision Immunology Institute at Mount Sinai integrates disease-focused immunological research programs across 42 labs and two leading-edge technology centers.
This report highlights:
- Harnessing the immune system to battle cancer
- Recent PrIISM Highlights: Publications
- NIAID award: Accelerating research into peanut allergy
- Developing a new generation of immunologists
- Message from the Director: Miriam Merad, MD, PHD
Harnessing the Immune System to Battle Cancer
The Precision Immunology Institute (PrIISM) at the Icahn School of Medicine at Mount Sinai has joined with The Tisch Cancer Institute, Mount Sinai’s NCI-designated Cancer Center, to build a state-of-the-art cancer immunology program to advance research in the most innovative and promising areas of investigation.
Leading an effort to identify new immune targets in cancer
PrIISM and The Tisch Cancer Institute have received one of four Cancer Immune Monitoring and Analysis Center (CIMAC) awards from the National Cancer Institute. The four recipients—Mount Sinai, Stanford Cancer Institute, The University of Texas MD Anderson Cancer Center, and Dana-Farber Cancer Institute—comprise a network to identify mechanisms of resistance to immunotherapy.
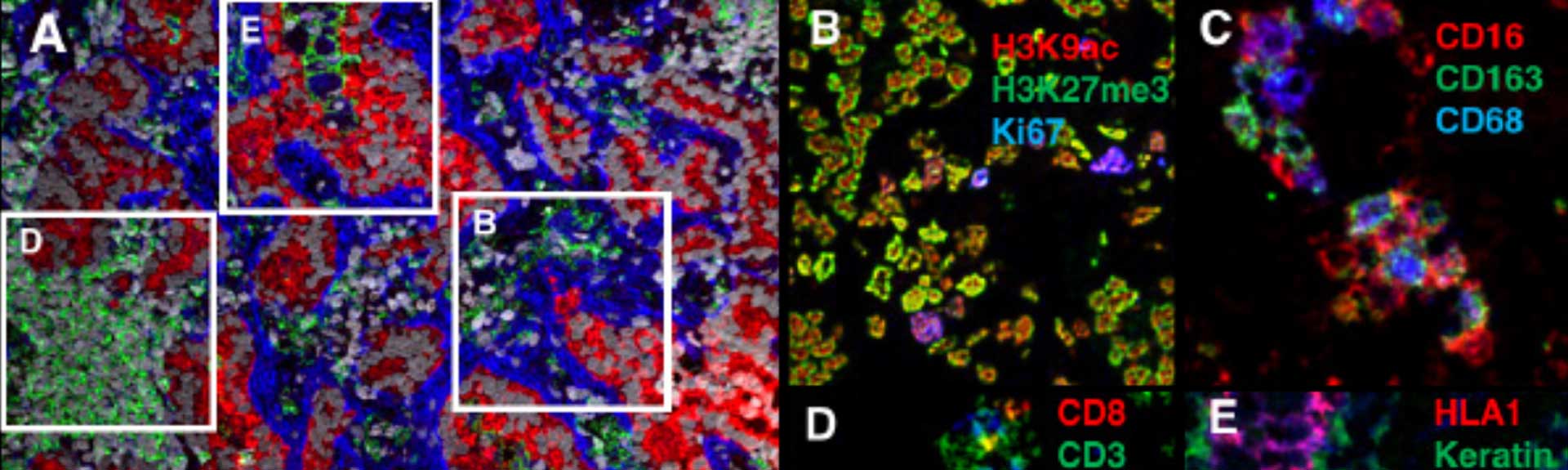
The CIMAC centers will systematically collect, process, and analyze blood and tumor tissue samples using cutting-edge technology including mass cytometry (CyTOF) and single-cell transcriptomics and proteomics, and will develop a data pipeline for shared use.
CIMACs will create the first system for integrated analysis across immunotherapy trials. Identifying predictive biomarkers that inform the likelihood of benefit or toxicities from therapies and the mechanisms of response or resistance, and identify novel potential immune targets, will help advance design of rational combinations of immunotherapies to optimize patients’ outcomes.
The CIMAC network is led by Sacha Gnjatic, PhD, Associate Professor of Medicine (Hematology and Medical Oncology) and Oncological Sciences at the Icahn School of Medicine, and co-Director of PrIISM’s Human Immune Monitoring Center.
Generating patient-specific neoantigen vaccines
PrIISM investigators are testing a personalized genomic vaccine (PGV) in pioneering clinical trials, the first of which has shown early success. Nina Bhardwaj, MD, PhD, Ward-Coleman Chair in Cancer Research and Director of Immunotherapy at the Icahn School of Medicine, has designed a PGV to treat cancers with high rates of mutations—lung, breast, and bladder cancers, among others. Dr. Bhardwaj’s team sequences the DNA and RNA of a patient’s tumor in our unique Vaccine and Cell Therapy Lab to identify specific mutations, and generates a PGV based on this genetic profile.
