Mount Sinai Researcher Receives NCI Grant to Study New Strategies to Prevent Cancer Metastasis
Study will explore mechanisms that enable cancer cells to remain dormant
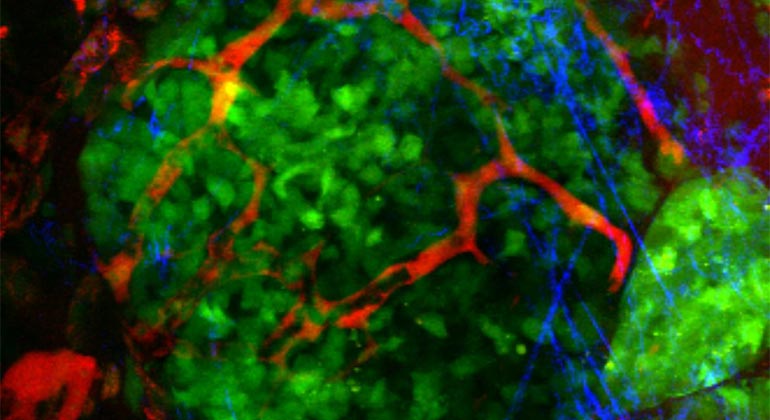
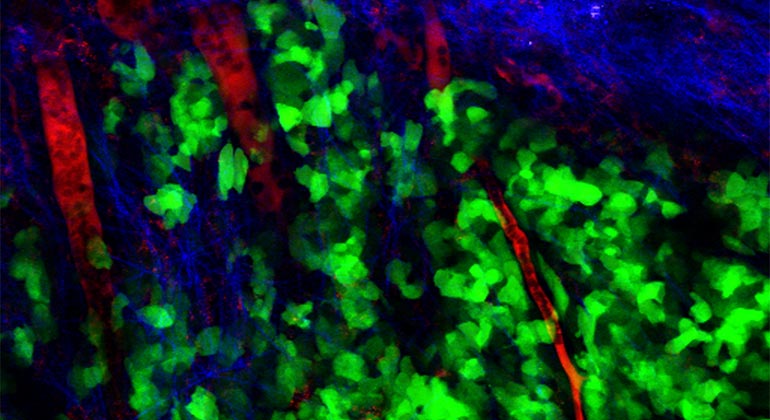
The Tisch Cancer Institute at Mount Sinai has been awarded a $1.9 million grant by the National Cancer Institute for an innovative study that will explore the mechanisms that enable tumor cells to spread from their original site but remain dormant for some time before becoming metastatic and threatening the lives of patients.
Jose Javier Bravo-Cordero, PhD, Assistant Professor of Medicine (Hematology and Medical Oncology) at the Icahn School of Medicine at Mount Sinai, and Scientific Advisor in the Advanced Bioimaging Center at The Tisch Cancer Institute, is the Principal Investigator for this five-year award.
Metastasis—cancer that has spread from its original site to grow in other parts of the body—is the main cause of death among patients with cancer, but the mechanisms involved in this phenomenon are unclear. Disseminated tumor cells, cells that have left the primary tumor and successfully seeded secondary target organs but have not yet formed tumors, have been recognized as crucial in initiating metastasis years or decades after patients have undergone cancer treatment.
This delay in growth is mediated by a process called tumor dormancy, in which tumor cells cease dividing but can be reactivated under the right environmental conditions. These cells are less vulnerable to the immune system and cancer treatments, including chemo and radiation. Studies have shown that tumor cell interactions with the extracellular matrix (ECM)—a molecular network that contributes to the structural integrity and strength of surrounding cells—regulate the hallmarks of cancer from tumor growth to metastasis. However, the role that ECM molecules and receptors play in the dormancy of disseminated tumor cells is unknown, as is whether the tumor cells develop dormancy-supportive niches that sustain them in this state.
“Our hypothesis is that the dormant disseminated tumor cells are constructing a dormancy-supportive ECM niche that is rich in collagen III, an ECM protein,” said Dr. Bravo-Cordero.
“This grant will enable us to better understand the role of collagen III in the construction of these niches at the metastatic organs and define the factors in the tumor ECM microenvironment that contribute to this phenomenon.”
The study will use high-resolution imaging tools combined with dormancy models to study tumor cell-ECM interactions during dormancy. The research team is developing an imaging approach that includes multiphoton imaging and second harmonic generation and activity reporters for dormancy-related genetic pathways.
“Uncovering the mechanisms that regulate dormant cell-ECM interactions will enable us to devise strategies that either prevent the awakening of the disseminated cells or that enable us to kill these cells by targeting the ECM microenvironment, which would result in a significant reduction in the annual number of deaths from metastatic cancers,” Dr. Bravo-Cordero said.
About the Mount Sinai Health System
Mount Sinai Health System is one of the largest academic medical systems in the New York metro area, with 48,000 employees working across seven hospitals, more than 400 outpatient practices, more than 600 research and clinical labs, a school of nursing, and a leading school of medicine and graduate education. Mount Sinai advances health for all people, everywhere, by taking on the most complex health care challenges of our time—discovering and applying new scientific learning and knowledge; developing safer, more effective treatments; educating the next generation of medical leaders and innovators; and supporting local communities by delivering high-quality care to all who need it.
Through the integration of its hospitals, labs, and schools, Mount Sinai offers comprehensive health care solutions from birth through geriatrics, leveraging innovative approaches such as artificial intelligence and informatics while keeping patients’ medical and emotional needs at the center of all treatment. The Health System includes approximately 9,000 primary and specialty care physicians and 10 free-standing joint-venture centers throughout the five boroughs of New York City, Westchester, Long Island, and Florida. Hospitals within the System are consistently ranked by Newsweek’s® “The World’s Best Smart Hospitals, Best in State Hospitals, World Best Hospitals and Best Specialty Hospitals” and by U.S. News & World Report's® “Best Hospitals” and “Best Children’s Hospitals.” The Mount Sinai Hospital is on the U.S. News & World Report® “Best Hospitals” Honor Roll for 2025-2026.
For more information, visit https://www.mountsinai.org or find Mount Sinai on Facebook, Instagram, LinkedIn, X, and YouTube.