Mount Sinai Researchers Identify Promising Drug Target for Depression
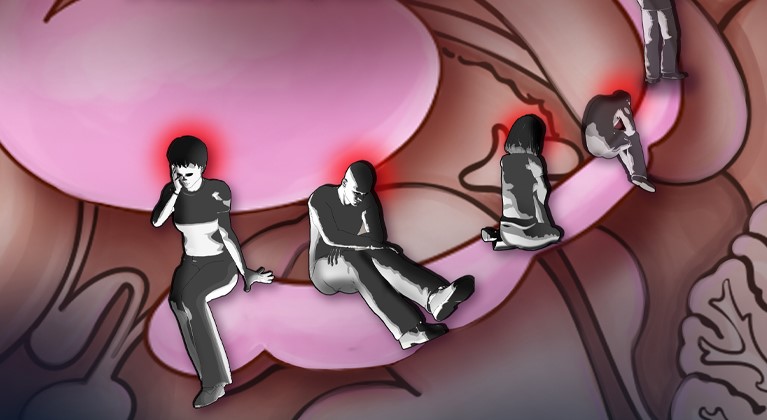
Decreased expression of a protein called Rac1 may be a primary cause of depression, according to a new study in human and mouse brains.
Decreased expression of a protein called Rac1 may be a primary cause of depression, according to a new study in human and mouse brains published in the journal Nature Medicine. Researchers from the Icahn School of Medicine at Mount Sinai also found that overexpression of Rac1 reversed depression-related behaviors in mice, indicating that this protein is a viable target for new therapies.
Scientists found that Rac1 was diminished in mouse brains after experiencing a chronic stressor, and in the postmortem brains of humans with major depressive disorder. By manipulating the expression of Rac1, they were able to control the depressive response in mice.
“Major depressive disorder affects millions of Americans, many of whom are no longer benefitting from currently available treatments,” said first author Sam Golden, a graduate student in the laboratory of Scott Russo, PhD, Assistant Professor of Neuroscience at the Graduate School of Biomedical Sciences at Mount Sinai. “There is a significant unmet need for new drug targets for treatment-resistant depression and for better understanding of the epigenetic underpinnings of this disease.”
Dr. Russo, Golden, and their team utilized an animal model in which mice were repeatedly subject to bouts of social defeat. They evaluated them 48 hours later to identify any changes in gene expression in the nucleus accumbens (NAc), a reward center in the brain. They found that Rac1 was significantly diminished, or down-regulated, in the brains of mice for 35 days after the chronic social defeat-stress ended. Rac1 was not affected by only one episode of stress, indicating that only prolonged stressors that induce depression are capable of down-regulating Rac1.
Golden next explored the epigenetic regulation of Rac1, or how the protein is expressed as a result of environmental factors. They found that social-defeat stress caused changes in chromatin, the structure that packages DNA, which caused the down-regulation of Rac1.
Dr. Russo and his team then evaluated postmortem human brain tissue to determine if their findings in mice were consistent in humans, and discovered that Rac1 was down-regulated in humans with major depressive disorder as well, and found similar epigenetic changes. In both mice and human brains, Rac1 was only normalized in half the subjects treated with antidepressants.
Armed with this knowledge, Golden manipulated Rac1 by either knocking out the expression of the protein or over-expressing it by using a process called gene transfer, in which Rac1 was encoded onto a modified version of the Herpes virus and infused directly into the NAc of mice. Knocking out Rac1 caused social defeat in both the genetically engineered mice and the mice who received the gene transfer. Mice receiving the gene transfer that overexpressed an active form of Rac1 were normalized after social defeat, indicating that an infusion of Rac1 may improve neuroplasticity in the brain.
“Our study is among only a few in depression research in which two independent human cohorts and animal models validate each other,” said Dr. Russo. “Rac1 has enormous therapeutic potential, and I look forward to investigating it further.”
Dr. Russo hopes to gain a better understanding of the role of Rac1 on the brain beyond the NAc, and to explore the possibility of biomarkers identified in blood tests to indicate dysfunction. After further investigation, he hopes to explore pharmacologic compounds that manipulate Rac1.
For more information, visit http://neuroscience.mssm.edu/russo/.
This study was supported by funding from the National Institutes of Mental Health (grants R01 MH090264, P50 MH66172, P50 MH096890), a division of the National Institutes of Health (NIH) and the Johnson and Johnson International Mental Health Research Organization Rising Star Award.
About The Mount Sinai Medical Center
The Mount Sinai Medical Center encompasses both The Mount Sinai Hospital and Icahn School of Medicine at Mount Sinai. Established in 1968, the Icahn School of Medicine is one of the leading medical schools in the United States, and is noted for innovation in education, biomedical research, clinical care delivery, and local and global community service. It has more than 3,400 faculty in 32 departments and 14 research institutes, and ranks among the top 20 medical schools both in National Institutes of Health (NIH) funding and by U.S. News & World Report.
The Mount Sinai Hospital, founded in 1852, is a 1,171-bed tertiary- and quaternary-care teaching facility and one of the nation’s oldest, largest and most-respected voluntary hospitals. In 2012, U.S. News & World Report ranked The Mount Sinai Hospital 14th on its elite Honor Roll of the nation’s top hospitals based on reputation, safety, and other patient-care factors. Mount Sinai is one of 12 integrated academic medical centers whose medical school ranks among the top 20 in NIH funding and by U.S. News & World Report and whose hospital is on the U.S. News & World Report Honor Roll. Nearly 60,000 people were treated at Mount Sinai as inpatients last year, and approximately 560,000 outpatient visits took place.
For more information, visit http://www.mountsinai.org.
Find Mount Sinai on:
Facebook: http://www.facebook.com/mountsinainyc
Twitter @mountsinainyc
YouTube: http://www.youtube.com/mountsinainy
# # #
About the Mount Sinai Health System
Mount Sinai Health System is one of the largest academic medical systems in the New York metro area, with more than 43,000 employees working across eight hospitals, over 400 outpatient practices, nearly 300 labs, a school of nursing, and a leading school of medicine and graduate education. Mount Sinai advances health for all people, everywhere, by taking on the most complex health care challenges of our time — discovering and applying new scientific learning and knowledge; developing safer, more effective treatments; educating the next generation of medical leaders and innovators; and supporting local communities by delivering high-quality care to all who need it.
Through the integration of its hospitals, labs, and schools, Mount Sinai offers comprehensive health care solutions from birth through geriatrics, leveraging innovative approaches such as artificial intelligence and informatics while keeping patients’ medical and emotional needs at the center of all treatment. The Health System includes approximately 7,300 primary and specialty care physicians; 13 joint-venture outpatient surgery centers throughout the five boroughs of New York City, Westchester, Long Island, and Florida; and more than 30 affiliated community health centers. We are consistently ranked by U.S. News & World Report's Best Hospitals, receiving high "Honor Roll" status, and are highly ranked: No. 1 in Geriatrics and top 20 in Cardiology/Heart Surgery, Diabetes/Endocrinology, Gastroenterology/GI Surgery, Neurology/Neurosurgery, Orthopedics, Pulmonology/Lung Surgery, Rehabilitation, and Urology. New York Eye and Ear Infirmary of Mount Sinai is ranked No. 12 in Ophthalmology. U.S. News & World Report’s “Best Children’s Hospitals” ranks Mount Sinai Kravis Children's Hospital among the country’s best in several pediatric specialties.
For more information, visit https://www.mountsinai.org or find Mount Sinai on Facebook, Twitter and YouTube.

Mount Sinai Launches Discovery and Innovation Center in Midtown West
Oct 05, 2023 View All Press Releases