Patients determined to be cancer-free after the initial tumor resection receive injections of the personalized vaccine over a period of six months. The goal is to identify the neoantigens (new proteins that arise as cells mutate and tumors evolve) and to create a vaccine specific to these neoantigens that will stimulate an immune response to tumor cells.
The first patient to receive the vaccine, a head and neck cancer patient, has shown evidence of immunity, and eight months after completing treatment, the cancer has not recurred. Five additional patients have now been enrolled in the trial, one with lung cancer and four with multiple myeloma. Dr. Bhardwaj was recently appointed co-Director of the Neoantigen Selection Alliance at the Parker Institute for Cancer Immunotherapy, a San Francisco-based national collaboration of leading research institutions.
Novel in situ vaccine alters disease progression
Joshua Brody, MD, a PrIISM investigator and Director of the Lymphoma Immunotherapy Program at the Icahn School of Medicine, is testing a novel approach to induce anti-T-cell immune response in lymphoma, with potential use for other cancers. Radiotherapy administered at the tumor site promotes the release of tumor antigens, followed by injection of a recombinant protein that expands dendritic cells (DCs) and TLR agonists that activate the tumor antigen-carrying DCs. Preclinical trials showed regression of the disease and paved the way for additional clinical trials, including a trial combining the in situ vaccine with anti-PD-1 or PD-L1 antibodies for patients with breast, head and neck, liver, or ovarian cancers.
Dr. Brody’s lab has also developed a related “immune-transplant” approach combining a checkpoint blockade with stem cell transplantation and T-cell transfer using the patients’ own cells, which has shown remarkable early results. The strategy is now being tested for patients with aggressive types of lymphoma, with similar success.
Using gut microbes to promote tumor resistance
The potential for treating disease or improving therapies by manipulating the gut microbiome is drawing increasing interest. Lab teams directed by Jeremiah Faith, PhD, Director of PrIISM’s Microbiome Translational Center, and PrIISM Director Miriam Merad, MD, PhD, are demonstrating the potential for such a strategy in cancer.
They have identified a specific human microbial community that actively promotes tumor resistance to checkpoint blockade (anti- PD-1) in melanoma. Gnotobiotic (germ-free) mice with melanoma are currently being transplanted with communities of these human microbes isolated from patient fecal samples. Transplantation is followed by deep mapping of gut and tumor tissue and lymph-draining nodes with the goal of identifying microbial-induced changes associated with tumor response or regression. Identifying microbial differences that could inhibit, or enhance, the effectiveness of the checkpoint blockade therapies for melanoma could have potential impact for many other cancers as well.
Recent PrIISM Highlights: Publications
“Protein Barcodes Enable High Dimensional Single-Cell CRISPR Screens” Cell, November 2018
Genetic screens using CRISPR pools are being widely employed to identify gene functions. However, current technology using DNA as barcodes has critical shortcomings, including limited phenotyping capability and bulk-cell resolution. To enable novel screening capabilities, Brian Brown, PhD, Associate Director of PrIISM, and his team developed a barcoding system operating at the protein level. By synthesizing modules encoding triplet combinations of linear epitopes, they generated hundreds of unique protein barcodes (Pro-Codes).
Pro-Code vectors were introduced into cells and analyzed by CyTOF mass cytometry. Using just 14 antibodies, the team detected 364 Pro-Code populations, establishing the largest set of protein-based reporter genes. Pairing each with a different CRISPR enabled simultaneous analysis of multiple phenotypic markers, revealing two interferon-stimulated genes implicated in killing cancer cells, as well as the Socs1 gene as a negative regulator of PD-L1. Pro-Codes open new possibilities for functional genomics, and for identifying immune and cancer regulators.
“A Neoantigen Fitness Model Predicts Tumor Response to Checkpoint Blockade Immunotherapy” Nature, November 2017
Is it possible to predict tumor response to immunotherapy? PrIISM member Benjamin Greenbaum, PhD, Assistant Professor of Medicine (Hematology and Medical Oncology), Pathology, and Oncological Sciences, and colleagues from the Icahn School of Medicine and Memorial Sloan Kettering Cancer Center developed the first mathematical model that can predict how a cancer patient will benefit from certain immunotherapies.
The model captures aspects of the tumor’s evolution, specifically neoantigens (the unique proteins that arise from tumor mutations) and the underlying interactions with the immune system in response to the drugs. The ability to predict and identify “low fitness” (less robust) neoantigens will be critical for the development of more effective immunotherapies, particularly for the design of neoantigen vaccines.
“Innate Immune Landscape in Early Lung Adenocarcinoma by Paired Single-Cell Analysis” Cell, May 2017
Approximately 50 percent of patients with early non-small-cell lung cancer relapse after treatment. Treating the cancer early could have a huge impact on both quality of life and survival. However, before this study, relatively little was known about immune response in early lung cancer.
PrIISM Director Miriam Merad, MD, PhD, collaborated with Raja Flores, MD, Chair of Thoracic Surgery for the Mount Sinai Health System, and a team of pathologists, thoracic surgeons, and Mount Sinai investigators to map and analyze the molecular and cellular components of early lung tumors with a level of resolution not previously achieved, leveraging platforms for high-dimensional profiling of patient samples available through the Institute’s Human Immune Monitoring Center. Their study revealed that early-stage tumors already contain immune components that likely compromise T-cells’ ability to protect against cancer.
