Cellular Therapy Service
The Mount Sinai Health System is a leader in cellular therapy research and clinical applications for cancer and other conditions. Cellular therapy is a type of immunotherapy. It uses your immune system to attack tumors and kill harmful cells.
The immune system helps the body fight infections and diseases. It also can prevent or curb the growth of many cancers. However, cancer cells have ways to avoid destruction by the immune system. For example, genetic changes can make them less visible to the immune system. As cancer progresses, the immune system may become more and more compromised. This can contribute to tumor growth. Our Cancer Immunology Research Program is working on this area. We strive to identify the mechanisms that make it difficult for the immune system to defend itself against cancer. With this information, we will be better able to develop new immunotherapies.
There are several types of immunotherapy, as listed by The National Cancer Institute. This video from the National Cancer Institute explains how immunotherapy works.
One of the tools we use to treat cancer is adoptive cell therapy. This technique is also called T-cell transfer therapy T-cell transfer therapy. One of the most effective types of adoptive cell therapy is CAR T-cell therapy. It is particularly helpful for patients who have not benefited from other treatments.
CAR T-cell therapy involves genetically engineering your T cells and then using them to recognize and kill your cancer cells.
We use these CAR T-cell therapies, approved by the U.S. Food and Drug Administration (FDA):
- Abecma for adults with relapsed/refractory multiple myeloma
- Breyanzi® for adults with large B-cell lymphomas
- Carvykti for adults with relapsed/refractory multiple myeloma
- Kymriah for patients up to 25 years old with acute lymphoblastic leukemia and adults with advanced lymphomas
- Tecartus ® for adults with advanced mantle cell lymphoma and adults with relapsed/refractory acute lymphoblastic leukemia
- Yescarta® for adults with advanced and relapsed/refractory lymphomas
At Mount Sinai, we also use CAR T-cell therapies and similar cellular therapies in clinical trials. We are testing them with relapsed/refractory multiple myeloma, a cancer of plasma cells. We plan to test CAR T-cell therapies in other blood cancers, such as leukemia as well as non-malignant conditions such as pemphigus vulgaris and viral hemorrhagic cystitis associated with allogeneic bone marrow transplants. Other areas of interest for cellular therapies include solid tumors, such as metastatic melanoma, urothelial cancers, breast cancer, and brain cancer.
CAR T-cell therapy and adoptive cell therapy are not available everywhere. You can only receive this state-of-the-art treatment at authorized cancer centers with highly specialized expertise, such as the Mount Sinai Tisch Cancer Center.
Clinical Trials
Our cutting-edge research offers more options and more hope. To learn about our cellular therapy clinical trials that are open to patient enrollment, you can contact us at crsu@mssm.edu or 212-824-7391.
Or you can search here for a list of cellular therapy clinical trials.
In the box that says SEARCH BY KEYWORD, TREATMENT NAME, ETC, type CAR T. Be sure to include a space between CAR and T.
Learn more about what joining a clinical trial may mean for you by visiting our clinical trials page.
CAR T-Cell Therapy Process
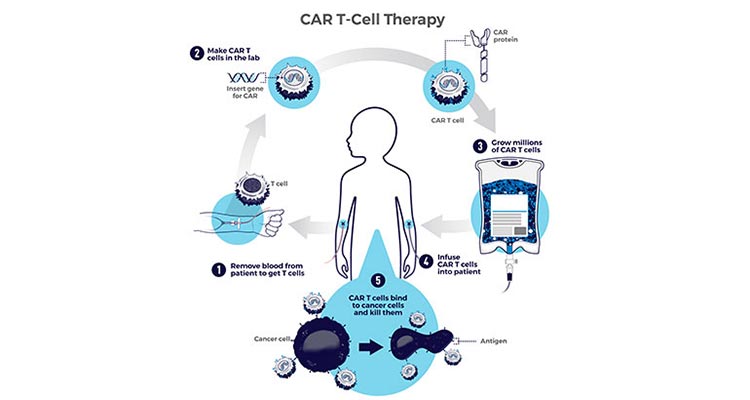
CAR T-Cell Therapy
This infographic shows the steps for creating CAR T-cell therapy, a type of treatment in which a patient's T cells (a type of immune system cell) are changed in the laboratory so they will attack cancer cells. Source: National Cancer Institute (NCI)
If you are a candidate for CAR T-cell therapy, we will review a detailed treatment plan with you so you know what to expect.
CAR-T CELL THERAPY PROCESS
The process is typically like this:
1. Pre-screening
To be considered for this treatment your doctor will evaluate you to determine if you are eligible to receive this therapy. We will review your full medical history, perform a physician exam, and check routine labs, the condition of your heart and lungs. Imaging may need to be performed to determine the status and extent of your disease. You will meet with our specialized CAR-T RN coordinator who will provide an extensive educational session as well as our dedicated CAR-T social worker who will assess any psychosocial needs prior to treatment. Once our doctors determine you are eligible to receive this therapy, you will be enrolled with the manufacturing company that produces the specialized T-Cells.
