Upper Airway/Hypoglossal Nerve Stimulation for Obstructive Sleep Apnea
Obstructive sleep apnea (OSA) is a common common condition that affects more than 18 million Americans. It is characterized by several breathing pauses, ranging from 5 to 30 times or more per hour throughout a single night of sleep. These frequent, deep sleep interruptions can lead to morning headaches, reduced attention span, excessive daytime sleepiness, and even depression. More alarming, health issues associated with untreated OSA include cardiovascular and metabolic disorders, such as hypertension, stroke, and diabetes, as well as dementia and cancer.
Patients diagnosed with OSA are usually prescribed a CPAP machine, which stands for continuous positive airway pressure. While this is the medical gold standard treatment, studies have shown that up to 50 percent of OSA patients fail to adhere to this treatment. At Mount Sinai, our Division of Sleep Surgery was one of the first New York City hospitals to offer Inspire therapy , the only Food and Drug Administration (FDA)-approved upper airway/hypoglossal nerve stimulation treatment, as an alternative for these patients struggling or unable to comply with CPAP.
“This implant is a revolutionary therapy, as it stimulates the base of the tongue, offering a wider airway for patients’ intake of oxygen when they would otherwise obstruct,” says Chief of the Division of Sleep Surgery at the Mount Sinai Health System Fred Lin, MD. “It’s also unique in that it is implanted, so patients no longer need to apply masks that cover the nose and mouth at night. Patients struggling with CPAP now have an exciting, fresh therapeutic choice.”

Upper Airway Stimulation for When CPAP Fails
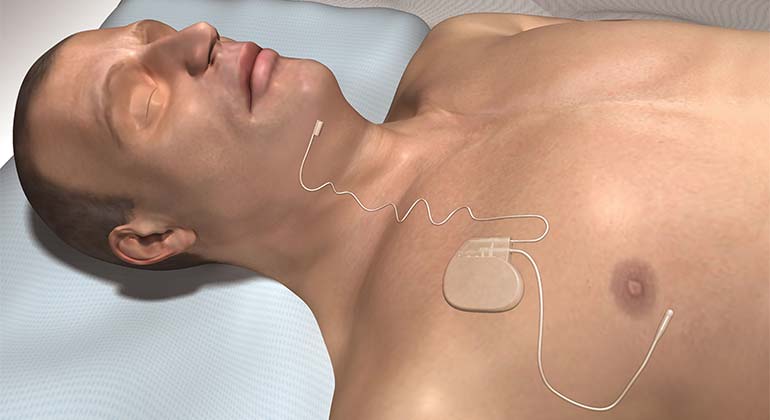
Approved by the FDA in 2014, Inspire therapy is designed to help patients who cannot tolerate CPAP. This system is comprised of three implantable components: a small programmable pulse generator, a breathing sensor lead, and a stimulator lead. Patients have full control of this device via a remote; they simply turn it on at night before bed and then off after awaking in the morning.
Watch the video to learn about how Inspire therapy can help.
Is Upper Airway/Hypoglossal Nerve Stimulation a Fit for You?
If you suffer from OSA and cannot tolerate the CPAP regimen, Inspire therapy might be the solution for you. Ideal candidates:
- Have a moderate to severe OSA
- Are unable to tolerate or benefit from CPAP
- Are not significantly overweight
- Are over the age of 18
- Have a BMI of <35 (may differ slightly with insurance)
To learn more about how Inspire can improve your sleep and quality of life, schedule a consultation by calling 212-241-9410.
Surgery and Follow-up Care
Implantation of the Inspire system is conducted in the operating room under general anesthesia as an outpatient procedure, which takes about 2-3 hours. Small incisions are made for the two leads and pulse generator. Patients generally experience some swelling at these incision sites after the surgery for a few days. However, they are able to return home the same day of the procedure or the next morning. Once the incisions have healed, the patient will return for device activation and receive education on how to operate the remote. After patients have used it for a couple of weeks, they undergo a titration sleep study at the sleep lab to optimize the device settings. Finally, patients are asked to return for a follow-up visit once a year for a simple device check.
Inspire Therapy Patient Outcomes
Results of the STAR trial, published in the New England Journal of Medicine, demonstrated that Inspire ™ therapy can significantly reduce sleep apnea events and improve quality of life measures. Specifically, data to date demonstrates there was a 78 percent mean reduction in sleep apnea episodes and 85 percent of bed partners reported little or no snoring.