NIAID Award: Accelerating Research Into Peanut Allergy
A million and a half children in the United States are affected by peanut allergy. While studies have so far focused on people with “exquisite allergy”—those who react to as little as one-50th of a peanut—the majority only react to larger amounts. There are currently no methods for predicting reaction thresholds to peanut allergy and no FDA-approved treatments for any food allergy.
M. Cecilia Berin, PhD, Deputy Director of the Elliot and Roslyn Jaffe Food Allergy Institute and Professor of Pediatrics at the Icahn School of Medicine, and Jaffe Institute colleagues have been awarded a $7.5 million grant from the National Institute of Allergy and Infectious Diseases (NIAID) to advance research into high-threshold peanut allergy.
The grant will support three projects: a clinical trial, investigation into the immune pathways that affect the threshold of reactivity and those that promote the development of tolerance to peanut, and a computational genomic study to identify biomarkers that predict reaction thresholds. Researchers will use monitoring and profiling assays and technologies in the Human Immune Monitoring Center, including CyTOF mass cytometry analysis.
Developing a New Generation of Immunologists

Training New Doctors
The Institute’s new Cross-Disciplinary Immunotherapy Clinical Fellowship Program welcomed its inaugural fellows in 2018. The Program—the first of its kind in the country— is designed around a novel curriculum created to train a new generation of clinical leaders in immune modulation. Postresidency clinical fellows will perform research in their primary laboratories and in the Human Immune Monitoring Center, participate in clinical trial protocols, teach, and work collaboratively with Icahn School of Medicine at Mount Sinai scientists, clinicians, and MD/PhD students. Boehringer Ingelheim is funding two immunotherapy fellowship awards in gastroenterology, while Pfizer is funding two fellowship awards in dermatology.
Expanding Graduate Training
We are expanding our immunology graduate program and welcome new applicants. Our program currently has 24 immunology students, including 14 PhD and 10 MD/ PhD students with very diverse backgrounds in mathematics, physics, engineering, biochemistry, biology, and industry. Housed on Madison Avenue, one block from Central Park and the Museum Mile in Manhattan, our program trains students in basic immunology, computational immunology, single-cell technology, and clinical medicine. Our students are involved in all aspects of the training programs, have the opportunity to help design novel classes, lead the work-in-progress immunology series, organize the yearly immunology retreat, and select and house seminar speakers from all over the world.
For more information, please visit our website at https://icahn.mssm.edu/research/immunology/education.
Recruiting New Faculty
We are recruiting faculty! Mount Sinai understands the growing importance of the immune system to medicine and is greatly expanding the Precision Immunology Institute. We are actively recruiting basic biologists, physician-scientists, and computational biologists. We provide competitive start-up packages and the opportunity to work in a highly collaborative multidisciplinary environment. We especially encourage postdocs ready for independence to apply. For more information, please visit our website at https://icahn.mssm.edu/research/immunology or contact immunology@gmail.com.
Message from the Director: Miriam Merad, MD, PHD

The realization that the immune system contributes to almost every disease process opens up enormous potential for transforming discoveries in basic immunology into effective therapies. Immunologists are bringing their expertise and talent to investigate not only diseases traditionally understood as immune disorders (such as allergy, immune deficiencies, autoimmune disorders, and inflammatory diseases), but also to new frontiers, including cancer, neurodegenerative disorders, and psychiatric and cardiovascular diseases.
Harnessing the power of the immune system can revolutionize medical care, but the task remains immense. Optimizing this potential requires rethinking the way we do science and building novel and integrated teams that bring basic immunologists, translational and clinical investigators, technologists, physicists, mathematicians, and computational biologists together to frame new questions, build novel technologies, and identify the precise pathways implicated in disease and the mechanisms of resistance and response to immunotherapies.
The Precision Immunology Institute (PrIISM) is a new entity at Mount Sinai, integrating disease-focused immunological research programs across 42 labs and two leading-edge technology centers, the Human Immune Monitoring Center (HIMC) and the Microbiome Translational Center. HIMC serves as the immune monitoring center for a number of large collaborative e_ orts with academic and industry partners. HIMC’s capabilities include novel technologies for analyzing single cells at an unprecedented level of resolution, including three CyTOF® 2 mass cytometers, a CITE-seq platform that combines RNA and antibody profiling at single-cell resolution, and a newly installed multiplexed ion beam imaging (MIBI) tissue analyzer system, which allows detection of up to 54 molecules (including RNA, proteins, and DNA) on a single tissue section.
I am excited by the breadth and strength of the Mount Sinai immunology community and inspired to help build a collegial, integrated, and impactful immunology program at our institution. Immunology is reinventing medicine, and it is a privilege to be a part of this transformative effort.
The Precision Immunology Institute at Mount Sinai
Download the Precision Immunology Report