2. Collection of T cells
We collect some of your blood through an intravenous line. We process the blood with a machine that separates your T cells from the other blood cells. We save your T cells. We return the rest of your blood to your body. This process is called leukapheresis. It can take between three to six hours. Your cells will be immediately sent to the manufacturing company to begin the genetic engineering and manufacturing process
3. Engineering and manufacturing
Your own T-Cells are genetically modified by introducing DNA into them to produce a gene called chimeric antigen receptor (CAR) on the surface of the cells. These are now called CAR-T Cells which now can recognize the antigens on your cancer cells. Your cells are “expanded” by growing hundreds of millions of them in a laboratory which can take up to 21 days. Once the CAR-T cells are genetically modified and manufactured, the product is shipped back to our hospital for your infusion to help you fight your cancer by recognizing and attacking your cancer cells.
4. Bridging Therapy
After your T-Cells are collected and while your cells are being manufactured, you may need to receive one or two cycles of chemotherapy with your doctor to control your disease.
5. Conditioning chemotherapy
Right before receiving the engineered CAR T cells, you will receive conditioning chemotherapy, usually over a three-day period. The conditioning chemotherapy removes other immune cells and makes room for the engineered CAR T cells. This can occur in the inpatient setting or outpatient setting, depending on your disease status.
6. Infusion of CAR T cells
The infusion of your engineered CAR T cells is done through an intravenous line. The infusion takes about 30 minutes, and there are generally no immediate side effects. The infusion can occur in the inpatient or outpatient setting, depending on your disease status.
7. Monitoring
We want to be sure you are OK after this procedure. We will monitor you closely in the hospital for side effects. You may need to stay in the hospital for up to two weeks. Mount Sinai is a full-service health care system. This means we have experts in all the medical specialties you may need. Once you are discharged from the hospital, you will be closely monitored by your CAR-T doctor and team for the first few months. You may need to receive supportive therapy in between visits which is expected and normal. If you have an oncologist outside of Mount Sinai Hospital, we will closely communicate with them of your status and ensure continuity of care throughout your journey.
Insurance coverage for CAR T-cell therapy
If this treatment is right for you, we will work with your health insurance company to arrange for coverage.
Side Effects of CAR T-cell Therapy
Although CAR T-cell therapy can produce great results, you may experience side effects. The most common side effects are cytokine release syndrome (CRS), neurotoxicity, cytopenias, and hypogammaglobulinemia.
Cytokine Release Syndrome (CRS): T-cells release cytokines, which are chemical messengers that stimulate and direct the immune response. The release of cytokines indicates that the T cells are actively working against your cancer. CAR T-cells can prompt a large release of cytokines in the body, activating a systemic immune response which can cause high fever, chills, and changes in blood pressure, heart rate and oxygen. For this reason, you will need to be monitored closely for a two-week period in the hospital. We manage CRS with supportive care that may include drugs such as Actemra®, to block interleukin 6 (IL-6) activity, Anakinra (Kineret) to block IL-1 activity, and steroids.
Neurotoxicity: Patients can experience a range of symptoms including headache, confusion, difficulty speaking, and trouble with balance. In very rare cases, brain swelling and seizures may occur. Fortunately, there are effective treatments such as steroids and anti-seizure medications. Our neurologic specialist members of your care team--are available to assist as needed. In the majority of cases, symptoms are temporary and reversible.
Cytopenias: After receiving your CAR T-cell therapy, you may experience cytopenias, which are low blood counts of red blood cells, white blood cells, or platelets. It is important to monitor these blood counts while you are in the hospital and after discharge from the hospital in case you require transfusions, injections, or other support to help bring them back to normal levels.
Hypogammaglobulinemia: This occurs because CAR T cells target cancerous B cells but also destroy normal B cells, resulting in a decrease in antibody production. This can place you at risk for infection. Patients often receive intravenous immunoglobulin after their infusion of CAR T cells to provide protection from infections.
Cellular Therapy Services Team

Our dedicated team includes physicians, nurses, pharmacists, social workers, laboratory technicians, and other specialists who have expertise in cellular therapy.
- Keren Osman, MD, Medical Director
- Jacques Azzi, MD
- Uroosa Ibrahim, MD, Associate Medical Director
- Shambavi Richard, MD, Co-Lead Physician for Multiple Myeloma CAR-T Program
- Adriana Rossi, MD, Co-Lead Physician for Multiple Myeloma CAR-T Program
- Jason Batalha, MSN, NP-BC, Clinical Program Manager
- Cellular Therapy Lab
- The Apheresis Center
Contact our Team: 212-241-6021
